Q: In atm what is the pressure in a 5.00 L tank with
A: Given volume5.00 L tank with 43.50 g of oxygen gas at 350 K We have to find pressure in atm
Q: Which of the following aqueous solutions are good buffer systems? (more than one answer maybe requir...
A: Buffer solution is the solution that has the ability to resist in change of pH when a small amount o...
Q: Propose a synthetic route for the following conversion: OH cyclobutanol 1,2-epoxycyclohexane
A: Alcohol will be first converted to alkene and then addition of ( Br2/ H2O) and then NaH is added wh...
Q: Choose the substance with the highest boiling point in the liquid phase. HOCH2CH2OH CH3CH2F CH3CH2...
A: Boiling point is the temperature at which liquid changes to gas (vapour) at atmospheric pressure. It...
Q: What is the process in which an ion is surrounded by water molecules arranged in specific manner.
A: This process is called hydration.
Q: For each of the following processes, determine the sign on the entropy and the best explanation for ...
A:
Q: A buffer solution is made that is 0.485 M in HNO2 and 0.485 M in NaNO2 . (a) If Ka for HNO2 is 4.50...
A: Solution - According to the question - Given - From Henderson-Hasselbalch equation of acidic buffer ...
Q: QUESTION 8 Select the structure of the major product formed from the following reaction. H;0 OH OH о...
A: Given : Reaction of alkene with H3O+ Product : ?
Q: 2.7. Predict the stereochemical outcome of the following reactions: CH3. CH3 a. b. epoxidation CH2 N...
A:
Q: Solve for pH & pOH a. 0.005 M HNO3 (Strong Acid) b. 0.85 M weack acid HX with Ka = 6.8x10-8 c. 1.5 ...
A:
Q: What, in general, is the order of decreasing reactivity of these carbonyl compounds towards nucleoph...
A:
Q: :CH 5 CH 2 CH2 3C=C6
A:
Q: The polymer shown can be formed by Fischer esterification. What monomer(s) would be used to make thi...
A: In the polymerization , the key step is the selection of appropriate monomer to get the desired prod...
Q: What is the hybridization of each atom in the following molecules? (Do not include hydrogen atoms.)
A:
Q: Which of the reagents listed below can convert a primary alcohol to an aldehyde? O AGNO3 O H2Cr04 KM...
A: The oxidation reaction of alcohols are reagent dependent which means that strong reagent leads to fu...
Q: The heat of fusion of pure silicon is 43.4 kJ/mol. How much energy in kJ would be needed to melt a 5...
A: Melting is defined as the process which convert from solid to liquid
Q: Suppose that Daniel has a 3.00 L bottle that contains a mixture of O,, N,, and CO, under a total pre...
A:
Q: Consider a binary solution for which the vapor pressure (in torr) of one of the components (say comp...
A:
Q: opose a synthetic route for the following conversion: OH cyclobutanol 1,2-epoxycyclohexane
A: It can be done by making alkene first and then doing epoxidation of alkene
Q: Use the data below to detemmine the Molarity of KMnO4 to THREE decimal places. (MM sodium oxalate = ...
A: Molarity of solution is defines as the number of moles of solute dissolved in per liter of solution ...
Q: Compare the de Broglie wavelength of a baseball moving at 90.0 miles per hour (40.2 m/s) to that of ...
A: The de-Broglie's wavelength can be given as : λ=hmv ....eq 1Here λ is de Broglie wavelengthh is P...
Q: Explain ion dipole interaction and ion induced dipole interaction. Give examples
A:
Q: estions: 1. Compare the results in experiment 1 and 3. What did you observe on the concentration of ...
A: 1.in exp 3 we keep the concentration of A as it is and increased the concentration of B 3 times of ...
Q: A buffer solution contains 0.341 M ammonium bromide and 0.480 M ammonia. If 0.0405 moles of hydroio...
A: On addition of an acid to the basic buffer solution, the concentration of weak base decreases wherea...
Q: Data Drawer Number Part I. Standardization of the Sodium Hydroxide Solution Mass of weighing bottle ...
A: Mass of oxalic acid sample is calculated as the difference in the weight between mass of weighing bo...
Q: Which of the following is formed when cyclohexanone reacts with hydroxylamine?
A: Carbonyl compounds when it reacts with hydroxylamine gives " oximes " by nucleophilic addition It...
Q: r an ideal gas has a pressure of 6.75 atm, a temperature of 90.80 °C, and a volume of 30.19 L, how m...
A: We know that the Ideal equation of state is : PV = nRT or, n = PV/RT ...(1) P = pressure of gas = 6....
Q: Which of the reagents listed below can convert a primary alcohol to an aldehyde? O AgNO3 O H2CrO4 KM...
A: Interpretation- To tell about which of the reagents listed below can convert a primary alcohol to an...
Q: Which of the following pairs of compounds would you choose as starting materials for the synthesis o...
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts. H30*
A: The given reactant have a double bond , so the proton ( H+ ) from H3O+ will be attached on this doub...
Q: a. From the given data, calculate the volume of the irregularly – solid and determine the dens...
A:
Q: Oxidation of alcohols involving the use of Na2Cr04 and H2S04 is also known as which of the following...
A:
Q: What would be the major product of the following reaction? NaOH `SH Br Br
A:
Q: A balanced, generic thermochemical equation is shown below. A, B, and C are substances involved in t...
A: The given thermochemical equation is: 2A + 3B + 42 kJ ----> 1C Since heat is on the reactant sid...
Q: Draw (1S,2S,4S)-1,2,4-trimethylcyclohexane.
A:
Q: An ideal gas is contained in a 1.5 L moveable piston at STP. Pext C) Without performing any calculat...
A: For a gas to be ideal, we assume that there is no force of attraction among the gaseous molecules an...
Q: eter to Fig. 5.15. Suppose you have a mixture containing 20 wt% acetic acid, 60 wt% water and the re...
A: Solution - According to the question - Given -
Q: The diagram below shows the energy levels and electron configuration for fluorine Which of the numbe...
A: F has a total of 7 valence electrons with a valence electron configuration of 2s2 2p5.
Q: a. Calculate the solubility of PbI2 (Ksp = 1.5 × 10¬8) in moles per liter. Ignore any acid-base prop...
A:
Q: The reaction rate for the decomposition of N2O5 to form NO2 and O2 was studied as a function of temp...
A:
Q: Select the structure that best fits the IR spectrum. CH;CH,CH,C=CCH3 CH;CH,CH,CH,OH CH;CH,CH,CH2C=CH...
A: By seeing the IR spectra one can easily predict the stretching frequency of different bonds by knowi...
Q: Analyze each statement (A and B) whether they are true or false. Use the choices below in answering ...
A: The given statements are, A) A dispersing agent such as Na-hexametaphosphate is needed prior to soil...
Q: Derive the half-life equation for a zero-order reaction. What is the half-life for the decomposition...
A: Here we have to derive half life equation for zero-order reaction and calculate the half life of abo...
Q: QUESTION 29 Which of the following structures will give the ozonolysis product below? 0, Me,s он OH
A: The above synthetically important reaction is an example of breaking the double bond of olefin follo...
Q: wavelength
A:
Q: QUESTION 38 What would be the final product of the following reaction series? HCN 95% H2SO4 heat HO ...
A: A question based on organic chemistry, which is to be accomplished.
Q: Which one of the following reactions is not a standard enthalpy of formation reaction? O A. Ca(s) + ...
A: Standard enthalpy of formation :- The enthalpy change involved when one mole of a compound is forme...
Q: Ctri H) Consiter the following Beaction: If 12.49 of PCI, is PeActeD Witn | 10.0g of Hy 0, what is t...
A: Given: Mass of PCl3 = 12.4 g Mass of water = 10 g Molar mass of PCl3 = 137.32 g/mol Molar mass of H2...
Q: What would be the major product of the following reaction? H* CH,OH он OCH, он он OCH, OH OCH3 OCH,
A:
Q: 10) 4A12(CO3)3 # of molecules: # of elements: Name of element: # of atoms:
A:
What is the IUPAC name for the compound shown?
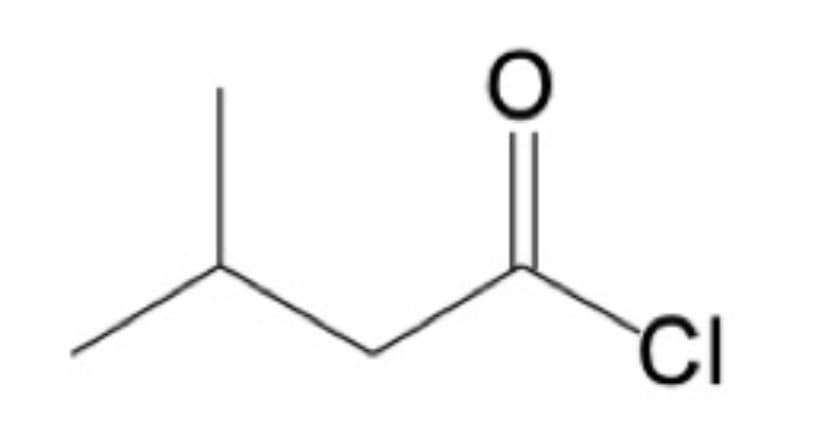
Step by step
Solved in 2 steps with 2 images


