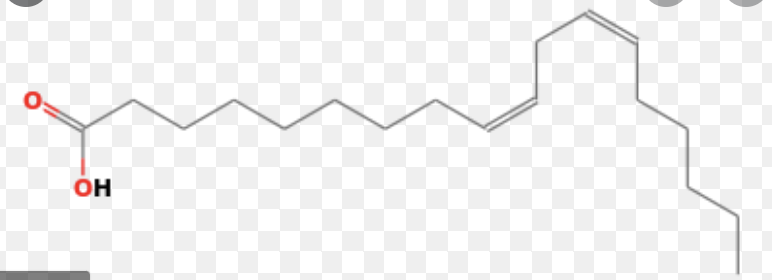
cis, cis-9,12-OCTAdecadiENoic acid Enumerate the number of carbons and unsaturations (double bonds) in this molecule
Q: Below is the structure of chitosan, a polymer used in wine fining to remove protein aggregates that…
A: The measure of acidic or basic property of a solution is referred to as pH. pH range of a solution…
Q: functional groups in palmitic acid, a saturated fatty acid and oleic acid, an unsaturated fatty acid
A: Biological lipids are a chemically diverse group of organic compounds which are insoluble or…
Q: Examine the structures of the following molecules. In each compound circle and identify by name each…
A: The answer is given in the image.
Q: Select the six atoms, including the labelled (*) one, in the tetrapeptide below that lie in the same…
A: The given peptide is composed of two alanine, one glycine, and one serine. The given peptide is…
Q: Now let us look at a real amino acid, alanine. Fill in the chart below for each ionizable group. You…
A: Amino acids are the building block of proteins. Amino acids are consisted of carboxyl group, amine…
Q: Histidine has three ionizable groups. On the titration curve below, show: a) 2 molar equivalents of…
A: Titration is the graphical representation of a plot, which shows the change in the pH as acid and…
Q: The physical properties of cholesterol are best described as amphipathic, charged amphipathic,…
A: Biomolecules that are composed of both hydrophobic and hydrophilic segments are termed amphipathic.…
Q: For the following peptide: group 3 O. C-NH, CH, Н 4 2 group 1 HO NH, 3 CH2 1 group 4 N. N-H group 2…
A: The given separation method has to be isoelectric Focusing or IEF as it is a method of separating…
Q: Glucose is a polyhydroxyaldehyde. Determine the structureof the product of the reaction of glycine…
A: Glucose was a simple sugar that was defined as polyhydroxy aldehydes associated with reducing…
Q: Spermaceti, a fragrant substance isolated from sperm whales, was commonly used in cosmetics until it…
A: Spermaceti can be described as a wax-like material present in sperm whales. It is present in liquid…
Q: which of the following functional groups are present in this molecule? amine amide amino acid…
A: Functional group is the group of atoms present within a organic molecule that provides the…
Q: Give one specific sample structure (Fischer Projection or Haworth) of the following carbohydrate…
A: Alditols are produced by reducing the aldehyde or a ketone group of monosaccharides or glycans at…
Q: Draw a skeletal structure for OCTADECANOIC ACID. A triglyceride contains thre octadecanoic acids as…
A: We'll answer the first question since the exact one wasn't specified. please submit a new question…
Q: Compound A is a dipeptide, optically inactive. While compound B is a tripeptide, and optically…
A: Peptides are polymers of amino-acid residues formed via peptide bond (which is a condensation…
Q: What is chemically nonsensical about this structure? H-C=C-H
A: Shared electron pairs, or covalent bonds, hold molecules together. These bonds are directional,…
Q: Basic chemical properties of amino acids with aromatic radicals in proteins. Use a few examples.
A: The aromatic amino acids found in protein are phenylalanine, tyrosine, and tryptophan. Phenylalanine…
Q: The following compound possesses two chirality centres labeled 'a' and 'B. Assign stereochemistry to…
A: Stereochemistry is the three-dimensional structure of the atoms in a molecule and their effect on…
Q: What is the structure of the general class of molecules that Ibuprofen belongs to? What is a…
A: Inflammation is a response of our body's immune system to certain environmental cues. The symptoms…
Q: The following lipids contain sphingosine as the alcohol backbone EXCEPT O Cerebroside O Ganglioside…
A: Sphingolipids have 18-carbon amino-alcohol backbone, sphingosine.
Q: Consider the intermolecular forces present in a pure sample of each of the following compounds:…
A: Molecular forces can be of two types, namely intramolecular and intermolecular. The intramolecular…
Q: Construct the primary structure of the following oligopeptides, preferably in their zigzag formulas.
A: Amino acids Proteins are the polymers of nitrogenous compounds called amino acids. Each amino acid…
Q: Column A shows the names of some of the important functional groups. Column B shows their…
A: A specific group of atoms that is present within a molecule and is responsible for the…
Q: D-Erythrose and D-threose are referred to as _____________ because their structures only differ in…
A: Stereoisomers are considered as the isomers, which differ to their spatial arrangements.
Q: Using octahedral symmetry, match each ligand SALC with its most likely interaction with a meta…
A:
Q: The following lipids contain sphingosine as the alcohol backbone EXCEPT O Cerebroside O Ganglioside…
A: Sphingosine is composed of an amino alcohol head group, with two hydroxyls, and one amino group…
Q: Trehalose, a disaccharide found in the blood of insects, has the following structure. What simple…
A: Trehalose, an unusual disaccharide sugar derivative derived from the insects, consists of two…
Q: Define the alternative chemical structure of deoxyribonucleic acid which could be copienfaithfully.
A: The Sanger chain termination sequencing method was invented by Sanger to identify the sequence of…
Q: Identify the monomers for the following polymers A.) Maltose B.) Sucrose C.) Lactose
A: INTRODUCTION Monomers are building blocks of polymers. The above mentioned compounds are…
Q: Give at least 10 examples of biological compounds having an alkyne functional group and identify the…
A: An alkyne contains at least one carbon-carbon triple bond. The simplest acyclic alkynes form a…
Q: Draw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic…
A: Acids are those chemical compounds, which are known to lose (H+) protons in the solution.
Q: Draw the complete structural formula of arachidonic acid (Table 23.1) in a way that shows the cis…
A: Arachidonic acid is a polyunsaturated fatty acid that was synthesized from dietary linoleic acid and…
Q: Sphingolipids can contain of all of the following except
A: Sphingolipids are a type of lipids that occurs in the cell membranes. They are extensively found in…
Q: The elements H, N, O, P, and S are commonly bonded to carbon in organic compounds. Arrange these…
A: Organic compounds : Organic compounds are generally any chemical compounds in which one or more…
Q: Glycine hydrochloride (Cl− H3N+CH2COOH) is a diprotic acid that contains a carboxylic acid group and…
A: Glycine hydrochloride is a non essential amino acids and is mainly found in gelatin and silk fibroin…
Q: These five anions: OH-, SH-, RO-, RS- and RNH- Please list out weakest to strongest in terms of…
A: Nucleophilicity: Nucleophile is a chemically reactive species that donate electrons to form bonds…
Q: Convert the following structural formulas into condensed structures.
A: Structural formulae are the structures that are drawn in a single line, whereas condense structures…
Q: The structure of D-arabinose is shown below. How many stereoisomers are possible for this molecule…
A: Arabinose is an aldopentose sugar. Arabinose found in the nature as part of the polymers such as…
Q: Check all that applies to the structure of fatty acid. H2C-O C1H35 CH2OH HỌ -C,7H35 + 3NAOH →…
A: Triacylglycerols are the esters of glycerol with the fatty acids. The hydrolysis of triacylglycerols…
Q: Glucose-1-phosphate has a ΔG°′ value of −20.9 kJ/mol, whereas that for glucose-6-phosphate is −12.5…
A: Glucose 6 phosphate is the first intermediate of glycolysis. Glucose-1-phosphate is formed by the…
Q: H HN-C-C-OH H-C-CH, CH2 CH3 The following short passage is about the compound with chemical…
A: Protein is a polymer formed from amino acid joined by peptide bond. There are twenty standard amino…
Q: Column A shows the names of some of the c functional groups. Column B shows their structure. Match…
A: As per the information given in the question, Column A represents groups and column B represents…
Q: Differences between Cis-fatty acid VS. Trans fatty acids
A: ANS: Unsaturated fatty acids have one or more double bonds. Each double bond may be in a cis or…
Q: For the following molecule, I need to identify several things, but I don't understand how to go…
A: Hi! Thank you for the question. We are authorized to answer three subparts at a time, since you have…
Q: Methionine has a sulfur atom in its formula. Explain why methionine does not form disulfide bridges.
A: The monomers, amino acids are linked by peptide bonds to form a polypeptide chain. One or more…
Q: HO-C-H H-C-OH HO-C-H H-C-OH ČH2OH
A: The Fischer projection was proposed by Emil Fischer in 1891. It depicts a three-dimensional molecule…
Q: Salol, the phenyl ester of salicylic acid, is used as an intestinal antiseptic. Draw the structure…
A: Phenyl salicylate is considered as a chemical substance that has several medical uses such as pain…
Q: The molecules shown below are: CH HN HN CH2 H. OH H OH OH O A= deoxyadenylate; B = thymidylate A=…
A: DNA consists of three types of component: phosphate, a sugar called deoxyribose, and four…
Q: The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by…
A: Proteins are composed of the polypeptide chain which has amino acids joined together by the peptide…
Q: Construct the primary structure of the following oligopeptides, preferably in their zigzag formulas.…
A: Proteins or peptide is the sequence of amino acids which is joined together by peptide bond…
cis, cis-9,12-OCTAdecadiENoic acid
Enumerate the number of carbons and unsaturations (double bonds) in this molecule
Step by step
Solved in 2 steps
