Q: Which compound in set undergoes more rapid solvolysis when refluxed in ethanol? Show the major pro...
A: Concept introduction: SN1 reaction: The displacement of atom or group by nucleophilic is known as nu...
Q: Give an example of suspension?
A: A homogeneous mixture of two or more than two particles which get dissolved into each other.
Q: Balance each of the following redox reactions occurring in basic solution. H2O2(aq)+ClO2(aq)→ClO−2(a...
A:
Q: Write the formula for ionic compound. Lead(II) chromate
A: The type of chemical bonding that consists of the electrostatic interaction of opposite charged ions...
Q: 16.48
A: SOLUTION: Step 1: 16.48 Given that pH = 10.05 POH = 14 – pH = 14 – 10.05 = 3.95.
Q: 46). Calculate the solubility of Pbl2 in a solution that already contained 0.10 M Nal. Ksp 3D 9.8 х ...
A: Since NaI is completely soluble. Hence it will dissociate completely as NaI ----> Na+ + I- Hence...
Q: 4. How is the temperature of the gas determined during an experiment? Group of answer choices A) By ...
A: In this experiment, it is required to measure the temperature of water bath and furnace by various i...
Q: The student Louise continued the analysis of acetic acid using half - neutralization technique. She ...
A: pH of any solution is given by pH = -log[H3O+ ] where [H3O+ ] = molar concentration of H3O+
Q: What is the formal charge of carbon in carbon monoxide (CO) when drawn with a triple bond? Multiple ...
A: Formal Charge is the charge assigned to the atom in the respective molecule pre assuming that the el...
Q: Name ionic compound containing a polyatomic ion. Fe(OH)3
A: Polyatomic ion is the ion having more than two atoms which are covalently bonded to each other and h...
Q: The pH of a household ammonia solution is 11.75. a.) Write the acid or base dissociation equation fo...
A: The pH of a household ammonia solution is 11.75. a.) The acid or base dissociation equation for ammo...
Q: How do the molecules of a compound differ from the moleculesof an element?
A: Molecules are group of atoms where the atoms are held together by a chemical bond. They can result i...
Q: Does p-d mixing in electronic spectrum of [Mn(Br)4]2- occur due the pi-donating bromine ligand . He...
A: Answer : Part 1: Yes, p-d mixing in electronic spectrum of [Mn(Br)4]2- occur due to the pi-donating...
Q: Aspirin is composed of 60.0% carbon, 4.5% hydrogen, and35.5% oxygen by mass, regardless of its sourc...
A: Given compound is aspirin. It is said to contain 60.0 % carbon, 4.5 % hydrogen and 35.5 % oxygen.
Q: What volume of water is required to make up 250ml of 0.5 NAcl solution by dilution of a 10.OM Nacl s...
A: Since the process is dilution. Hence only water is being added to 10.0 M NaCl solution. Hence moles ...
Q: Draw structural formulas of all chloroalkanes that undergo dehydrohalogenation when treated with KOH...
A:
Q: Chemistry Question
A: Answ
Q: Chemistry Question
A:
Q: Convert the following
A:
Q: Which of the following groups least electronegative? Group 1 Group 18 Group 17 Group 2 G...
A: Generally the electronegativity follows the trend that in increases as we move across period and dec...
Q: 30). Identify the Lewis acid in this reaction. Zn+2 + 4NH3 ---> Zn(NH3)4*2 a. Zn+2 b. NH3 c. Zn(NH3)...
A: The reaction given is
Q: Using data in the following image. Please determine the reaction order with respect to each reactan...
A:
Q: D and H please
A: PBr3 is a source ob bromide ion, therefore it is called as a brominating agent. CrO3 is an Oxidizi...
Q: A 12-oz vanilla milkshake at a fast-food restaurant contains547 Calories. What quantity of energy is...
A: 1 calorie = 4.2 joules
Q: If you can please show work and be clear which one will be 1(slowest)-4(fastest) thank you
A: The average speed of a gas depends on the number of collisions between gas molecules and does not de...
Q: The structure of vitamin Bs, pantothenic acid, is shown below. Highlight and identify all the functi...
A: Please find below the functional groups present in Vitamin B5 as shown below. 1) COOH : Since the c...
Q: Chemistry Question
A: a. When we talk about the major source of energy in the diet the first position is of the carbohydra...
Q: Heavy metal azides, which are salts of hydrazoic acid, HN3, are used as explosive detonators. A solu...
A: The dissociation reaction of HN3 is => HN3 --------> H+ + N3-
Q: Predict the major organic product of the following reaction sequence. Styrene (1.53 g, 0.011 mol) in...
A: The reactant is styrene which is reacted with methanol and mercuric acetate. After that sodium hydro...
Q: A student measured a 6.790 g sample of a hydrated salt. After heating, the mass of anhydrous salt wa...
A: a) Given that weight of hydrated salt is 6.790 g and mass of anhydrous salt is found to be 4.501 g. ...
Q: Consider the following statement in reference to SN1, SN2, E1, and E2 reactions of haloalkanes. To ...
A: This comes in SN1 and E1 reactions. In case of bulky base or strong base with haloalkane, the mechan...
Q: 1-For the reaction, 4 Cl- + Co(H2O)62+ <--> CoCl42- + 6 H2O (color 1) ...
A:
Q: Which of the following molecules has the smallest dipole moment? h2o HCl CO3 NH3
A: Given data : Molecule that has the smallest dipole moment need to be write. H2O HCl CO2 NH3 Dipole M...
Q: Imagine a reaction that has reached equilibrium an din which the forward reaction at equilibrium is ...
A: Assuming the reaction is A ----> B
Q: Draw an MO energy diagram and determine the bond order for the N2 + ion.
A: MO diagram : Molecular orbital will form from linear combination atomic orbitals. Total number of a...
Q: 1-What is the pH of the blood?Include all digits given on the digital scale. 2-In the blood, Is th...
A: 1) Blood is in slightly basic range with pH = 7.4 approx Hence the pH of the blood is 7.4
Q: The structure of estrone, a female sex hormone, is shown below. Highlight and identify all of the fu...
A: The structure of estrone given is Hence the below shown functional groups present in it. 1) C=O :...
Q: How are physical properties different from chemical properties?
A: Difference between physical properties and chemical properties:
Q: what will change the value of an equilibrium constant?
A: Value of an equilibrium constant is changed by??
Q: By what factor would a scuba diver's lungs expand if she ascended rapidly to the surface from a dept...
A: Given: The depth of the surface is 134.5 ft The density of sea water is 1.01 g/mL The density of Hg ...
Q: 1-Does tap water conduct electricity? Group of answer choices yes no No answer text provided. No an...
A: 1-Does tap water conduct electricity? Tap water contains some amount of dissolved salts in it. These...
Q: 16.84
A: SOLUTION: Step 1: (a) AlCl3 acts as Lewis acid. Aluminium has 6 electrons in its valence shell, and ...
Q: What is the product of the reaction when α-amylase acts on amylose? (A) amylose molecules (B) mon...
A: Amylose is obtained by hydrolysis of starch. Hydrolysis of starch gives amylose and amylopectin. Amy...
Q: Provide the IUPAC name from stilbene dibromide.
A:
Q: Which of the following compounds has the lowest boiling point? `OH IV Multiple Choice || IV II
A: Boiling point of any molecule depends on 2 things ( in the same priority order as they are mentioned...
Q: 16.78
A: (a) Kb for ammonia is 1.8*10^-5 Kb for hydroxylamine is 1.1*10^-8
Q: Calculate the amount of H2S in cubic meters measured at 49°C and at a pressure of 0.2 barG, which ma...
A: Hydrogen sulphide (H2S) can be prepared by action of FeS with dilute hydrochloric acid (HCl). The eq...
Q: 1. H2, Pd/CaCO3, quinoline 2а. ВH;:THF 2b. H2O2, NaOH
A: The given reaction sequence produces a less substituted alcohol.
Select the member of each pair that shows the greater rate of SN2 reaction with KI in acetone.
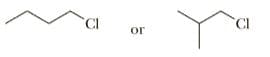
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images


