Q: How many molecules are in sample? 389 g CBr4
A: Given information, Mass of CBr4 is 389 g
Q: How many moles of a gas are present if, when the gas is collected over water at 24 oC, it has a tota...
A: According to Dalton's law, the total pressure of gaseous mixture is equal to the sum of partial pres...
Q: Propose a mechanism to account for the following transformation. OH H,SO, -H,0 ОН
A: A mechanism for the transformation has to be proposed
Q: The following reaction is completed in a sealed container: 2AB(g) ↔ A2(g)+B2(g) Use Le Chatelier's P...
A: Since we are entitled to answer up to 3 sub-parts, we’ll answer the first 3 as you have not mentione...
Q: Calculate the emf of the following concentration cell at 25°C: Cu(s) / Cu**(0.043 M) || Cu**(1.400 M...
A: EMF of the cell can be calculated by using the Nernst equation: Where, E= cell potential E0= cel...
Q: A. What mass of Ba(OH)2 is needed to make 250 ml of 0.155 m Ba(OH)2(aq) B. What if 25 ml of the 0....
A: NOTE : since density of solution is not mentioned in the question and volume of solutions are given,...
Q: -5 %3D A chemistry graduate student is given 450. mL of a 0.30 M acetic acid (HCH,CO,) solution. Ace...
A: Acetic acid is a weak acid. Hence dissolving NaCH3CO2 i.e salt of acetic acid in the acetic acid sol...
Q: What is AH° for the following reaction? 2C,H>(g) + 50,(g) → 4CO2(g) + 2H20(1) Substance AH®, (kJ/mol...
A: ∆H0 for the reaction will be 2 C2H2(g) + 5 O2(g) -------------> 4 CO2 (g) + 2 H2O(l) Given, ∆fH0...
Q: Name compound
A: Given structure of the compound,
Q: (D-H)
A: d) Since the molecule given is having a COOH group which is carboxylic acid functional group as show...
Q: According to the equation below, the partial pressure of SO2Cl2 at 3.0 x105s is _______ atm.
A: The given data are: Rate constant k = 3.0 x 10-6 s-1, Po = 3.3 atm and t = 3.0 x 105 s We have to fi...
Q: Aluminum hydroxide reacts with stomach acid (hydrochloric acid). Calculate the molarity of the stoma...
A:
Q: 1. i)Give the structure of the product from the reaction of propanal with 1M ethanol in dry acid . (...
A: Organic compounds are the compounds which are mainly composed C and H atoms. The branch of chemistry...
Q: Original gas under has a pressure of 4.40 atm at 25.0°C. At what temperature in Celsius will it reac...
A: According to Gay-Lussac's law, the pressure and temperature of an ideal gas are directly proportiona...
Q: Given the following rate law, how does the rate of reaction change if the concentration of X is doub...
A: Given: Rate Law: Rate = k [X][Y]2 Concentration of x is getting doubled.
Q: Draw structural formulas for the alkenes formed on acid-catalyzed dehydration of each alcohol. Where...
A: In acid catalyzed dehydration of alcohols, alkenes are formed.
Q: the compound can be synthesized by an SN2 reaction. Suggest a combination of haloalkane and nucleo...
A: In SN2 reaction, the rate of substitution reaction is depended on both nucleophile and substrate. Th...
Q: Chemistry Question
A: Compound NaI is neutral because in aqueous solution it will give strong acid (HI) and strong base(Na...
Q: A sample of potassium nitrate (49.0 g) is dissolved in 101g of water at 100˚C, with precautions take...
A: All known compounds can be dissolve in certain solvents whereas they remain insoluble in other solve...
Q: The K, for benzoic acid is 6.5 x 10. Calculate the pH of a 0.13 M benzoic acid solution.
A: Benzoic acid i.e HC7H5O2 is a weak monoprotic acid. Hence it can be presented as HA. Hence the parti...
Q: What is the e/m ratio of an electron?
A: e = charge of an electron, m= mass of electron J.J.Thomson was able to determine the e/m ratio by h...
Q: How much heat is released at constant pressure if a 15.0-L tank containing 47.0 atm of hydrogen sulf...
A: Here, P=47.0 atm, V = 15 litre R = 0.0821 L atm / Kmol T= -60 + 273 = 213K
Q: Complete the following acid-base reaction . Show all valence electrons on the interacting atoms and ...
A:
Q: What is the mass in grams of 1.00 gal of water? The density of water is 1.00 g/mL.
A: 1 gallon of water has a volume of 3.785L. Convert 1.00 gal into milliliters.
Q: What is the product of the following reaction equation after balancing? If no reaction state that " ...
A:
Q: a) Define vapor pressure of a liquid, b) how does vapor pressure vary with temperature?
A: a) Vapour pressure of any liquid is defined as the partial pressure of vapours of that liquid presen...
Q: 6b-please see attached
A: The species van’t Hoff factor value may be used to examine the maximum range to which the species ca...
Q: I only have 30 minutes to answer so please help me out if you can, I REALLY appreciate it!!! :) The ...
A: Stronger is the inter-molecular forces between the molecules, less will be the distance between the ...
Q: write UNCERTAINTY IN MEASUREMENT
A: Uncertainty as used here means the range of possible values within which the true value of the measu...
Q: Draw structural formulas for the major organic product reaction
A: A primary alcohol is oxidized to a carboxylic acid by chromic acid.
Q: Ribose, a carbohydrate with the formula shown, forms a cyclic hemiacetal, which, in principle, could...
A: Ribose, a carbonhydrate with the formula shown, forms a cyclic hemiacetal, which in principle could ...
Q: Is oxymetazoline acid or base?
A: Oxymetazoline is a member of the class of phenols.
Q: Butanol is treated with H2SO4 and heat. Identify the reaction and give the product that can be forme...
A: When alcohols are treated with H2SO4 and provided heat, then elimination reaction takes place and fo...
Q: How does the energy of three photons from a blue light source compare with the energy of one photon ...
A: The relationship between the energy of photon and wavelength is given by where E = energy of each ...
Q: Write Postulates of Dalton’s atomic theory.
A: Atoms and molecules are the building blocks of matter. Various experiments performed to find out the...
Q: Which of the following is NOT a form of potential energy? Group of answer choices Compressed spri...
A: Potential energy is the energy of the object by virtue of its position. Potential energy is the ener...
Q: Container of oxygen has a volume of 349 mL at a temperature of 22.0°C what volume will the gas occup...
A: According to Charles' law, Volume and temperature are directly proportional to each other. That is w...
Q: A solution is prepared at 25 °C that is initially 0.35 M in propanoic acid (HC,H,CO,), a weak acid w...
A: Given: Concentration of propanoic acid = [HA] = 0.35 M Concentration of potassium propanoate = [A-] ...
Q: A first order reaction has a rate constant of 1.3x10-8s-1.How long in minutes will it take for the r...
A: Given, first order rate constant k = 1.3 x 10-8 s-1 Initial concentration = 0.8 M ...
Q: An organic compound A is widely used as a preservative in pickles and has amolecular formula C2H4O2 ...
A: Given: Molecular formula of compound A = C2H4O2 When compound A reacts with ethanol, it forms a comp...
Q: Identify the Bronsted-Lowry acid and base? K2Cr2O7 + H2SO4 ⟷ H2Cr2O7 +K2SO4
A: According to Bronsted-Lowry acid and base theory, An acid is a substance which can donate a proton. ...
Q: What intermolecular forces is/are in the following molecule? H.
A: The molecule given is a covalent molecule. Hence there are no ions present in the solution. Hence th...
Q: Determine the quantity (g) of pure CaCl2 in 7.5 g of CaCl2•9H2O. Show your work.
A:
Q: Balance First five only
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and s...
Q: 0.26g CH4 are burned and all the heat added to a silver cup with 90g H2O(l) inside. If the water was...
A: The combustion reaction of CH4 is => CH4 + 2 O2 --------> CO2 + 2 H2O Since the ΔH of reaction...
Q: Determine the pH of a 0.62 mol L-1 NH4NO3 solution at 25oC. The Kb for NH3 is 1.76 x 10-5. Select ...
A: Ammonium nitrate is the salt of weak base and strong acid. When it is dissolved in water it dissoc...
Q: 4d-please see attached
A: since question first is incomplete molality of solution is not given so I am calculating second ques...
Q: Name ionic compound. Cr2S3
A: It is an inorganic compound. It is a brown-black solid.
Q: Write a short paragraph describing chemical bonding according to the Lewis model, valence bond theor...
A: Lewis model: Lewis model is based on octet rule. This model explains each atom have a tendency to co...
Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol
more rapidly
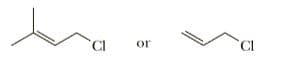
In SN1, the rate determining step is formation of carbocation and order of the substrate that favored in SN1reaction is 30 > 20 > 10.
Step by step
Solved in 2 steps with 1 images


