Q: A solution contains Cr³+ and Mg2+. The addition of 1.00 L of 1.55 M NaF solution is required to…
A: By determining total mass and total no. of moles of a mixture, we can calculate the mass of…
Q: Calculate the pH of 0.2 mol/L of NH4+. NH4+ H+ + NH3 The Ka = 5.8 x10-10 for the above weak acid.…
A:
Q: 1. Barium chloride solution is mixed with potassium sulfate solution to produce solid barium sulfate…
A: A reaction is a balanced reaction if it contains equal number of atoms on both sides of the…
Q: Use the information page to give the acid name and the acid formula for each of the ate anions…
A: Anion NameAnion Formula
Q: How many of the following statements are true about the reaction below? 1 ~ 2 i. The name of the…
A: This an Friedal-Craft acylation reaction: Aromatic substitution reaction.In this reaction to the…
Q: 2. For each pair of the following compounds, identify which compound and conditions would react more…
A: Biomolecular nucleophilic substitution reaction (SN2 reaction) involves the attack of nucleophile…
Q: Vapor pressure (mm Hg) 900 800 700 600 500 400 300 200 100 Carbon disulfide 0 10 20 30 40 °C Methano…
A:
Q: Click the "draw structure" button to launch the drawing utility. What product is formed when the…
A: K2Cr2O7 is names as potassium dichromate. It behaves likes and oxidising agent. It will oxidise…
Q: For the simplest serial first order reactions A→ R→S; the concentration of component A -increases…
A: A first-order reaction is one in which the rate of the…
Q: Question 7 1 Name 2 3 4 Albuterol НО. НО. How many chirality centers are in the molecule shown?…
A: Chiral center - carbon having four different group attached
Q: Write the formula of the conjugate Brønsted-Lowry base, PH3 acid of the PH₂
A: Conjugate acid is a species that contains one more H atom (proton) than the base from which it is…
Q: Complete the reactions by drawing the structure of the O a) Ob) Oc) d) CIH O + H₂ :OH OH aOH FOH aoH…
A: -> H2/Pt can reduce aldehyde and ketone to alcohol but it can not reduce carboxylic acid.->…
Q: A student prepared 5.82 M solution of sodium sulfate, Na2SO4 (molar mass-142 g/mol) in a 1-L…
A: Sodium SulphateConcentration = 5.82 MVolume = 1 LNo. of moles = 0.206Molar mass = 142 g/mol
Q: Consider three isolated ions: Mg2+, O²- and Na*. Which is true about these ions? A. All of the ions…
A: Atomic radius is the distance between the central nucleus and the outermost electron. Atomic radius…
Q: The strength of a covalent bond depends upon the size of the atoms and the bond order. In general…
A: Bond energy is directly proportional to the bond order and inversely proportional to the bond…
Q: Reaction Kinetics and Asymptotic Behavior Suppose the formation of a product P depends on the single…
A: "Since you have asked a question in which third part requires an external link to visit, as per…
Q: A galvanic cell at a temperature of 25.0 °C is powered by the following redox reaction: 3 Cu2+ (aq)…
A:
Q: Current Attempt in Progress In an industrial process for making nitric acid, the first step is the…
A:
Q: Consider the molecule trans-1-ethyl-2-methylcyclohexane. What can you say about the most stable form…
A: -> For most stable form contains alkyl substituent at equatorial position to reduce 1,3-diaxial…
Q: 3. One component of the waste water that enters a waste water plant for treatment is urea, (NH2)2CO.…
A: The process involves the following steps:Urea hydrolysis: Urea reacts with water in the presence of…
Q: What is the formula for copper(I) oxide ? What is the formula for silver chloride ? What is the…
A: In ionic compound the total positive charge of the cation is equal to the total charge of the anion.
Q: Which compound will give a broad and intense band at 3100 cm-1 in its IR spectra? (a) Pentane (b)…
A: A question based on IR spectroscopy of organic compounds. 4 options are given from which the most…
Q: A weak acid, HA, has a K, of 1.00x105. If 0.100 mole of this acid is dissolved in one liter of…
A: Calculation of degree of dissociation (α) for weak acid (HA) :According to Arheenius' theory of…
Q: This is prostaglandin PGF2 ball & stick There are + labels carbon atoms in prostaglandin PGF2α-
A: The number of atoms can be easily determined by the line-angle formula. The Line-angle formula…
Q: A marine biologist is preparing a deep-sea submersible for a dive. The sub stores breathing air…
A: The width of the sphere, w = 69.0 cm Volume of air, V1 = 3900 L at a pressure (P1) of 1 atm
Q: 1. For the following molecule, draw its enantiomer as well as one of its diastereomers. Designate…
A: Enantiomer is mirror image of original structureDiastereomers are a subset of stereoisomers, which…
Q: Identify the following as soluble or insoluble. Na3PO4 AgBr CaCO3…
A:
Q: A nonmetal element in the third row of the periodic table exists as two allotropes (two different…
A: Given that, a nonmetal element has two allotropes. Among them, one has extended network and other…
Q: ify each of the molecules as one of the central electron geometries and molecular shapes. Molecule:…
A: -> electron geometry refers to arrangement of electron density around the central atoms. Geometry…
Q: Calculate the solubility of CuCO3 in water at 25 °C. You'll find Kp data in the ALEKS Data tab.…
A: From standard data, Ksp for CuCO3 = 1.4 × 10-10
Q: What is the name of the compound with the formula Cu₂O ? What is the name of the compound with the…
A: The inorganic compounds do not form by the combination of C and H only. There may be other atoms,…
Q: 5. Which of the following C-H bands has the highest energy vibration? A CH4 6. Which of these m B…
A: C-H bond vibrations in molecules can have varying energies based on the specific molecular context…
Q: Hydrogen peroxide, H2O2, is a strong oxidizing agent. It is used as an antiseptic in a 3.0% aqueous…
A:
Q: Question 15 10.50 mL of 0.100 mol/L NaOH(aq) is needed to neutralize 30.00 mL of a HCl(aq) solution.…
A:
Q: Which product would form for the reaction below? OH Me HB (aq) Br Me Me Me Br. Me Me Br ? Me Me O Me…
A:
Q: Please explain both questions
A: A periodic table is a tabular arrangement of chemical elements organized based on their atomic…
Q: A buffer solution is 0.337 M in H2CO3 and 0.239 M in KHCO3 . If Ka1 for H2CO3 is 4.2x10^-7 , what…
A:
Q: 1) Which of the following elements is a metal? A) nitrogen 2) Which of the following elements is a…
A: 1.nitrogen is a non metalic element Which belongs to group 15.2.fluorine is also a non metal which…
Q: A medical technician is working with the four samples of radionuclides listed in the table below.…
A: we have to complete the given table with respect to the radioactivity of the given samples
Q: 1. The reaction below gives four different aldol condensation products. Draw all four products. oli…
A: This is an example of self and crossed aldol reaction.
Q: Predict the product H3C H2 IO CH3 ОН CH2 [0]
A:
Q: What is true considering the bonding of atoms in clusters? O When atoms are bound in small clusters…
A: The correct answer is 1) When atoms are bound in small clusters (i.e., nanoparticles), their…
Q: Which of the selections completes the following nuclear reaction? 40 y+ 20 Ca Ca+ 20
A:
Q: Question A10 Complete the missing blank in the following question: Rf value is (a) (c) (d) Distance…
A: A question based on analytical processing. 4 options statements about Rf value are given, from which…
Q: Draw complete structural diagrams of the following compounds. (a) 2-methyl,pent-2-ene (b) propyne…
A: 2-methylpent-2-enePropyne2,3-dimethylbutaneCycloheptane
Q: 11. Methylation of sodium azide with trimethylsulfanium iun would give off gas: [a] dimethylsultide…
A: sodium azide (NaN3) undergoes methylation with trimethylsulfanium ion (Me3S+), it leads to the…
Q: Calculate either [H₂O*] or [OH-] for each of the solutions at 25 °C. Solution A: [OH-] = 2.13 x…
A:
Q: Choose the resonance structure that results from the arrow pushing scheme below:
A:
Q: How many neutrons are present in a 137Ba²+ ion? (a) 81 (b) 83 (c) 56 (d) 58
A: Given that: Element = 137Ba2+Mass number = 137
Q: The hydronium ion concentration in an aqueous solution at 25°C is 8.8×10-2 M. The hydroxide ion…
A:
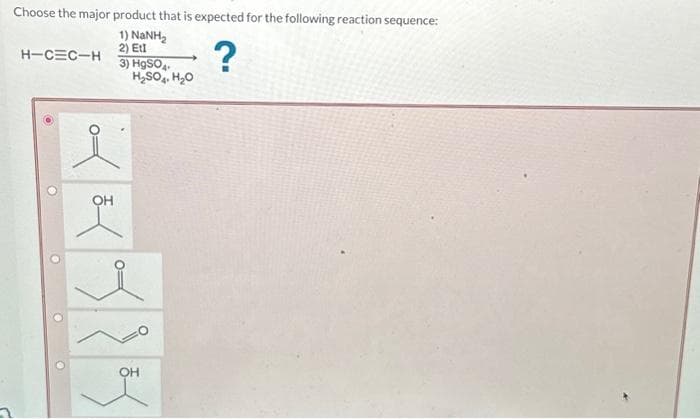
Step by step
Solved in 3 steps with 1 images

- Select the necessary reagent(s) for the following transformation. a. I2 b. H2/Pt c. Cu^2+ d. H+/H2O e. NaOH f. K2Cr2O7What order of reagents should be used to synthesize the following product from methylcyclopentane?(A) Br2(B) Br2, hv(C) KOH, heatWhat is the major product of the reaction of 2-methyl-2-butene with each of the following reagents? a. HBr b. HI c. Cl2/CH2Cl2 d. O3, −78 °C, followed by (CH3)2S e. H2/Pd f. MCPBA (a peroxyacid) g. H2O + H2SO4 h. Br2/CH2Cl2i. Br2/H2O j. Br2/CH3OH k. BH3 /THF, followed by H2O2, HO-, H2O
- We are asked to prepare (CH3)2CHCH2OH (2- methylpropan-1-ol) from an alkyl halide and any required reagents ?Predict the products of reaction of pent-1-yne with the following reagents. warm, concd. KMnO4, NaOHThese reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4 choices:A,DB,CA,B,CA,B,C,D
- Draw the favored product(s) of the following reactions (organic chemistry)Predict the product C of the following reaction below and briefly explain youranswer.[PtCl3(C2H4)]¯ + NH3 → CPredict the products from reaction of 2-hexyne with the following reagents:(a) 2 equiv Br2(b) 1 equiv HBr(c) Excess HBr(d) Li in NH3(e) H2O, H2SO4, HgSO4

