Q: Q4: - Determine if the following systems are fermions or borons? And why? (1) Kernel He. (2) Corn 19...
A: Fermion- A subatomic particle, such as a nucleon, which has half-integral spin. Boson- A subatomic p...
Q: calculate the volume (mL) of 0.402g of argon gas at a temperature of 11.5 and a pressure of 8.5 atm....
A:
Q: An empty steel container is filled with 4.40 atm of H₂ and 4.40 atm of F₂. The system is allowed to ...
A:
Q: For nos. 18 and 19, consider the following choices of acids and their corresponding pKa values at 25...
A: pKa is defined as the negative logarithm of ka i.e. pKa = -logKa
Q: Provide a multistep synthesis for each compound from the given starting material. Provide the reagen...
A: We have given the organic reaction and we have to multistep synthesis for each compound from the giv...
Q: when are surfactants used In seperation techniques
A: Surfactants are substances that lower the surface tension of water, making the molecules slipperier ...
Q: element ionization energy nitrogen (Choose one) ♥ arsenic |(Choose one) gallium |(Choose one) fluori...
A: list in order of ionization order--
Q: The substance nitrogen has the following properties: normal melting point: 63.20 K normal boiling po...
A:
Q: LOH (RL NaOH (S) H2O This nucleophilic substitution occurs with rearrangement. Draw curved arrows to...
A: In this question, we will write the Nucleophilie reaction mechanism by using carved arrows. You can...
Q: A chemical reaction occurring in a cylinder equipped with a moveable piston produces 0.641 mol of a ...
A:
Q: Two of the most abundant noble gases in the Earth's atmosphere are Argon and Neon. Calculate the rel...
A: Given: The gases are argon (molar mass of 39.95 g/mol) and neon (molar mass of 20.18 g/mol).
Q: 2al(s) + cr2o3(aq) > al2o3(aq) + 2cr2(s)
A: The given chemical reaction, 2Al(s) + Cr2O3(aq) -------> Al2O3(aq) + 2Cr(s)
Q: An electrochemical cell consists of a half-cell in which a piece of platinum wire is dipped into a s...
A: The standard reduction potential values are: Mg2+(aq) + 2e- ---> Mg(s) ; Eo = - 2.37 V I2(s) + 2...
Q: When a 50-gram piece of aluminum at 100 deg C is placed in water, it loses 735 calories of heat whil...
A: We have to calculate the joules if heat lost, and specific heat of aluminium.
Q: the equation for the react 1 2 3 4 acid-base precipitation
A: 1) balanced chemical reaction, Pb(NO3)2 (aq.) + 2NaI(aq.) + PbI2(s) + 2NaNO3(aq. 1 B) As Solid PbI2...
Q: The units of PRACTICE Using the reaction above, identify the limiting reactant and the theoretical y...
A: given moles of PCl3 = 1 mole mass of H2O = 50 g
Q: For Nie) + 2H* (? M) H2 (1 atm) + Ni*2 (1 M) E = .01v Calculate the pH of the solution. Given: Nics)...
A:
Q: a.) Incorrect number of valence electrons b.) Incorrect entral atom c.) Octet rule not satisfied ...
A:
Q: Calculate the hydronium ion concentration in an aqueous solution that has a hydroxide ion concentrat...
A:
Q: I Review | Constants | Periodic Table An 8.45 L tire contains 0.532 mol of gas at a temperature of 3...
A: Given V = 8.45L no of moles = 0.532 T = 320K
Q: Find the mass of helium that can be stored in this container at 289 K Express your answer using two ...
A:
Q: If 100 ml of an aqueous solution of HNO3 contains 0.0056 moles, what is the pH?
A: Given Volume of HNO3 solution = 100 mL = 0.1 L Moles of HNO3 = 0.0056 moles pH of the solution = ? N...
Q: what is the chemical equation that represents the standard heat of formation for methanol, CH3OH
A: Here, we have to write the chemical equation that represents the standard heat of formation for meth...
Q: A student was performing an acid-base extraction of a mixture of three compounds. The three compound...
A: The solution is as follows:
Q: 4. a) Calculate AG° for the following reaction, using the provided information. 2 C,H61) + 15 02() →...
A:
Q: A 0.15 M aqueous solution of some weak acid (HA) has a pH of 4.55 at 25 °C. What is the ΔG° for the ...
A:
Q: [20] ml of [4] M HC2H3O2 is titrated with 2.25 M sodium hydroxide. Ka for HC2H3O2 is 1.8 x 10-5 a. H...
A: Since you have posted questions with multiple sub-parts, we are entitled to answer the first 3 only....
Q: )Given the following reactions Fe 03 (s) + 3CO (s) → 2Fe (s) + 3CO2 (g) AH = -48.0 k 3Fe (s) + 4CO2 ...
A:
Q: conc. HCI
A:
Q: The noble gas in the second period. O H2 O He 0.02 O Ne O Ar
A:
Q: Reagents 2 equivalents of NaNH2 H2, Lindlar's catalyst Na / NH3 a. HBr m. HBr, H2O2, hv c. H2O, H2SO...
A: We have to identify the reagents nessato accomplish the following given reaction as follows in step ...
Q: Consider a solution of 0.00500 M HBr for numbers 22-24. Determine the following: M 22. equilibrium m...
A: [HBr] = 0.005 M
Q: Please use the values in the resources listed below instead of the textbook values. From the standar...
A:
Q: ege.com/course.html?courseld%3D169856748&OpenVellumHMAC=b6918aU5d7d0ddc 14 of 26 Exercise 10.51 - En...
A: Given P1 = 755 mmHg T1 = 35°C P2 = ? T2 = 1245°C Since we are using a can the volume is constant.
Q: Benzoic acid and aniline are both sparingly soluble in water. Their respective salts, however, are b...
A: intermolecular H-bonding is formed when any molecule which has a hydrogen atom attached directly to ...
Q: 6. The plot given below was constructed from data that was collected for the following reaction. N2(...
A:
Q: Draw the structure corresponding to each of the following name. 1,2-epoxy-4-methylcyclohexane
A:
Q: You are given a mixture of compounds and asked to isolate a component that is believed to be a base....
A:
Q: Compare picture I and II and choose: A = If the indicated property is greater in picture I than in ...
A:
Q: Which was likely lost to generate peak A? a.)C b.) CH2 c.) Br d.)Cl
A: Given spectrum is mass spectrum (Intensity Vs M/Z value .) We know that peak around 130 is molecular...
Q: If you add an acid to water and you get me. Can you draw my Lewis structure with formal charges?
A:
Q: 2. Explain the meaning of AH:H2Om = -286 KJ. Include a chemical equation in your answer, and %3D inc...
A:
Q: Compare picture I and II and choose: A = If the indicated property is greater in picture I than in ...
A: We have to compare the stability of the given compound 1 and 2
Q: Hg(OAc)2 then NaBH4
A:
Q: Determine the products in this reaction:
A:
Q: [References] One half-cell in a voltaic cell is constructed from a copper wire electrode in a 3.1 x ...
A: Given data :- Zn2+(aq) + 2e- → Zn(s) E0 = -0.763 V Cu2+(aq) + 2e- → Cu(s) E0 = -0.33...
Q: Experimental observations show that thiocyanate (SCN- ), the conjugate base of thiocyanic acid (pKa ...
A:
Q: Type your numerical answers in the box provided in three significant figures (e.g., 0.123). Do not r...
A:
Q: Consider a solution of 0.00200 M HCIO4 for numbers 22-24. Determine the following: M. 22. equilibriu...
A:
Q: How much energy does an aluminum (M = 27.0) sample gain if its molar heat capacity is 24.2 J/mol•K, ...
A:
Complete the following transformations by indicating the reactant structure, all necessary reagents, or the major organic product(s).
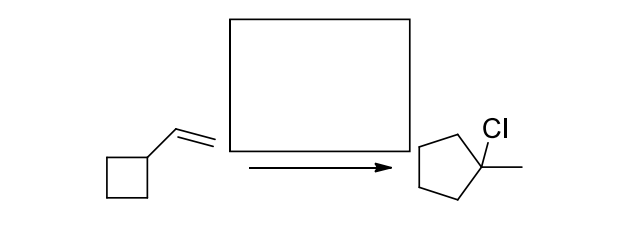
Step by step
Solved in 2 steps with 2 images


