Q: 8 TSHETKEIX 4800 001 TINSHETTINKE 8 1000 3000 1000 A B 2000 2008 MIYEHUNGER AVEMBER- 1100 1500 m…
A:
Q: For the following reaction, 15.5 grams of phosphorus (P4) are allowed to react with 41.6 grams of…
A:
Q: Quinine is a natural product extracted from the bark of the cinchons tree, which is native to South…
A:
Q: a). What does the chemical equation for a synthesis reaction tell us is happening in the flask? ble…
A: According to the guidelines of multiple question I will provide answer of first 3 part. Chemical…
Q: The molar heat of vaporization of water is 42 kJ/mol. How much energy is released by the…
A:
Q: he enthalpy change for the reaction of hydrogen gas with iodine to produce hydrogen iodide is 53.1…
A: Given reaction is, H2(g) + I2(s) -----> 2 HI(g) ΔH = 53.1 kJ
Q: 23- Write the detailed mechanism of synthesis of the compound (A) using suitable starting materials.…
A:
Q: The density of an (NaCl) ionic solid is 3.34 g/cm3 and the length of an edge is 482 pm. a. How…
A: Given: density of an (NaCl) ionic solid = 3.34 g/cm3 length of an edge (a) = 482 pm…
Q: (a) Determine whether this hydrogen peroxide reaction is first-order or second-order by first…
A: A first-order reaction is one in which the rate of reaction depends only on one reactant…
Q: For each of the salts on the left, match the salts on the right that can be compared directly, using…
A: Answer: For sparingly soluble salts it is considered that all their dissolved part in aqueous…
Q: what is the purpose of adding concentrated sulfuric acid to the product, 1-bromobutane after…
A:
Q: Three samples of three different gases are listed in the table below. All the samples contain…
A: Introduction The ideal gas equation, also known as the ideal gas law, states that PV = nRTwhere, P…
Q: Match the following aqueous solutions with the appropriate letter from the column on the right.…
A: with increasing the number of particles or ions, depression of freezing point occurs. Thus solution…
Q: o which element does the following electron configuration correspond? 2 4 a. 18²2s²2p 3s²3pª silicon…
A:
Q: Give mechanism of this reaction ASAP
A:
Q: 8= NBS hv
A: In this compound we have to find the total possibly major monobrominated product.
Q: is reduced in the following reaction: Cr₂O72- + 6S2032 + 14H+ → 2Cr³+ + 3S406² +7H₂O O Cr₂O7²- O…
A: Introduction The oxidation number of an atom is a measure of the degree of oxidation of an atom in a…
Q: Melting point is used in the recrystallization experiment to assess purity of solid samples.…
A: The melting point of a substance is the temperature at which it changes from a solid to a liquid…
Q: The density of helium gas under certain conditions is found to be 0.164 kg/m³. What is the volume in…
A:
Q: 2a) The molecule ethene (or ethylene), which has the molecular formula C₂H4, contains two carbon…
A: In the lewis structure of ethene, there is a double bond between carbon atoms, four C-H bonds. There…
Q: 4. Two volatile liquids, A & B, are mixed together. Under the conditions in the room, a pure sample…
A: The problem is based on Raoult's Law and it is a subtopic of Solution. The solution is given below.
Q: Draw all of the monochlorination products you would expect for radical chlorination of…
A: Cl2 in the presence of light undergoes radical formation. It then attacks various positions of…
Q: For which pieces of lab equipment will mass measurements be recorded using the analytical balance?…
A: =>Analytical balance-: this measurement balance is used to measure small amount of masses…
Q: If an oxide of carbon is 40% oxygen by mass, what is its empirical formula?
A: we have to determine the empirical formula of the compound
Q: Which of the two molecule represents the following ¹H-NMR spectrum? g 3H O The one on the right The…
A: triplet, 3H obtained at 1.05 ppm due to -CH3 of -CH2CH3 and quartet, 2 H indicate there is…
Q: OH HOO, но H₂O A ?
A: In the given reaction the reactant contains ketone and alcohol functional groups. In the presence of…
Q: Calculate the percent ionization of an acid if the [HA]initial = 0.4 M and at equilibrium the pH of…
A:
Q: Final Aqueous Layer Burette Reading Initial Aqueous Layer Burette Reading Final Thiosulfate Burette…
A: For third titration, Initial Thiosulfate burette reading = 6.83 mL Final thiosulfate burette reading…
Q: What mass of NH4Cl must be dissolved in 100 mL of H₂O to make a 0.250 M solution?
A:
Q: How to Convert 7.12 inches in kilometers?
A: Introduction Inches (in) is a unit of length in the imperial and United States customary systems of…
Q: If a solute is polar, then the solvent that theoretically should work for recrystallization should…
A: In the recrystallization experiment, the impure compound is purified by dissolving it in a minimum…
Q: a. CH3CH=CHCH3 c. CH3CH2CH3 b. d. Н. ΗΙ Н H на н -С-СЕС-С Н HA 'H H нн 6-bromo-4-methylhex-2-yne C-t…
A: We will determine bond line formula for each of the formula given in the question.
Q: You need to use a dilute hydrochloric acid solution in an experiment. However, the only bottle of…
A: We have to calculate the pH of the solution
Q: Four liquids are described in the table below. Use the second column of the table to explain the…
A: Using elevation in boiling point and depression in freezing point equation, order of boiling point…
Q: What is the multiplicity expected in the hydrogen NMR spectrum for the hydrogen atoms marked by a…
A: Given : structure of molecule Tip : multiplicity in NMR = n+1 n = adjacent hydrogen
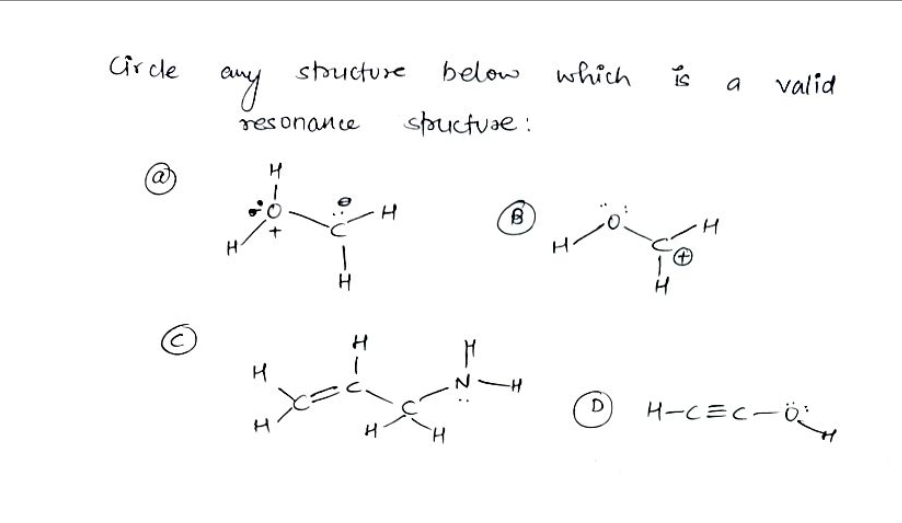
Q: In the structures below, circle all atoms that bear aportial positive charge as a result of apolo…
A:
Q: 26 Write equations for the reaction of 1-butanol, a primary alcohol, with these reagents. heat (a)…
A: Given : name of reactants and reagent
Q: A plot of ln[CH3CHO] versus time is linear. After 530 s, [CH3CHO] decreases to one half of its…
A: A question based on chemical kinetics. Few hints about chemical kinetics are prescribed based on…
Q: Using IUPAC guidelines, name the following organic compound HHHH |||| H-C-C-C-C-H AAAA Answer:…
A: IUPAC nomenclature: In IUPAC nomenclature a molecule's longest chain of carbons is taken as parent…
Q: A 480-pound alloy (which is just a mixture of metals ) containing 30% silver was mixed with an alloy…
A: Mass of alloy with 30% silver = 480 pound Mass of alloy with 55% silver taken = x pound % silver in…
Q: Assuming that Ar and Cl2 can be described as corresponding gases, what is then the pressure of the…
A: Given: Ar and Cl2 are corresponding gases. Pressure of Ar = 18.4 atm Critical pressure of Ar= 48 atm…
Q: Under certain conditions, the substance ammonium chloride can be broken down to form ammonia and…
A:
Q: 24. Upon titration, a mixture of weak acid and another weak acid whose Ka values are not too…
A: Equivalence point is the volume of the titrant at which the moles of titrant added equal to the…
Q: Starting from the wedge-and-dash structure below, rotate the back carbon to provide the Newman…
A: The Newman projection represents the dihedral angle and the different conformations of the molecule.…
Q: 25. Lactic acid (also known as milk acid) is composed of only carbon, hydrogen, and oxygen and has a…
A:
Q: OH CH₂OH H OH Н OH OH In the box below, draw the open-chain structure (as a Fischer projection)…
A: We have to convert this compound from Haworth to fisher projection.
Q: Using the information in the table, the value of the rate constant for the reaction 2 A(g) + B(g) →…
A:
Q: < 2:19 A) S-¹ B) M S-¹ What are the units for the rate constant of a first-order reaction? C) M-¹ s¹…
A: Introduction The rate constant of a reaction is an important parameter that describes how quickly a…
Q: The kinetics of a gas phase reaction of the form A → Products results in a rate constant of 0.00781…
A: Introduction Kinetics is the branch of chemistry that studies the rates of chemical reactions and…
Q: ОН н ОН н H₂N "I 5 - 10 min 22 °C Acetic Acid Acetic Anhydride heat ОН Н NaBH4 Ethanol 22°C ОН н H2O
A: The given reaction is a condensation reaction followed by acetylation on the amine group. The first…
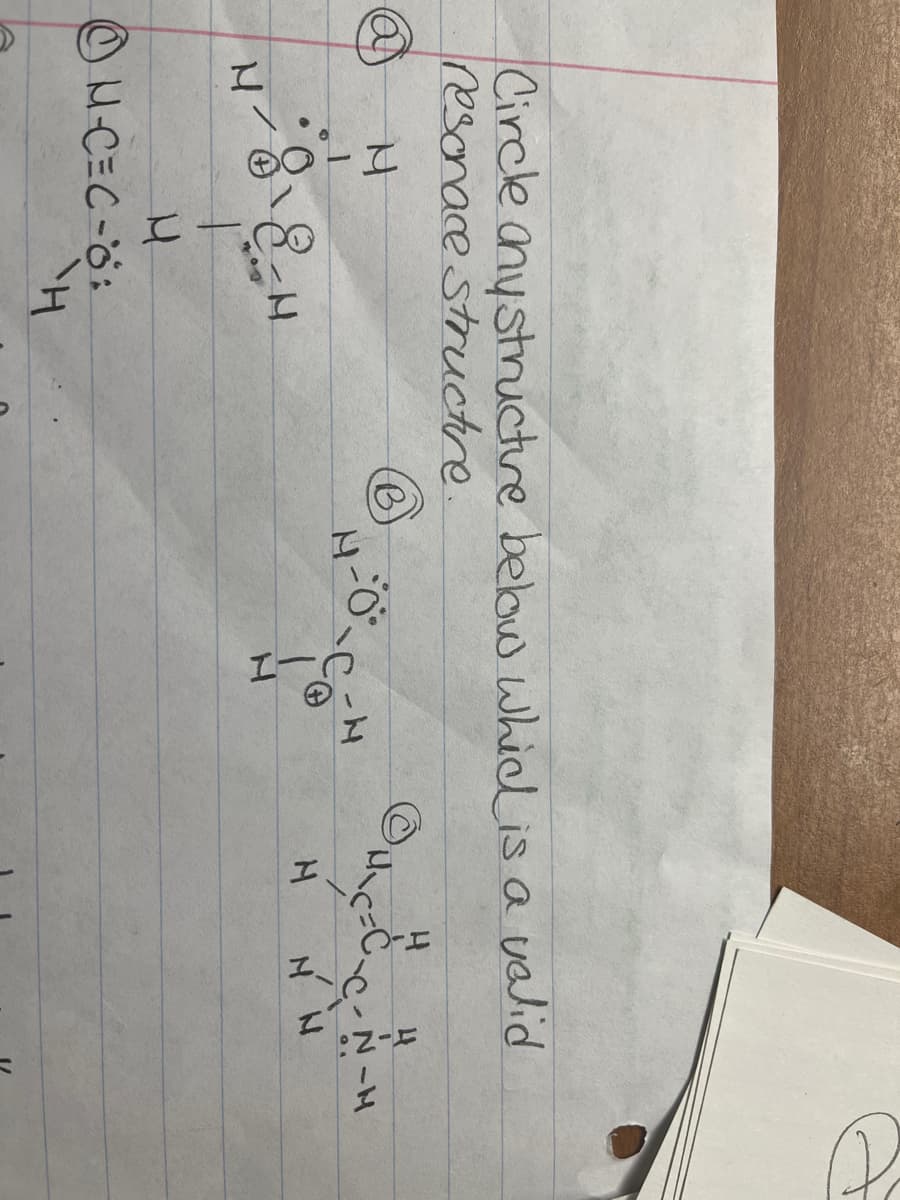
Step by step
Solved in 4 steps with 4 images

- A 1, 2-cis disubstituted cyclohexane, such as cis-1, 2-dichlorocyclohexane, must have one group axial and one group equatorial. Explain.Complete this graph of relative potential energy vs. rotation of the C1C2 bond of ethane. Thepoint drawn for you indicates the P.E. at 0° (eclipsed).(a)(2R,3S)-2,3-dibromohexane, draw a three-dimensional representation.
- Rank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2ClShow the delocalization of charge in the following structure, draw the resonance forms and indicate the movement of electrons with a curved arrow CH2=CH-OTrue or False: 1. The eclipsed conformation of a linear alkane IS called cis, while the anti conformation of an alkane IS called trans. 2. In the conversion of open-chain D-glucose to the ring form, the aldehyde carbon (carbon #1) bonds to the oxygen on carbon number 5 to form a ring that is both PYRANOSE and UNLOCKED.
- Propose structures for hydrocarbon molecules that meet the following descriptions: (2 pts.each)(a) Contains 4 sp2-hybridized carbons and 2 sp-hybridized carbons (b) Contains only 5 carbons, all of which are sp3-hybridized (c) Contains 2 sp3-hybridized carbons and 4 sp-hybridized carbonsGiven the following partial structure, add a substituent X to C-1 so that it satisfies the indicated stereochemical requirement. What is the A -C - C - X torsion (dihedral) angle in each?Rings + Unsaturation --- Hydrogenation If compound A C51H81BrN5O3P3 is hydrogenated to give compound B C51H101BrN5O3P3. How many rings does compound A have? Assume that P has a valency of 5. Would the answer be 4 rings? Formula -> unsat + rings = 1+C +N/2 - H/2 - X/2
- onsider the methyl group at the junction of rings A and B. Is it axial or equatorial to ring A? Is it axial or equatorial to ring B?draw double-newman projection for 1. neopentane, 2. isobutyl bromide, 3. sec-butylcyclopentane, 4. trans-1,2-dichlorocyclopropane.A.) On any clean sheet of paper, draw both the condensed and line-bond structure of the following compounds: 4) bromocyclobutane (Br-, Bromo branch) 5) 5-sec-butylnonane 6) 4-t-butylheptane 7) 4-ethylcycloheptene 8) 4,4-dimethylpent-2-ene 9) 3-chloro-2,7-dimethylnon-4-yne (Cl- Chloro branch) 10) 1-cyclohexylbut-2-yne



