Q: Which is the better leaving group in each pair?a. Cl-, I-b. NH3, -NH2c. H2O, H2S
A: Generally good leaving group is weak base. This weak base will have stronger conjugate acids with…
Q: Br2 H20 Br
A: The chemical reaction of an alkene with halogen gas and water will result in the addition of a…
Q: Which SN2 reaction is each pair is faster? OBr O. OOCH3 Br A. OBr Br OCH3
A:
Q: Which carbocation is more stable?H2C=C+H OR HC=C+ b HC= C+ OR CH3C+H2
A: 1) Since a carbocation which is present on a less electronegative element will be having more…
Q: t-BUOK -> A is the reacich SN1,SN2 ET or E2 and wnat is the Br majar product. er EtONa
A:
Q: 1. Which alkene is the major product of this dehydration? HO H2SO4 heat А В C D
A:
Q: H CH,--CH, HS0, H2SO4 CH3-C-CH3 CH3-C=CH2 a. OH These 2 C's are identical. When either one loses a…
A:
Q: Show how each alkene can be synthesized by a Wittig reaction
A: Wittig reaction is an organic synthesis reaction which is used to synthesize alkene by reacting a…
Q: Which alkene in each pair is more stable?
A: Stability of alkene: Trans alken is more stable than cis alkene. More substituted alkene is more…
Q: Draw an energy diagram for an E2 reaction: (CH3)3CBr + −OH →(CH3)2C=CH2 + H2O + Br−
A:
Q: Draw the organic products formed when 2-bromopentan-3-one (CH3CH2COCHBrCH3) is treated with each…
A: The products formed when 2-bromopentan-3-one is treated with Li2CO3, LiBr, DMF is given as,
Q: NH2 major product?
A:
Q: Draw a stepwise mechanism for the attached reaction
A:
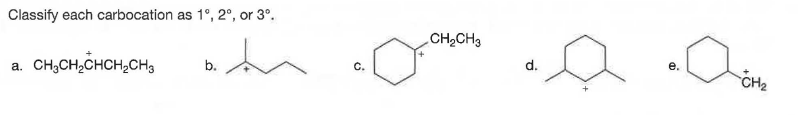
Q: Classify each carbocation as 1°, 2°, or 3°. b. (CHa),CCH2 a. С. d.
A: NOTE: Since you've posted multiple sub-parts, we'll solve only the first three sub-parts for you. To…
Q: Of the choices, the predominant product is CH3 :Br-Br: NH ? FeBr3
A: The above reaction is an example of the elecorophilic aromatic substitution where the attaching…
Q: Which alkyl halide in each pair reacts faster in an SN1 reaction?
A: SN1 is a nucleophilic substitution reaction It is a two-step reaction First step formation of a…
Q: Which compound in each pair undergoes a faster SN2 reaction? а. or b. Br or CI Br
A: The bimolecular nucleophilic substitution reaction is said to be SN2 reaction that means the rate of…
Q: G. Н. I.
A: "Since you have posted a question with multiple sub-parts, we will solve the first thee subparts for…
Q: What alkene is the major product formed from each alkyl halide in an E1 reaction?
A: E1 reactions are two step reactions in which the first step is the cleavage of carbon halogen bond…
Q: 30. Draw all of E2 elimination product with the correct stereochemistry. Label the major product. Br…
A: Following is the major product of the given chemical reaction.
Q: Circle the least reactive compound in an SN2 reaction. Explain your choice. H H. CH2I CH,CH,I
A: "Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: Draw enol tautomer(s) for each compound. Ignore stereoisomers.
A: a) Please fine below the enol tautomer for compound 1
Q: Br "OCH,CH3
A: Reaction of alkyl halides with nucleophile is termed as nucleophilic substitution. It is competed by…
Q: Which compound in each pair undergoes a faster SN2 reaction?
A: SN2 is a reaction in which attack takes place from back side, so less hindrance means faster or…
Q: What alkenes are formed from each alkyl halide by an E2 reaction? Use the Zaitsev rule to predict…
A: Given compounds,
Q: Which SN1 reaction is each pair is faster? А. CI OH2 H20 .CI OH2 H20 CI В. H20 CI OH2 H20 OH2 B,
A: Unimolecular nucleophilic substitution reaction (SN1): This reaction proceeds into two steps. The…
Q: Which group in following pair is assigned the higher priority? −CH2OH, −CHO
A: A priority of atoms or groups depends on an atomic number of atoms or groups. In other words, the…
Q: H;C-CH- -CH2- -CH2 CH3 CH3 Common Name 5. What resources could potentially be used to stabilize a…
A: Common name :- Propyl isobutyrate
Q: A is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. (a) Label each alkene that…
A: Given data : A is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. Each alkene…
Q: Do ethers have a good leaving group ?
A: Ethers are the organic compounds which have the general formula R-O-R'. A group which leaves as a…
Q: How can pentan-2-one be converted to each attached compound?
A: (a) (b)
Q: 1. H3C. CH3 ? ? + 1,2-Product 1,4-Product HBr (1 eq) 2. + Br2 ? ? (1 eq) 12-Product 1,4-Product 3. ?…
A: All these are examples of conjugated dienes which are more stable than simple or isolated dienes…
Q: Which alkene is lower in energy? r The alkene on the left is lower in energy The alkene on the right…
A:
Q: а) b) c) OEt
A: When aldehyde or ketone react with diol or alcohol it form acetal.
Q: Classify each substituent as electron donating or electron withdrawing.
A: Electron donating group: The group that increases the electron density on the ring. Electron density…
Q: Draw a stepwise mechanism for the following reaction.
A: The given reaction is haloydrin formation reaction where a halogenated enol intermediate is formed…
Q: One of these molecules reacts faster via an E2 reaction. Draw the products from each E2 reaction.…
A: Welcome to bartleby !
Q: The most stable carbocation a
A: A carbocation is a planar molecule in which a carbon atom has a positive charge and three bonds.
Q: Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2
A: Introduction: NaNH2 is a strong base. It is often used for deprotonation in weak acids or also for…
Q: OCH3 rovide the major product for gach reaction. 露CH。 EDG octtz Bro' Br2 FeBr3 EW G H2SO4 SO3 S-0H…
A: Electrophilic aromatic substitution is an organic reaction in which an atom that is attached to an…
Q: a) CH3CH2CH2CH2CH2CH2BR + CH3CH2ONA/CH3CH2OH b). 3-Kloropentan + CH;CH2ONA/CH3CH2OH, isi c)…
A:
Q: b) CH3-CH-CH-CH; ОН ОН frpm CH3-CHOH-CH,-CH3
A:
Q: OH OH OH II III
A:
Q: Why is a substituion (carbocation) more stable for a (OH) more than a teritary substituion?
A: Tertiary carbocation is a more stable carbocation thats why OH substitution is faster for tertiary…
Q: Why is - CN a weaker leaving group than - NHCH3.
A: Weak Bases are the Best Leaving Groups. (Recall that the stronger the acid, the weaker the conjugate…
Q: 7. For each reaction, circle the correct product. Br N2OCH3 or NaOCH3 or E2 E2
A: A species with a larger size can easily accommodate negative charge. In other words, in a large size…
Q: Classify each carbocation as 1°, 2°, or 3°.
A: Primary or 1o carbocation is the carbon in which the positive charge is attached to the carbon which…
Q: C. Which substituent has higher priority? CEN CH3
A:
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 5 images

- (a) What product(s) are formed when the E isomer of C6H5CH = CHC6H5 is treated with Br2, followed by one equivalent of KOH? Label the resulting alkene(s) as E or Z. (b) What product(s) are formed when the Z isomer of C6H5CH = CHC6H5 is subjected to the same reaction sequence? (c) How are the compounds in parts (a) and (b) related to each other?Which alkene in each pair is more stable?Draw all products formed by treatment of each dibromide (A and B) with one equivalent of NaNH2
- Rank the species in each group in order of increasing nucleophilicity.a. CH3CH2S-, CH3CH2O-, CH3CO2- in CH3OHb. CH3NH2, CH3SH, CH3OH in acetonec. -OH, F-, Cl- in acetoned. HS-, F-, Cl- in CH3OHDraw an energy diagram for an E2 reaction: (CH3)3CBr + −OH →(CH3)2C=CH2 + H2O + Br−Name each alkene: a. CH3CH=CHCH2CH2CH3 b. CH3CH=CH2 c. CH2CH3 l CH2=CHCH2CHCH3

