Q: Which is the least stable A. 'Br B. E. O Br ? Br O C. O Br D. O =0 O Br
A: The delocalization of electrons through pi bonds is simply called resonance. The resonance is driven…
Q: In the laboratory, a student dilutes 16.8 mL of a 6.40 M hydrochloric acid solution to a total…
A: • Before dilution,Concentration of HCl solution (M1) = 6.40 MVolume of HCl solution (V1) = 16.8 mL•…
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A: pKa of ethanol = 16.0pKa of hydrogen cyanide = 9.3
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting structure,…
A: Alkene reacts with HCl
Q: 1 hentary reaction N₂0(g) → N₂(g) + O(g) - k₁ N₂(g) + O₂(g) k₂. Suppose also k₁k₂. That is, the…
A: We have to find overall, rate and rate constant values.
Q: 2 moles CH₂Cl₂ AICI 3
A: In Friedel-Crafts alkylation in benzene ring, any alkyl group is attached with the benzene ring via…
Q: Suppose 0.452 g of sodium iodide is dissolved in 100. mL of a 14.0 m M aqueous solution of silver…
A: Mass of NaI = 0.452 gVolume of AgNO3 solution = 100 mLConcentration of AgNO3 = 14.0 M
Q: You are playing the role of a high school chemistry teacher. Provide an analogy to explain the…
A: The state of reaction at which the contraction of the reactants and products does not change with…
Q: Objectives Write the objectives of this experiment. 1. 2. 3. Prelab exercises 1. List the reagents…
A: objectives of the experiment:The objective(s) are what it is you are supposed to accomplish in the…
Q: What is the pH of a solution containing 1.033 mol L-¹ of a diprotic acid with PKA1 = 4.03 and PKA2 =…
A: HA concentration = 1.033 mol L-1pKa1 = 4.03pKa2 = 9.04
Q: Which of the following does not have an electron configuration ending with 2S^2 2P^6? chlorine ion…
A: we have to select the ion that does not have an electron configuration that ends as
Q: Draw both resonance structures of the enolate formed when the following ketone is treated with a…
A:
Q: 3. Draw the shape of the monoclinic structure of S. Describe the temperature and pressure conditions…
A: The monoclinic crystal system is described by vectors of unequal lengths, as in the orthorhombic…
Q: A 10 g sample of each substance is placed in a -20°C freezer for 10 minutes. Which sample undergoes…
A:
Q: What reagents are required to convert the alkene 1-propene to the alcohol 2- propanol? A) H₂ B) H.O,…
A:
Q: the following data were obtained from replicate 1.00g samples of cement dissolved in HCI and diluted…
A: To calculate the percent Cr₂O₃ in each sample and determine the absolute and relative standard…
Q: Choose the reagent(s) that would be most likely to complete this reaction. NBS Br A B C D E NBS Brz…
A:
Q: The trityl cation intermediate is a highly stable species due to the resonance stabilization…
A: Trityl cation also known as triphenylmethyl cation is very stable because the positive charge on…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. HO Na2Cr2O7 H2O, CH3CO2H…
A:
Q: What is the pH of a 3.6 x10^-2 M Mg(OH)2 solution assuming 100% dissociation
A:
Q: ¹₂(g) + ICl(g) → HI(g) + HCl(g) HI(g)+ ICI(g) 1₂(g) + HCl(g) Suppose also k₁«k₂. That is, the first…
A:
Q: 1. From the following enthalpy changes, S (s) + 3/2 O₂ (g) → SO3(g) 2 SO₂ (g) + O₂(g) → 2 SO3 (g)…
A: Hess's law states that if a process can be expressed as the sum of two or more steps, the enthalpy…
Q: Draw the product of this reaction. Ignore inorganic byproducts. H D 1. NaNHz
A: Alkynes are the hydrocarbons that contain carbon-carbon triple bond. These may be classified as…
Q: Curved arrows are used to illustrate the flow of electrons. ollow the arrows and draw the…
A:
Q: 8. How many unique protons are present in each of the following compounds? CI A) I=6; II= 3; III = 6…
A:
Q: Hydride Reactions xod ali ni aslidqortools diw taas ao sinon Review: Provide a mechanism for the…
A: In the given question there is formation of alcohol from benzaldihyde with reagent LiAlH4. now we…
Q: The value of Kc for the reaction below is 8.72 at 487 K. 2NO2(g) ---->N2O4(g)…
A:
Q: PROBLEMS What reagents would you use to accomplish each of the following transformations: 13.22 -X +…
A: Conversion of alkene to recemic mixture of less substituted alcohols.
Q: CaCO3(s)→CaO(s)+CO2(g) Express your answer as a chemical equation. Identify all of the phases in…
A: Concept of writing the balanced chemical equation in which number of atoms of each elements in…
Q: Given that the acid ionization constant of hypochlorous acid is 2.8 x 10-8, in which pH range is an…
A: Given,The buffer solution: HOCl/ NaOClKa of HOCl = 2.8 x 10-8
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Assume that the water side…
A: In this reaction, when ketone react with hydrazene we get hydrazone as the major product.This…
Q: A reducing agent can be described as a substance that loses electron and causes reduction gains…
A: As we know,Reducing agent is a substance that cause reduction & can lose electrons and be…
Q: A chemist carefully measures the amount of heat needed to raise the temperature of a 0.85 kg sample…
A: Molar heat capacity = q/n∆T q = heat absorbedn = number of moles of substance∆T = T2 - T1 T1 =…
Q: Draw the major 1,2- and 1,4-addition products obtained in the reaction shown. Assume that both are…
A:
Q: Organic Compounds: Alkanes 249 Questions and Problems Q5 Draw the condensed structural formula for…
A: We have to write the condensed structural formula.
Q: Provide the major organic product of the following reaction sequence. 1. PHCOCI, AICI 2. Zn(Hg), aq,…
A:
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. H₂ Raney Ni Drawing H OL
A:
Q: ALKYLATION AND ACYLATION Br FeBr3
A: The reaction in which benzene reacts with alkyl halides in the presence of lewis acids to produce…
Q: Draw the skeletal (line-bond) structure for 2-isopropylheptanoic acid. Drawing
A: A representation of molecular structure in which covalent bonds are represented with one line for…
Q: BONUS! Draw the Newman projection of the following molecule as viewed along the C=O bond (it may be…
A: Mechanism of the organic reactions
Q: Formal stability evaluations of OTC drug products should be conducted using in an FDA registered…
A: Formal stability evaluation of over-the-counter (OTC) drug products should be conducted using…
Q: For the following reaction 6.0 moles of H2 were combined with 4.5 moles of Cl2. The reaction was…
A:
Q: Suppose 13.5 g of barium acetate is dissolved in 300. mL of a 0.20 Maqueous solution of ammonium…
A: Details
Q: ОН heat
A:
Q: Use standard reduction potentials to calculate the standard free energy change in kJ for the…
A:
Q: The formation of hydrophobic interactions are the primary driving force behind aggregation of…
A: Micelles are formed by a cumulative formation of amphipathic molecules in an aqueous solution. In…
Q: One electron occupies each of several orbitals at the same energy before a second electron can…
A: One electron occupies each of several orbitals at the same energy before a second electron can…
Q: O N H catalytic H₂SO4 H₂O heat
A:
Q: a summary of the positive and negative effects of the oil and gas industry in Alberta how your…
A: Positive Effects:Economic Contribution: The oil and gas industry plays a significant role in…
Q: Balance the reaction between Hg2+ and SO₂ to form Hg and SO4²- in acidic solution. When you have…
A:
What are the major products(s)?
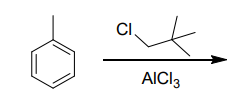
Step by step
Solved in 3 steps with 2 images


