Chromate-dichromate system 1. Prepare a clean spot plate and label four wells. Place 5 drops of 0. 10 M K,Cro into wells 1 and 2, and 5 drops of 0. 10 M K_Cr.0, into wells 3 and 4. 2. Add 2 drops of 2. 0 M H ,SO̟ into wells 1 and 3. 3. Add 2 drops of 2.0 M NaOH into wells 2 and 4. 4. Observe the colors of the solutions. Well 1 2 3 Color of solution Predominant species responsible for color Give the reaction equilibrium that describes this system:
Chromate-dichromate system 1. Prepare a clean spot plate and label four wells. Place 5 drops of 0. 10 M K,Cro into wells 1 and 2, and 5 drops of 0. 10 M K_Cr.0, into wells 3 and 4. 2. Add 2 drops of 2. 0 M H ,SO̟ into wells 1 and 3. 3. Add 2 drops of 2.0 M NaOH into wells 2 and 4. 4. Observe the colors of the solutions. Well 1 2 3 Color of solution Predominant species responsible for color Give the reaction equilibrium that describes this system:
Chapter22: Bulk Electrolysis: Electrogravimetry And Coulometry
Section: Chapter Questions
Problem 22.30QAP
Related questions
Question
[C] *refer to the (2) photos below*

Transcribed Image Text:Chromate-dichromate system
1. Prepare a clean spot plate and label four wells. Place 5 drops of 0. 10 M K,Cro.
into wells 1 and 2, and 5 drops of 0. 10 M K¸Cr,0, into wells 3 and 4.
2. Add 2 drops of 2. 0 M H,SO̟ into wells 1 and 3.
3. Add 2 drops of 2.0 M NaOH into wells 2 and 4.
4. Observe the colors of the solutions.
Well
1
3
4
Color of solution
Predominant species
responsible for color
Give the reaction equilibrium that describes this system:
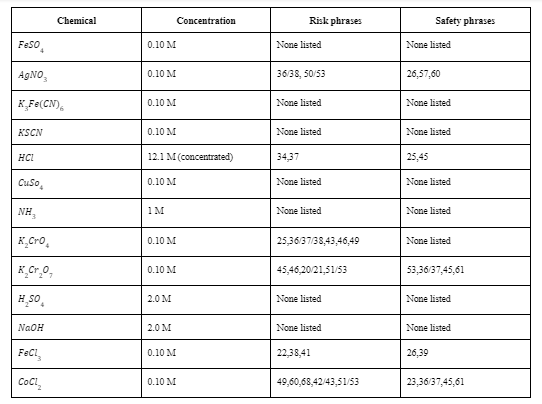
Transcribed Image Text:Chemical
Concentration
Risk phrases
Safety phrases
Feso.
0.10 M
None listed
None listed
AgNO,
0.10 M
36/38, 50/53
26,57,60
KĻF¢(CN),
None listed
0.10 M
None listed
KSCN
0.10 M
None listed
None listed
HCI
12.1 M (concentrated)
34,37
25,45
Cuso,
None listed
0.10 M
None listed
NH,
1м
None listed
None listed
K,Cro,
0.10 M
25,36/37/38,43,46,49
None listed
K_Cr.0,
0.10 M
45,46,20/21,51/53
53,36/37,45,61
H sO,
2.0 M
None listed
None listed
NAOH
2.0 M
None listed
None listed
FeCl,
0.10 M
22,38,41
26,39
CoCl,
0.10 M
49,60,68,42/43,51/53
23,36/37,45,61
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning