Q: a. Alcohol CH3-CH=C-CH3 180°C ČH3 b. Alcohol H,SO4 CH3-CH=CH2 180°C c. Alcohol H2SO4…
A:
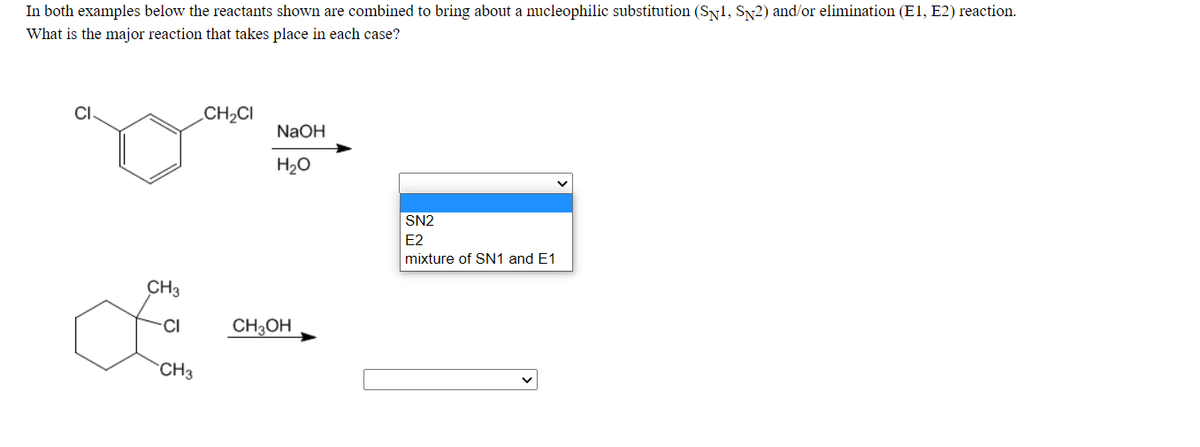
Q: Draw the substitution and elimination products.
A: “Hello. Since you have posted a question with multiple sub-parts, we will solve the first three…
Q: -OCH3 d. CH;CHCH,CH3 а. ÓH CH3 OH b. CH3OCCH3 CH3 с. CH-CH,0CHCH-CH; f. CH;CH,CHCH;CH,CH3 CH3 ОН
A: Ether can be synthesized from alkene by oxymercuration-demercuration reaction.
Q: t-BUOK 5 t-BUOH CH CH2 CH3 ČH3 (R)-3-methylpent-1-ene 3-methylenepentane…
A:
Q: 1. CH;CH20- Na* 2. H*, CH;CH2OH он H. I ll. ...
A: We have to tell the specific information about product.
Q: 1. /TSOH Cyclohevanone B 2. H;C=CHCH;CI
A: This reaction is an example of stark enamine reaction. The enamines are regioselective with…
Q: 8. * CH3 CH3 CH3 CH30C, CH2CH NaOCH3 HC CH2CH3 Br Sy2 CH2CH3 VWX XwV O vWX O xwv
A: SN2 reaction:- It stands for substitution nucleophilic bimolecular. It is a bimolecular reaction…
Q: a. -CH2OH b. -CH2B1 c. –CH(CH3)2 -CH3 OH- -CH,CH,Br H- -CH3 -CI -CH,CH2OH –CH¿OH -CH,CH,CH,Br
A: Priority to the atoms/group of atoms are given according to Cahn-Ingold-Prelog rules/principles.…
Q: CH3 HCI CH3 CH3 CH3-CH-OH ZnCl2 CH3-CH-CI CH3-C=0 BCD DCB
A:
Q: 1. Hg(OAc)2, H2O 2. NaBH4 CH2OH HO -HO- I| OH II IV
A:
Q: CI SN2 + HO CH;OH or CI SN2 + НО II CH3-S-CH3
A:
Q: h. + i. H* H3C- -0-CH2CH3 H,0 j. H* OH H3C O~H + H3C.
A: (h) Nucleophilic addition of secondary amine occurs with ketone and after that loss of water…
Q: CH CH3 CH3 (I) „CH3 (II) „CH3 (III) CH, (IV) (V) IOH CH HO Step I
A:
Q: (d) (c) CH3 0 1. LIAIH4 ? 2. H2O H2C-CHCH,CH3 HBr. ? H2C=CHCHCH2COCH3
A: Lithium Aluminium hydride is a strong reducing agent which reduces esters into alcohols. The…
Q: 1. ВН+ THF (с) CH,=CH-CH2 -Ĉ-CH3 2. НО, Н.О2, Н20 CH3 1.Hg(OАC),, Нн,О, THF 2. NABH4 (d)…
A:
Q: 1) ? 2) ? 3) ? BRCH2CH2CCH3 HC=CCH2CH2CCH3
A: The conversion from reactant to product requires 3 step process including protection of the ketone…
Q: CH3 HạC+ to CH3 OCH3
A: The given compound has molecular formula C5H12O
Q: CH CH CH3 H,C CH2 CH2 2-pentene-4-one A) Compound I 4-pentene-2-one I. I. B) Compound Il CH H3C. C)…
A: The structures of the given compounds can be drawn as -
Q: (a) (b) CH2CH2CHO (c) CH3CH2CCHCH, CHyČCH;CH2CH;ÖCH,CH3
A: Since we only answer up to 3 sub-parts, we'll answer the first 3. Please resubmit the question and…
Q: Grive the meckamiem of given जप्बत्जेनक र i) H2NNH2 2) KOH dCH 3 HI B HCN H30 B (10)
A:
Q: CH3O H* CHСCH-CH(CH)2 + H,0 CH3CCH2CH(CH3)2 + ? CH3Ó
A: Ketals and acetals are formed by reaction of the carbonyl with alcohols (methanol or ethanol) in the…
Q: (a) CH3 O (b) Br (c) CH3CHCH,COH CH3CHCH2CH2COH CH;CH,CHCH,CH,CH3 (d) (e) H H C=C HO2C" H3C…
A: Given compounds are : IUPAC names of the given carboxylic acids = ?
Q: 1. HC =C-G- ÇH -CH- CH=CH 2 CH3 'CHg た。 Br CH3 Br CH = CH2 2. CH3 CZC-GH--CHs 3. CH = CH- CH-…
A: Given that : We have to write the IUPAC names for the following :
Q: 5. (X)C,H¬OC1- NH3 →C¢H„ON- Br2 →CH3CH2CH2NH2; Compound (X) i : КОН CH3 (b) CH3 (a) (c) -HO- (d) cl-…
A: To solve this problem we have to complete the given chemical reaction .
Q: (a) Br2 FeBr, (b) NaCN (c) HO H,SO, 180 °C Major product
A:
Q: a) ? -CI ? b) с-он -CH2OH с) ? -C-H С-он d) ? CH3CH2CH2C-OH CH,CH,CH2C-ONa
A:
Q: write a mechaniSHI 181 CH₂=CH- Los -CH3 + CH3–CH2–CH,—CH 120 OH A -0 d CH₂CH3
A: The given reaction is a example of basic aldol reaction. Aldehyde is more reactive than keton .
Q: What is an i somer of CH =C? CH3 CH's CH3
A: Given: To find: isomer of the given compound.
Q: + OCH3 DMSO Br H Br + CH3OH 'CH2CH,CH3
A:
Q: H. (b) Но HO WHO, H (с) H. (d) CH3 CH3 CI ОН H. (е) CH3 H3C O ? CH3CH=CHCHCH3 I| CH3CH + CH3CHCH
A: a) The reagent used in the given reaction is alkaline KMnO4. The reaction is as follows,
Q: CH3 Cro3 CH3 Reagent N, PCI5 OOCPH H;C CH3 Но. H2SO4 Reagent M K H;C *CH3 CH3 Menthol Cro, CH,COOH…
A:
Q: i some CH =CH2 r ? It CHz Cl
A: The IUPAC name of the given compound is : 5-Chloropent-1-en-3-ol. In this compound third carbon is…
Q: hamess CH3-CH cH3- cH3 -CH cH3-CH cH
A: In IUPAC nomenclature of ether the most complex group is named as alkane as root word where as the…
Q: Which is a stronger base?
A: A molecule or an ion that is capable of donating a proton or accepting an electron pair in a…
Q: DIBAH toluene H.O 1. H,O* HGSO 2. он HO- 3. CH,CHMgBr 4.
A: 1) The ethyl phenyl acetet reacting with the Di-isobutyl Aluminum Hydride (DIBAL) and…
Q: H;C „CH3 ? CH3 starting material final product
A:
Q: CI H,0 А (substitution) e) -CH3 H,0 B + C (elimination) major minor
A: A substitution reaction is a class of chemical reactions in which an atom, ion, or group of atoms…
Q: What is the configuration of the asymmetric centers in the following compounds?
A: Step 1: Actually, the asymmetric center is the center in a compound where all the groups attached…
Q: H* H3C-C-o-CH2CH3 H,0 j. HO H3C O-H + H3C
A:
Q: Name each of the following alkanes.
A: The given structure is
Q: (CH:)CHMGC1 (6) H:O (CH:).CHCHOHCH:CH CH:MEC1 + CH: - CH: (7) (8) + CH:CH:ONa CH:CH:OCH:
A:
Q: CH,-CH=CH, | hv (i) –CH3 CH3
A:
Q: CiCrh ONa Br eter CH CH
A: This is condition of E2 elimination reaction by which alkyl halide undergoes in elimination reaction…
Q: OCH3 "OH H3C CH2CH3 CI CH3 H3C H CI H OH H3C H. CH2CH3
A: IUPAC RULES : Find and name longest continuous chain(Parent chain) with functional group if…
Q: ОН а) КН, 18C6 -CH,CH=CH2 CH3 80 °C
A:
Q: Convert each cyclic monosaccharide to its acyclic form.
A: “Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: 1. NaOEt Rxn I 2. Нао CH3OH cat. H* Rxn II
A:
Q: CH3 H. H CH3 H H;C- H. CH3 (а) II or mationcohexane has a ter e H. CH,CH,CH3 H. CH,CH,CH3 H. (b) II…
A:
Q: (i) CH3 CH2CI (h) (е) (g) BrCH2 CH2Br CH3 CH2COOH HOCH2 CH2OH CH3 CH2MG Br
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Identify which substitution mechanism best fits the following statement: The reaction proceeds through a concerted mechanism. A) SN1 B) SN2Write the products for (A-I). Label products with the mechanism (Sn2, Sn1, E1, E2) that produced it and write out and label major vs minor elimination products.i need help filling out the following SN2 reactions with appopiate reactants, products, or reagents,
- For each of the questions below – determine whether the following combinations of reactants, will react by substitution (SN1 or SN2 mechanism), elimination (E1 or E2 mechanism) or will not react under the conditions given. draw the final structure and explain the logic for each decision. There may be multiple possible options for some reactions. You must include all 4 aspects of these reactions, substrate, nucleophile, solvent and leaving group in the decisionBelow is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?Substitutions vs elimination. Pls help me identify the major products and the dominant mechanism. (SN1/SN2/E1/E2 or their combination)
- Complete the reactions given below, write down the type of mechanism (SN1, SN2, E1, E2)?Multi-step synthesis. Give the reagents over the arrows and the intermediates between thearrows that are required to synthesize the product compound on the left from the reactant givenon the right for each retrosynthetic analysis.They are Sn1 and Sn2 reactions Answer step by step with explanation Please answer correct i will give you upvote.
- Several reagents and several organic structures are shown below. Construct a multistep synthetic route for the synthesis of acetylene from ethane by dragging the appropriate pieces into the bins. Note that each bin will hold only one item, and not every given reagent or structure will be used. (Stoichiometry is omitted.)For each of the following reactions diagram the steps from the substrate to the product. For each step, a) over the reaction arrow supply the needed reagents b) name the type of reaction/mechanism {ROS (SN1, SN2, radical) E(E1, E2) A [acid-base, addition (carbocation, onium bridge, radical)]}Decide which compounds from the list below are best suited for nucleophilic addition reactions and which ones are more appropriate for nucleophilic substitution reactions.

