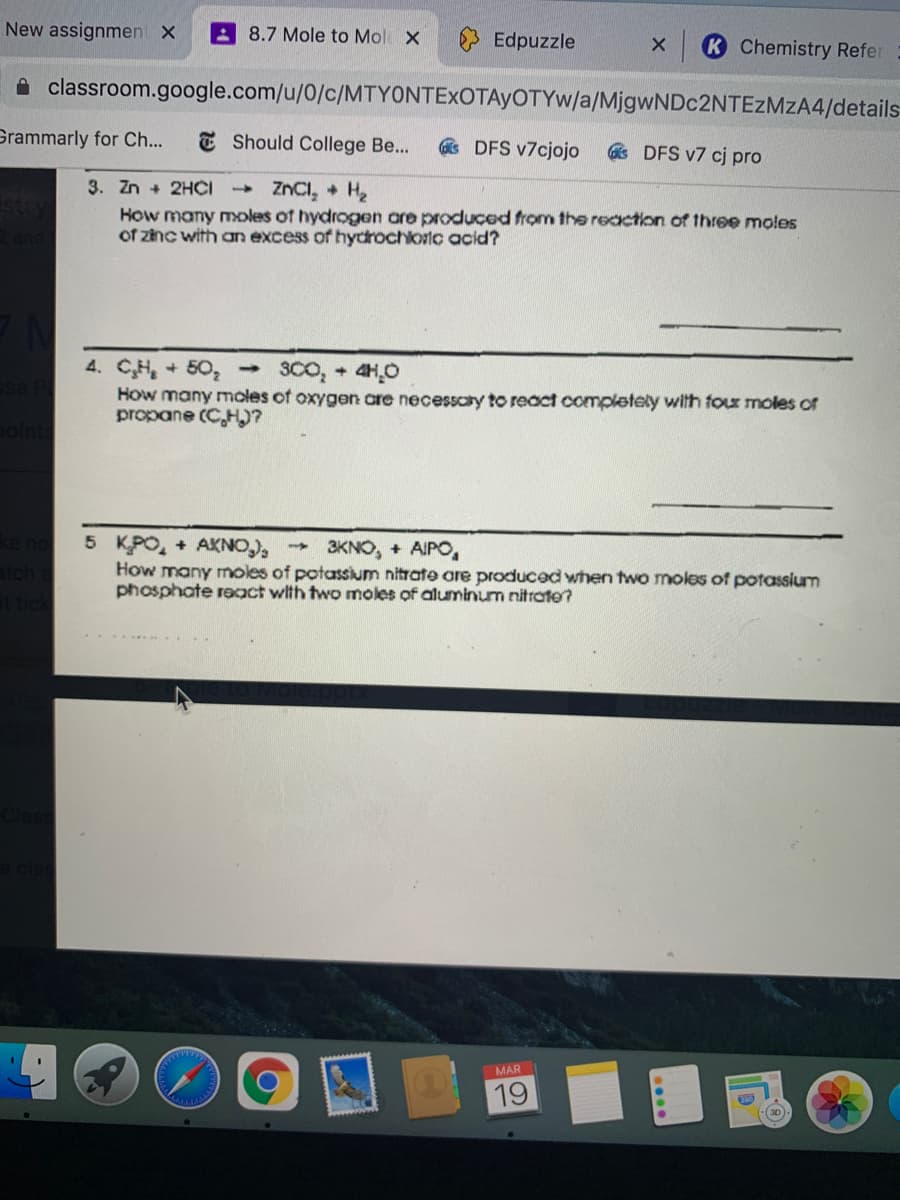
Čj pro ZNCI, + H2 3. Zn + 2HCI How many moles of hydrogen are produced from the reaction of three moles of zinc with an excess of hydrochloric acid? 3Co, + 4H,0 4. CH, + 50, How many moles of oxygen are necessaty to react completely with tour moles of propane (CH)? 5 KPO, + AKNO), How many moles of potassium nitrate are produced when two moles of potassium phosphate react with two moles of aluminum nitrate? - 3KNO, + AIPO,
Čj pro ZNCI, + H2 3. Zn + 2HCI How many moles of hydrogen are produced from the reaction of three moles of zinc with an excess of hydrochloric acid? 3Co, + 4H,0 4. CH, + 50, How many moles of oxygen are necessaty to react completely with tour moles of propane (CH)? 5 KPO, + AKNO), How many moles of potassium nitrate are produced when two moles of potassium phosphate react with two moles of aluminum nitrate? - 3KNO, + AIPO,
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter2: Atoms, Molecules, And Ions
Section: Chapter Questions
Problem 126CP: You have two distinct gaseous compounds made from element X and element Y. The mass percents are as...
Related questions
Question
Transcribed Image Text:New assignment X
8.7 Mole to Mol X
Edpuzzle
K Chemistry Refer
A classroom.google.com/u/0/c/MTYONTEXOTAYOTYW/a/MjgwNDc2NTEzMzA4/details
Grammarly for Ch...
E Should College Be...
s DFS v7cjojo
s DFS v7 cj pro
3. Zn + 2HČI
ZNCI, + H2
How many Yoles of hydrogen are produced from thee reaction of three moles
of zinc with an excess of hydrochloric acid?
4. CH, + 50, 3CO, + 4HO
How many moles of oxygen are necessaty to react completely with four moles or
propane (C,H)?
5 KPO, + AKNO),
How many moles of potassium nitrate are produced when two moles of potassium
phosphate react with two moles of aluminum nitrate?
3KNO, + AIPO,
MAR
19
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning



Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning



Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning