Q: Given the following lewis structures... 1) Identify the most electronegative and least…
A: 1) As both the atoms are the Nitrogen (N) atoms, that is both are same. Therefore, there is no most…
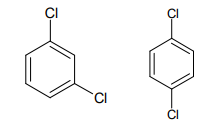
Q: Which of the following would you expect to have the higher boiling point? Explain why. OH 3.
A:
Q: What causes the dipole-dipole force in molecules? O a. Small differences in the electronegativity in…
A: Dipole dipole interactions are between two dipoles. Means permanent polarity is there.
Q: Determine the shape of the following molecules and identify what intermolecular forces exist in a…
A: a) SO2Cl2 = The structure of the given compound is, The shape of the compound is Tetrahedral. The…
Q: what intermoleculor forces does CS2 have
A: Interpretation - To tell what intermoleculor forces does CS2 have - Introduction-…
Q: What kind of intermolecular forces act between a hydrogen cyanide (HCN) molecule and a chlorine…
A: Intermolecular forces are the attractive or repulsive forces that operate between opposing particles…
Q: Given the following lewis structures... 1) Identify the most electronegative and least…
A: The electroneagtive element can be determined by a trend in the periodic table i.e element with…
Q: A molecule. force such as occurs between atoms in the same a) intermolecular, covalent bonding O b)…
A: Intramolecular forces are the forces that hold atoms together with in the molecule. Intermolecular…
Q: What are the network covalent atomic solids are composed of?
A:
Q: Explain the difference between nonpolar molecules andpolar molecules.
A: The covalent bond among atoms or elements is the foundation of chemical reactions. It is the process…
Q: what intermolecular forces can be expected between NH3 and a molecule of HF
A: We would check the type of molecules and Nature of forces can exist between them .
Q: Given the following lewis structures... 1) Identify the most electronegative and least…
A: 1) The most electronegative element is O and the least electronegative element is S. 2)
Q: Which of the following ionic solids has the largest lattice energy? O Sro LICI O KCI RBCI O Bao
A: We have to choose solid having highest lattice energy.
Q: Identify the most probable intermolecular force of attraction in the following: NaCl -----------…
A: Since you have posted a question with multiple subparts, we will solve first three subparts for you.…
Q: Which intermolecular force correlates with dipole moment?
A: Polarity is the property of liquid which shows that any liquid that has a high dipole moment are…
Q: Electron group Name the Molecular Total valence Bonding Polar or Intermolecular Arrangement *…
A: Molecular Geometry: The molecular geometry of any molecule is determined by the bond pair as well as…
Q: For each of the molecules or ions below, (1) draw Lewis dot structure (Remember the Octet Rule),…
A: Electron dot structures of molecule or ions : 1. CH3Br : The molecule has total 14 valence…
Q: ns as the polarity of the molec
A: Given What happens to the degree of dipole-dipole interactions as the polarity of the molecules…
Q: Given the following lewis structures... 1) Identify the most electronegative and least…
A: Given molecule is hydrogen peroxide To choose most and least electronegative element To show Delta…
Q: Predict the intermolecular forces of attraction in the following 1. HF 2. CH3CO2H
A:
Q: Calculate the lattice energy of ionic solid MX, given the following thermodynamic data: M(s) + X2(9)…
A: ΔH(n→n)=ΔH"Sub"+1/2ΔH"dis"+ΔH(E⋅A)+ΔH(J.E)+ΔHLE ΔH(L-E)=4H(n×2)-ΔHsent+1/2ΔH(dij1)+ΔH(E⋅A+)AH(I.E)…
Q: (f) Na2SO4(s) O covalent bonding O H-bonding O ionic bonding O dipole-dipole O London forces
A:
Q: e the attracti orce described and ar Figure 1 H H H H---N:---H---N: H--N:---H ---O: H H H --D 00 B -…
A:
Q: (a) State whether or not the bonding in each substance islikely to be covalent: (i) iron, (ii)…
A: Iron (Fe) is a metal that forms metallic bond in which electrons are free to move from one atom to…
Q: why are H bonds the strongest example of a dipole dipole bond?
A: The H-bonding is a special type of dipole-dipole interaction. In some of the molecules containing H…
Q: Which substance do you expect to have the greatest lattice energy: MgF2, CaF2, or ZrO2? Why?
A: The lattice energy gets increases when charges of ions are increased. It depends on the charge…
Q: e B. water C. sodium chl
A: Intermolecular forces exist between the molecules which are close to each other while moving around.…
Q: Solid ionic molecules are not electrically conductive due to the free movement of their ions
A: Ionic solid: Such solids are formed by the 3-dimensional arrangements of cations and anions bound by…
Q: Should H2S or COCl2 have the higher boiling point? Briefly explain why
A: Boiling point of a compound depends upon which type of forces binding the constituent molecules,…
Q: Define the term Ion–Dipole Interactions?
A: The term ion-Dipole interactions has to be defined.
Q: in
A: The London dispersion force is the weakest intermolecular force. The London dispersion force is a…
Q: Is lactose an ionic or covalent compound? What type of intermolecular forces exist in lactose
A: lactose is a covalent compound.
Q: The type of chemical bond in which there is a complete transfer of electrons from one atom to…
A: option C is correct - an ionic bond is formed option a polar covalent bond is formed by sharing…
Q: Use electronegativit ies to predict the direction and magnitude of the dipole moment in a carbon…
A: The direction and magnitude of the dipole moment in a carbon monoxide has to be given,
Q: State 2 conditions necessary for the formation of a dative or coordinate bond.
A: NOTE : Since you've posted multiple questions, we'll solve first question for you. If you want any…
Q: This sketch of a neutral molecule is shaded red or blue wherever the electrostatic potential at the…
A: From figure, we can see + ion : bigger cation. - ion : smaller anion. (a) HI ==> Smaller…
Q: а. COH HO H,C O,N I II b. LO HO H,C I II
A:
Q: There are three flask they are all 1.00L flask a is NH3 flask B is NO2 flask c is N2 which flask are…
A:
Q: Which compound will be more likely to exist as a hydrated solid? O KCl O CuSO4
A: Some compound when comes in contact with water vapors present in the atmosphere absorbs them into…
Q: CI HO
A: 1. Given compounds,
Q: 58. This picture is an example of: " Vander Wals Force of Attraction O Dipole-dipole Interaction O…
A: The attraction forces between the different molecules are known as intermolecular forces. Different…
Q: NH3 & H2O a. illustrate the lewis dot structure b. polar or nonpolar?
A: a. To write the Lewis dot structure we should know the total number of valence electrons b.…
Q: Use the thermochemical expression PCl3(g) + Cl2(g) ——-> PCl5(g) delta r G° = -37.2 kJ/mol…
A: Consider the given information is as follows; PCl3(g) + Cl2(g) → PCl5(g)…
Q: KBr has a lattice energy -671 kJ/mol . Consider a hypothetical salt XY. X2+ has the same radius of…
A: Given data, Lattice energy of KBr = -671kJ/mole The lattice energy of an ionic salt is given by…
Q: Discuss the basis of certain properties of substances through the concept of intermolecular forces…
A: Discuss the basis of certain properties Of substances through the concept of intermolecular forces…
Q: Direction: Complete the table by writing all types of intermolecular forces (IMF) that will exhibit…
A: Hydrogen bonding can be formed between a hydrogen atom and F, N and O atoms.
Q: Consider the molecule KrF2. What is the: a) VSEPR notation b) molecular geometry c) bond angle d)…
A: VSEPR - This theory is used to predict the arrangement of electron pairs around the central atoms in…
Q: Each molecule listed below is formed by sharing electrons between atoms when the atoms within the…
A: Please find your solution below : 34.
Will the two have the same dipole moment? why or why not? Express in terms of their structure and intermolecular force.
Step by step
Solved in 2 steps with 2 images


