Q: 10. The molecular formula of the unknown is C,H,N. Deduce the structure using the given spectra.
A: Here Given that, The molecular formula of a compound is C9H9N. So the degree of unsaturation in the…
Q: What are the notable peaks in the molecule? Please answer completely.
A: IR spectra helps to determine the actual structure of organic compound. It will be gives the…
Q: Determine the structure for the molecule that most likely produced these spectra.
A: From the IR data 3002 it's indicate that the presence of -OH group. 1724 it's indicates that the…
Q: What is the tR of peak A? What is the tR of peak B?
A:
Q: I would like to know the peaks for this IR spectrum
A: A question based on IR spectrum, which is to be accomplished.
Q: Give an advantage and disadvantage of the spectra obtained from an electron impact(EI) mass…
A: Adavantages and disadvantages of EI spectrometer
Q: Which molecule is consistent with this mass spectrum? Click on letters A to D to answer. 100 - 80 60…
A:
Q: Briefly describe the four stages of mass spectroscopy.
A: The answer is given below:
Q: The group most likely responsible for the peak at "1" is most likely: 3
A: Nuclear Magnetic resonance(NMR) spectroscopy is a type of technique that is used to determine the…
Q: I need help identifying and labeling the spectra please .. Can I get help explaining also Thank you
A: The structure of the given organic molecule can be determined using the mass, IR, and NMR…
Q: For the following mass spectra determine: presence or absence of Cl and Br, molecular weight, of it…
A: If the ratio of M, M+2 peaks is 3:1, then the compound contains 'Cl'. If the ratio of M, M+2 peaks…
Q: why is this mass spectrum named 2-propanol. Explain briefly include a fragmentation explanation in…
A:
Q: n the mass spectrum the highest peak is known as the base peak is also considered as the parent…
A: ->In mass spectrum the parent peak is due to the compound there is no fragmentation .it's…
Q: Answer the following questions : A. When analyzing an analyte solution in a specific solvent, what…
A: Two questions based on spectroscopy, which are to be accomplished.
Q: How can you interpret the Rf value to study any molecule?
A: How can you interpret the Rf value to study any molecule?
Q: 2. The baseline in the spectrum at right looks twisted. What is this due to? a) Poor shimming b)…
A: The structure of the unknown compound can be determined by the spectroscopical results such as…
Q: What is the base beak in an electron impact mass spectrum? Name one property of the positive…
A: Mass spectrometry is a technique for analysis of a substance in which molecules of substance ionised…
Q: What are the peak splitting for each peak?
A: NMR spectroscopy is a very important tool for the determination of the structure of the organic…
Q: which group is responsible for peak 1, 2, 3
A: Infrared Spectroscopy (IR) is useful for the identification of organic compounds. Identify what…
Q: analyse the spectra and write the name of the compound . also write brief explanation for each…
A: According to the IR spectroscopy, the peak obtained at 1715 represents a carbonyl group whereas the…
Q: Starting material was [(Me5dien)CoCl2] and reacted with SCN to produce [(Me5dien)Co(SCN)2]. Using…
A: Co2+ is present in octahedral state which can show John Teller distortion, elongation or…
Q: please help analyze the major peaks of this ir
A: Please find the attachment.
Q: These five spectra were recorded from different isomers with our molecular formula C,H,CIO. They all…
A: SPECTRA 1:
Q: What a molecular ion peak has characteristics and how we can identify it? Explain with Examples.
A: The Mass Spectrometry has basically three types of peaks present in a mass spectrum.
Q: draw the structure of ıons responsible for the peaks in mass spectrum in BENZOİN
A: Given Compound Benzoin Peaks in mass Spectrum = To be determined
Q: Is it possible to have an Rf value greater than 1? Why or why not?
A: We know that Rf value retention factor, and it is the ratio of distance traveled by the component…
Q: Why mass spectrometry is not called as mass spectroscopy:
A: Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio…
Q: ucumberlands.blackboard.com Question 18 MATCH a structure below to the IR spectra shown below. Но.…
A: IR frequency (cm-1): It is the number of wave crests that pass by a given point in one second…
Q: What a molecular ion peak has characteristics and how we can identify it? Explain.
A: Mass spectrometry is a spectroscopic technique which is helpful to find out the m/z value of the…
Q: The M+ peak for an alcohol in the mass spectrum is 74. Find the structure of the alcohol.
A:
Q: Why was yellow most absorbed at 420nm on my spectrophotometer in the lab but is at 600nm on the…
A: However, as it is generally followed at 600 nm goof to fo to compare with others. Some measure at…
Q: A mass spectrum with M+ at m/z = 101 with a minor M+1 peak contains what diagnostic atom? Please…
A: Mass spectrometry is a tool which is useful for measuring the mass to charge ratio of one or more…
Q: A mass spectrum with M+ at m/z = 101 with a minor M+1 peak contains what diagnostic atom?
A: In mass spectroscopy, the highest value of m/z is the value of the relative formula mass or…
Q: Q1. Why is it important to acquire a reference spectrum and subtract it from the sample’s spectrum?…
A: Since you have posted multiple independent questions in the same request, we will solve the first…
Q: Three students examined the signals in the HNMR spectrum shown below. The first student concludes…
A: Doublet and quatrate peaks are like this
Q: Why acid is added to the solution when analyzing mixtures in the UV-Visible absorption spectrometry
A: Acids may impact the pH of the analyte solution significantly.
Q: how molecular spectroscopy works? what are its advantage and disadvantages
A: Spectroscopy is based on the interaction between light and matter.
Q: Determine the structure for the molecule that most likely produced these spectra. Use all pictures
A:
Q: Beside each spectrum, list the important IR absorbances and the type of bond causing each signal.…
A: IR spectrum is an accurate tool to find the type of functional groups present in a molecule. It…
Q: One of the spectra below is produced by 1-chloropropane and the other by 1-iodopropane. Which is…
A: Chemical shift goes to downfield when a electronegative atom is attached to molecule that means…
Q: Which of the following molecules matches the given data? (Note: All the molecules have a M+ = m/z…
A: Given: Molecular Formula: C5H10O2 All the molecules have an M+ = m/z 102 base peak = m/z 43 To…
Q: Determine the molecular formula and possible structures for the unknown based on the spectra given.…
A:
Q: Please list the relevant IR peaks
A: IR spectrum is used to identify the functional group present in an organic compound.
Q: a) which bond is present in alcohols but not alkanes? b) is this band strong or weak (in terms of…
A: This question is related to IR spectroscopy. IR spectroscopy is a branch of spectroscopy that deals…
Q: elect one: Oa. The concent
A: An intermediate reaction is neither of the product or neither of the reactant it exists during the…
Q: Determine the structure of the molecule, provide the missing splitting pattern, and label which…
A:
Q: What is the effect of an increase in temperature on the absorbance value?
A: Molecular movement (vibration and rotation) increases at a higher temperature; more energy is needed…
Q: the most intense mass spectrum signal for 12 m/z 29 m/z 41 m/z 57 m/z
A: We have to predict most intense signal value and the structure.
Q: 1. Describe a black body radiator and its spectrum.
A: spectrum:
Q: Given the following molecule, draw its predicted standard spectrum along with DEPT 90 and DEPT 135…
A: Standard 13C, DEPT 90 and DEPT 135 spectrum of given organic compound.
Q: If the reference cuvette is dirty, will the spectrometer give an absorbance reading that is too…
A:
Q: e compounds shown is consistent with the given IR spectrum?
A: In this question we have to choose correct option of the compounds shown in consistent with the…
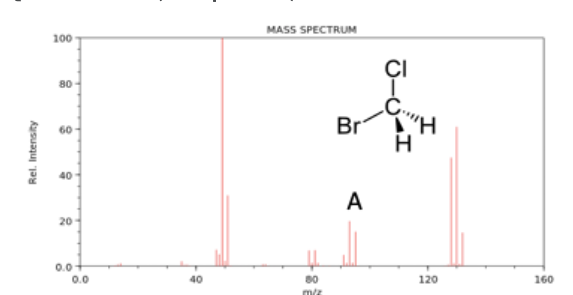
Which was likely lost to generate peak A?
a.)C
b.) CH2
c.) Br
d.)Cl
Step by step
Solved in 2 steps

- Provide a concise explanation of Mass spectrometry (MS) in regard to food samples. include the specific application name (e.g., Identification andQuantification of Mercury in Salmon), outline a brief description of samplepreparation procedures, and provide a summary of the obtained results from the analytical processSample Physical Appearance 0.1 M Na2CO3 0.1 M CaCl2 Na2CO3 + CaCl2 mixture Part II filtrate + CaCl2After creating her standard curve for absorption versus the concentration (M) of FD&C Red 40 and processing her data, Sally found that her drinking strength Kool-Aid sample had a concentration of 4.692 x 10–5 M. If 0.132 grams of Kool-Aid powder was used to prepare an 8-fl oz cup of her assigned flavor, what is the percent by mass of FD&C Red 40 in her Kool-aid powder? Report your answer as a percentage with two places after the decimal. Do not include the percentage sign in your answer. Pay attention to units. You are trying to find the mass percentage of red dye, so you need to know each variable in the equation in Appendix B. Note that the percent by mass equation should be multiplied by 100. There may be some useful conversion factors in the experimental procedure.
- A loss of 0.35 mg of Zn occurs in the course of analysis to determine the element. Calculate the percent relative error due to this loss if the masses of Zn in the replicate samples are the ff: (a) 40.0 mg (b) 400.0 mg (c) 175.0 mg (d) 600.0 mg.explain in short terms the following vocab: exact mass, elemental analysis, mass spectrometry and their applicationsWhat volume (in cm3) cm3 of glacial acetic acid (100 % of acetic acid, with a density of 1.050g/cm3 ) is needed to produce 1.0 dm3 of pH 3.3 acetic acid?Ks = 1.75 x 10-5MM [CH3COOH] = 60.05 g/mol
- Explain three different types of mass spectrometers. Differentiate them, construction,working and advantages.Calcium ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) Ca (s) 0 0 41.4 Ca (g) 178.2 144.3 158.9 Ca2+ (g) 1925.9 CaC2 (s) -59.8 -64.9 70.0 CaCO3 (s, calcite) -1206.9 -1128.8 92.9 CaCl2 (s) -795.8 -748.1 104.6 CaF2 (s) -1219.6 -1167.3 68.9 CaH2 (s) -186.2 -147.2 42.0 CaO (s) -635.1 -604.0 39.8 CaS (s) -482.4 -477.4 56.5 Ca(OH)2 (s) -986.1 -898.5 83.4 Ca(OH)2 (aq) -1002.8 -868.1 -74.5 Ca3(PO4)2 (s) -4126.0 -3890.0 241.0 CaSO4 (s) -1434.1 -1321.8 106.7 CaSiO3 (s) -1630.0 -1550.0 84.0 Carbon ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) C (s, graphite) 0 0 5.7 C (s, diamond) 1.9 2.9 2.4 C (g) 716.7 671.3 158.1 CCl4 (l) -135.4 -65.2 216.4 CCl4 (g) -102.9 -60.6 309.9 CHCl3 (l) -134.5 -73.7 201.7 CHCl3 (g) -103.1 -70.3 295.7 CH4 (g) -74.8 -50.7 186.3 CH3OH (g)…After creating her standard curve for absorption versus the concentration (M) of FD&C Red 40 and processing her data, Sally found that her drinking strength Kool-Aid sample had an FD&C Red 40 concentration of 1.086 x 10–5 M. If 0.133 grams of Kool-Aid powder was used to prepare an 8-fl oz cup of her assigned flavor, what is the percent by mass of FD&C Red 40 in her Kool-aid powder?



