Q: d. O D H* HO, HO. OCH3 H, H2O HO. e.
A: The reaction in the presence of proton can undergo rearrangement via carbocation formation. For e.g-
Q: Propose a mechanism for the following reaction CH3 CH3 1. (CF3CO0'),Hg, H2O CH3-CH CH CH2 CH3-CH CH…
A: Given is oxymercuration demercuration reaction.
Q: S.7. Complete the following reaction with its mechanism. wm www www isi benzaldehit + KCN + E1OH →
A: When benzaldehyde which don't have α- hydrogen reacts with KCN in presence of EtOH it will form…
Q: он HO ONa 1. CO, A. P 2. H,SO, NaOH он
A:
Q: OH RCOCI „NH2 EtO OEt A Base H,0*
A:
Q: Propose mechanisms and show the expected products of the following reactions. (a)…
A: b) 2,4-dimethylchlorobenzene + sodium hydroxide, 350 °C The mechanism for the given reaction is,
Q: Propose a suitable synthesis to accomplish the following transformation E OCH 3 OH
A: We have to carry out the given transformation.
Q: Provide a mechanism for the following transformation: OH Cro3 но. H30*
A: The mechanism of the given reaction is given below,
Q: Propose an efficient synthesis for the following transformation но- The transformation above can be…
A: The required reaction sequence is shown below.
Q: Propose an efficient synthesis for the following transformation: OH The transformation above can be…
A:
Q: 2-methyl cyclohexane gave a mixture of both cis- and trans-products. What might be the reagent and…
A: The answer to the following question is-
Q: When benzene is treated with diazomethane and irradiated with light, cyclohepta-1,3,5-triene is…
A:
Q: Propose a mechanism for the following reaction:
A: The given reaction is represented as follows:
Q: Give the major product for the following reactions. A Оз Zn/CH3COOH Br2 H2 B H20 Pd Ch HCI E H* H20…
A:
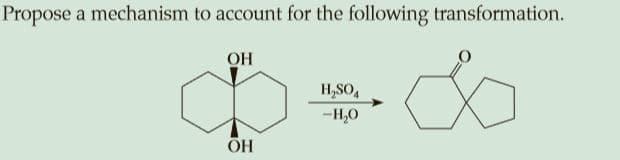
Q: Propose a mechanism (step by step) for the following transformation. Explain each step. HO. H,SO,…
A: Alkenes contain loosely bonded pi-electrons. In the presence of an acid, the alkenes are protonated…
Q: Br O,N O,N AIBR3
A:
Q: Propose an efficient synthesis for the following transformation: OH i H Ph 1) MeMgBr 2) H3O+ Ph The…
A:
Q: 7. Propose a mechanism for the following reaction under conditions that favor a SN1 reaction. H,O OH…
A: Nucleophilic substitution reactions are those reactions in which nucleophile attacks the alkyl…
Q: Provide a mechanism for the following transformation: 1. PPh 2. Buli Ph Br Ph 3.
A: THIS IS BASICALLY WITTIG REACTION,WHERE FIRST WE GENERATE THE WITTIG REAGENT WITH: PhCH2Br,PPH3,BuLi
Q: 2. Propose a mechanism for the following transformation and explain why this type of reaction is…
A: Triethylsilane acts as protecting group for alcohol in presence of mild acid.
Q: Br 1. НаС B CH2 Br2 Mg А FeBr3 Et,0 2. H3O*
A:
Q: 9) Using curved arrows to indicate electron movement, propose a mechanism tor the following…
A:
Q: OCH3 CH3 CH2CH HO B Rank the compounds shown in order of increasing reactivity towards electrophilic…
A: electrophilic aromatic substitution are those reaction where aromatic C-H is replaced by…
Q: Provide a mechanism for the following transformation: CN CO2H 1. NaOH 2. H30*
A:
Q: d. HO, OH e. OMe OMe Ph OMe Ph. OH f. OH NH NH2 OMe OMe
A:
Q: Propose a way to carry out the following transformation beginning with the starting material bong ng…
A: The question is based on the concept of organic reactions. we have been given a reactant. we have…
Q: Propose a mechanism to account for the products formed in the following reaction:
A: Bromination reaction starts with the homolytic cleavage of N-Br bond in N-bromosuccinimide (NBS)…
Q: Provide a mechanism for the following transformation and show all resonance forms of the important…
A: Reaction : The 1-chloro 2-nitrobenzene reacting with the sodium hydroxide, Water…
Q: Give the major product for the following reactions. A O3 Zn/CH3COOH Br2 H2 В H20 Pd HCI Ch E H*
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: For the following transformation, predict the major product, and suggest a mechanism for its…
A: The product of given reaction has been provided with mechanism of the reaction.
Q: U18 18 HCI + H2O С. 18 `OH + H2O CH ОН CH
A: Acid catalyzed reaction with water : In presence of acid , and water acetic acid can inter-convert…
Q: 3. The Mannich reaction can take many forms, including reactions where aromatic heterocycles are…
A: Note : Sn2 reaction between alkyl bromide and pyridine nitrogen. Michael addition followed by…
Q: Propose an efficient synthesis for the transformation shown below. Provide explanations for each…
A:
Q: 5. Provide a mechanism for the following transformation: OH H-A Ph Ph-
A:
Q: 1. NaOH OH 2. PHPH Br
A: The given transformations involves a phosponium ylide intermediate which react with carbonyl…
Q: Propose an efficient synthesis for the following transformation: OH The transformation above can be…
A:
Q: Propose a mechanism for the following transformation: to OH (H,SO4] MEOH Meo
A:
Q: Propose an efficient synthesis for the following transformation: он ?. OH The transformation above…
A:
Q: Stel 'h meganisme vir die volgende reaksie Propose a mechanism for the following voor. reaction.…
A: Aniline on treatment with ethyl chloride forms N-ethyl aniline.
Q: Propose an efficient synthesis for the following transformation: OH The transformation above can be…
A:
Q: c. Give mechanisms for the following transformations i. PhSeNa, DMSO Ph ii NaH, then allyl bromide…
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Propose an efficient synthesis for the following transformation: OH The transformation above can be…
A: In the given reaction the product is formed in three steps. The first step of the reaction is the…
Q: Propose a mechanism to account for the following reaction: CH3 CH₂Cl H₂C AICI H₂C
A: We have to predict the mechanism for the given reaction.
Q: OH CH3CI 1. NH, NazCr207, H* B A C AICI, 2. CH3B Нeat ČHĄCH3
A: The reaction schema given is,
Q: OH (a) OH (c) (d) OH (f) OMe
A: Organic reaction mechanisms: As per our guideline we have to answer first three steps only…
Q: Propose a mechanism for each of the following reactions
A: The organic reactions are carried out with the help of various reagents. This helps in the…
Q: Propose a plausible step by step mechanism for the following transformation H30*/heat `OH
A: To get more stabilized carbocation ring expansion takes place and the elimination will give the…
Q: 1. NaOH 2. H0 1. TsC, pyridine 2. EION 1. NaH 2.EIBr
A: In first question three memebered cyclic state is higly unsatable it break and carbocation come on…
Q: Br +. NBS peroxide Br
A: INTRODUCTION: NBS is N-Bromosuccinimide.It is used for allylic bromination via free radical…
Q: 1 Predict the product for the following transformation. Br 1. Me-PPH3 n-BuLi, THF 2. cat. Cp2La(NM…
A: These are the examples of imination of alkene and Haffman elimination
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Q2 Propose mechanisms for the following reactions. (a) (b) (c) (d) (e) Br HBr ROOR hv cat. H₂SO4 H₂O HBr > HCI EtOH cat. H₂SO4 H₂O Br OH Br xBr+ Br CI CH₂CH3 OH H H -OH Br CH₂CH3In a strongly acidic solution, cyclohexa-1,4-diene tautomerizes to cyclohexa-1,3-diene.Propose a mechanism for this rearrangement, and explain why it is energetically favorable.PPropose mechanism (showing explicity how observed mixtures of products are formed) cyclopenta-1,3-diene + Br2 ---> 3,4-dibromocyclopent-1-ene + 3,5-dibromocyclopent-1-ene

