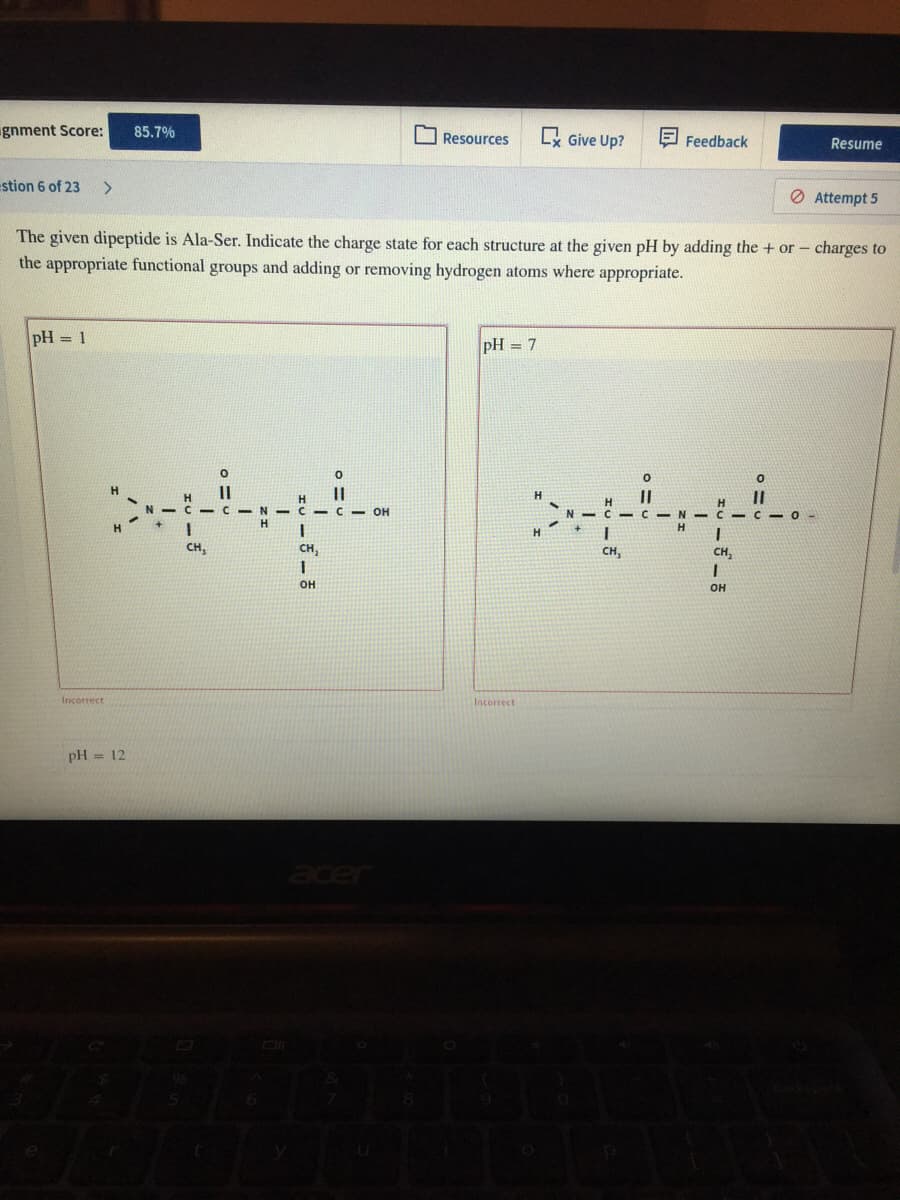
The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by adding the + or- charges to the appropriate functional groups and adding or removing hydrogen atoms where appropriate. pH = 1 pH = 7 %3D H H H. II C-N- C- C - OH H. CH, CH, CH, CH, он он Incorrect Incorrect
Q: A polypeptide is shown below. Please answer the following questions. OH В A C H;N E H D H a. Match…
A: A polypeptide is a long, unbranched chain of amino acids joined by peptide/amide bonds. The peptide…
Q: Give the NAME(S) of the functional group(s) present in the following structure? SELECT all that…
A: The functional groups are the moeity in the molecule which are attached to the compound and are…
Q: Daclatasvir is formulated at the dihydrochloride salt. Which functional group in Daclatasvir will be…
A: Daclatasvir is one of the potent, first-in-class, and a selective inhibitor of the nonstructural…
Q: As we have seen in Chapter 28, monosaccharides can be drawn in a variety of ways, and in truth,…
A: Monosaccharides are the simplest unit of carbohydrates. They can be divided into trioses, tetroses,…
Q: Convert each of the following chair conformations to an open-chain from and to a Fischer projection.…
A: Fischer projection is the representation of the sugar molecule in the open-chain form with the solid…
Q: Identify the circled functional groups and linkages in the compound below. OH CH3 H-N+c+CH2CH2-N-C+c…
A: A functional group is a substituent in a molecule that determines the characteristic features of the…
Q: Below is the titration of histidine. Calculate the average charge of histidine at pH 6.50. ÇOON…
A: Hi! Thank you for the question, as per the honor code, we are allowed to answer the first…
Q: Which of the following shows the reaction by which valine and leucine form a peptide bond H. H…
A: Valine and leucine are two amino acids that have branched-chain structures and are essential to the…
Q: H C-H O: H- OH HO H- C OH H C-H OH Which of the following best describes the saccharide shown? a…
A: Sacharides are defined as sugar molecules which are nothing but carbohydrates. The sacharides are…
Q: For the following peptide: group 3 O. C-NH, CH, Н 4 2 group 1 HO NH, 3 CH2 1 group 4 N. N-H group 2…
A: The given separation method has to be isoelectric Focusing or IEF as it is a method of separating…
Q: All of the following molecules are branched-chain amino acids EXCEPT 2 points H H TH C. H I H. HH H.…
A: Introduction: The correct choice is molecule C
Q: ◆ Arginine Phenylalanine ◆ Cysteine ◆ Serine Glutamic acid ◆ Glutamine ◆ Isoleucine Leucine A.…
A: There are 20 naturally occurring amino acids. In a polypeptide chain, the alpha amino and the alpha…
Q: which of the following functional groups are present in this molecule? amine amide amino acid…
A: Functional group is the group of atoms present within a organic molecule that provides the…
Q: The given dipeptide is Ala-Ser. Indicate the charge state for each structure at the given pH by…
A: The given dipeptide Ala-ser have different charge distributions at different pH, which are shown at…
Q: Expanded structure (Zwitterion Form] of the Essential amino acids' 1. 7. 2. 5. 8. 3. 9. Amide Bonds…
A: Amino acids are the primary building blocks of proteins. There are two categories of amino acids-…
Q: Which of the following explains why methyl anion has a pyramidal geometry while methyl cation is…
A: The methane (CH4) is the lowest possible alkane that can be formed, where a carbon atom is attached…
Q: H2N. H3CO- NAI IYK КАН BCS
A: Hydrolysis is the process of cleaving a bond with the help of water. During hydrolysis the water…
Q: This image shows us a of G6PD. O A. monomeric (tertiary) structure O B. homodimeric (quaternary)…
A: Glucose-6-Phosphate Dehydrogenase (G6PD) is the cytosolic enzyme that is a part of the pentose…
Q: What is the isoelectric point of casein? Please choose one correct answer only. A. pH 6.6 B. pH 5.6…
A: Peptides are molecules consisting of 2 to 50 amino acid residues covalently linked together by…
Q: Which of the following sub
A: To assign " R " and "S " configurations to chiral carbons in optically acive compound , Cohn…
Q: The functional group represented by N2 is: Amide Amine Lactone Lactam
A: The properties of any chemical molecules depend upon the functional groups present in them. Many…
Q: In the structure shown, which arrow is pointing to a peptide bond? OH LOH NH2 H OH HO-P=0 H. `A ÓH…
A: Peptide bond: It is a covalent bond used to join two amino acids to form a peptide chain. Generally,…
Q: H H |H I peptide chains H Type of bond Choose ペーUーエ アー O=U 1.
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: The pH vs charge graph for a triprotic amino acid is shown below. Please answer the following…
A: A. This is histidine triprotic amino aciD. It's the triprotic, with a third pKa value associated…
Q: In X-ray crystąllography, the wavelength of an X-ray is - 1.5 Å. This is useful in determining a…
A: X-ray crystallography is a technique used in the determination of three-dimensional atomic…
Q: The configuration of the given structure below is: * CH,OH он ÓH O alpha-L O alpha-D O beta -L O…
A: The carbohydrates can be represented by two types of configurations: L-isomer and D-isomer. When the…
Q: Refer to the figure below.The functional groups shown are the group,the group, and the group C…
A: All living organisms are made up of cells, which are the most basic unit of life. They are…
Q: Which of the following statements concerning the titration curve of glycine is correct? 2.0 E D 1.5…
A: Glycine is a hydrophobic amino acid with an alpha-carboxylic group and an alpha-amino group. Glycine…
Q: which functional group remains charged at pH of 9. „NH2 H2N COOH
A: The pKa of the ionizable groups on an amino acid determines the net charge of the amino acid. Since…
Q: Which of these reagents is commonly used to determine the number of polypeptides in a protein?…
A: In living organisms, all the physiological and biochemical actions are performed by the appropriate…
Q: The following are the absorption range for the functional group in IR spectrocopy below: D) alkyne…
A: Infrared (IR) spectroscopy is a technique that relies on the absorption of infrared radiations by…
Q: For each of the following compounds, indicate the pH at which 50% of the compound will be in a form…
A: According to concept of acids and bases, an acid is defined as a substance that donates a proton (a…
Q: Ph Level given: Acetic Acid = pH value of 3 Monocholoroacetic Acid = pH value of 2 Acetamide = pH…
A: pH value of a compound denotes whether the compound is acidic or basic. Lower the pH value more…
Q: Shown below is the titration curve for the amino acid glycine (side chain -H). What is the…
A: An amino group and an acid group are present in organic molecules called amino acids.…
Q: Shown below is the structure of a polypeptide with the pK, values of its acidic hydrogens. H pK, =…
A: Proteins are formed when different amino acids join together via peptide bonds. Proteins serve…
Q: Convert the following structural formulas into condensed structures.
A: Structural formulae are the structures that are drawn in a single line, whereas condense structures…
Q: Which of the following best describes the glycosidic bond in the disaccharide shown? CH,OH CHOH CH…
A: A bond formed between the anomeric carbon atom of a monosaccharide and the oxygen atom of the…
Q: H3C- -CH3 CH3 H3C CH3 Но ČH3 -CH3 A B D H2N NH2 OH CH3 -CH3 -CH3 H3C- CH3 H3C- H3C CH3 H;C ČH3 F G H
A: A functional group is a substituent in a molecule which governs it's characteristics of chemical…
Q: H HN-C-C-OH H-C-CH, CH2 CH3 The following short passage is about the compound with chemical…
A: Protein is a polymer formed from amino acid joined by peptide bond. There are twenty standard amino…
Q: Convert each of the following chair conformations to an open-chain from and to a Fischer projection.…
A: Carbohydrates are biomolecules.
Q: 2. Circle & Name functional groups -C-N, H. CH2 но CH H,C CH, HICICI
A: Amino acids: It is considered the building block of life as a polymer chain of amino acids forms a…
Q: Study the given structures below. Which of the following are enantiomers? * H- OH но- - H OH но- -H…
A: Carbohydrates or carbs are maconutrient consisting of Carbon, hydrogen and oxygen atoms. In nature…
Q: The following amine acids are given. SH CH3 *HạN-CH- ČH2 *H,N-CH- or Cysteine Alanine Draw…
A: Introduction Fatty acids These are long-chain hydrocarbons with carboxylic group end. Three fatty…
Q: Please draw all four bonds at chiral centers. Use wedge and dash bonds at chiral centers only. Do…
A: Enantiomers are the compounds that are mirror image to each other but are not superimposable .
Q: НО IZ NH₂ HO
A: The given molecule is composed of carbon, hydrogen, oxygen, and nitrogen. Molecules composed of…
Q: Define the following terms:a. optical isomerb. isoelectric pointc. peptide bondd. disulfide bridgee.…
A: Proteins are organic molecules that are present in living organisms. They serve a wide range of…
Q: Which of the following compounds would be most soluble in carbon tetrachloride, CCl4? a. NH3 d.…
A: Carbon tetrachloride is an organic compound. It has no flammability, used in refrigerators, fire…
Q: Sphingolipids can contain of all of the following except O more than one choice O phosphate O…
A: Sphingolipids are highly bioactive compounds. They participate in the regulation of cell growth,…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- The-carotene molecule has λmax 450 nm and ɛ = 15.000 m2 / mol. Calculate the expected absorbance value for a solution in which 0.1 mg is dissolved in 10 mL of water. ( β-carotene, C40H56, 536 g/mol path length 1 cm)A peptide has the sequence: Glu–His–Trp–Ser–Gly–Leu–Arg–Pro–Gly1. What would be the net charge of the molecule at pH a) 3, b) 8, and c) 11? (Use pKa values. Do not calculate the value per se, but instead estimate considering only fully protonated, or deprotonated states. Then estimate the pI for this peptide. Show full, clear and complete procedureans fast I will upvote A sample of PbSO4 solid has reached equilibrium in a beaker of water. If the ionic strength of the solution is then increased by adding LINO3 salt what happens to the activity coefficients of the Pb+2 and what happens to the solubility of the PbSO4 choose the option and explain the activity coefficient will increase and the molar solubility will decrease Both activity coefficient the molar solubility will decrease the activity coefficient will decrease and the molar solubility will increase Both activity coefficient the molar solubility will increase
- Structure activity relationship of pindolol80mL of a 0.3M solution of hexapeptide Leu-His-Cys-Glu-Asn-Arg is adjusted to pH=pI. The solution is then titrated with 0.2M HCL to a final pH of 2.1. Sketch the titration curve, labelling the pH and volume axes. Indicate the volume of HCL needed to reach relevant pKa value and equivalence point(s)z Relevant pKa values are: 2.1, 4.3, 6.0, 8.3, 9.8, and 12.5.10. o . Briefly discuss Ramchandran plot. . What is quaternary structure of proteins? Mention the role of various bond in its structure with the help of an example give its biochemical function. What are liposomes? Discuss their role in medicine. . Differentiate between cerebroside and ganglioside and one disorder associated with each. R
- Modify isoleucine to show the predominant forms at pH 1, 7, and 13. Isoleucine has p?ap�a values of 2.4 (carboxyl group) and 9.7 (amino group).using the expanded structure of Levothyroxine. Determine the molecular geometry of each central atom. Color each central atom based on its molecular geometry and include a key for your colors. For example, color tetrahedral C’s red, trigonal planar C’s , blue, etc.Which statements are true? Explain why or why not.1 A 10–8 M solution of HCl has a pH of 8.2 Most of the interactions between macromoleculescould be mediated just as well by covalent bonds as bynoncovalent bonds.3 Animals and plants use oxidation to extract energyfrom food molecules.4 If an oxidation occurs in a reaction, it must beaccompanied by a reduction.5 Linking the energetically unfavorable reaction A→ B to a second, favorable reaction B → C will shift theequilibrium constant for the first reaction.6 The criterion for whether a reaction proceedsspontaneously is ΔG not ΔG°, because ΔG takes intoaccount the concentrations of the substrates and products.7 The oxygen consumed during the oxidation of glu-cose in animal cells is returned as CO2 to the atmosphere.
- Assuming one Free Radical per molecule, draw all of the hydroperoxide isomers based on possible resonance forms of the fatty acid 18:1w7.draw the primary structure of the polypeptide at physiological pH range of 7.35-7.45 and put each charges beside the amino acids then compute for the isoelectric point please show complete solution DFACKT. Separating Glycated Hb From Normal Hb (Integrates with Chapters 5and 6.) Human hemoglobin can react with sugars in the blood(usually glucose) to form covalent adducts. The a-amino groups ofN-terminal valine in the Hb b-subunits react with the C-1 (aldehyde)carbons of monosaccharides to form aldimine adducts, whichrearrange to form very stable ketoamine products. Quantitation ofthis “glycated hemoglobin” is important clinically, especially fordiabetic individuals. Suggest at least three methods by which glycatedHb (also referred to as HbA1c) could be separated from normal Hband quantitated

