Q: Draw all stereoisomers formed in each reaction. Cl2 Br2 Br2 b. с. CHg а.
A: Stereoisomers are geometrical isomers with same molecular formula and connectivity but different…
Q: Answer each question using the ball-and-stick model of compound A. Draw a constitutional isomer that…
A: SOLUTION: Step 1: Hello. Since the question has multiple sub-parts, the first three parts are…
Q: Match each reaction with its correct energy diagram. Он OH H2SO4 H2SO4 HCI HO A) B) C) .......…
A:
Q: t-BUOK -> A is the reacich SN1,SN2 ET or E2 and wnat is the Br majar product. er EtONa
A:
Q: Draw the structure (including stereochemistry) of an alkyl chloride that forms each alkene as the…
A: Both ‘H’ and ‘Br’ group should be trans to each other. Thus the alkyl halides needed for the…
Q: Draw a second resonance structure for each carbocation. Then draw the hybrid.
A: In the below steps , please find the Resonating structure & hybrid
Q: Rank the attached alkenes in order of increasing stability.
A: The given alkenes are represented as follows:
Q: Which alkene has (E) configuration? а. b. c. d.
A: E–Z configuration is the method of describing the absolute stereochemistry of double bonds.
Q: Of the choices, the predominant product is CH3 :Br-Br: NH ? FeBr3
A: The above reaction is an example of the elecorophilic aromatic substitution where the attaching…
Q: Convert each compound to its enol or keto tautomer.
A: Keto-enol tautomerism represents a chemical equilibrium between ketonic and enolic form. Keto and…
Q: Which alkyl halide in each pair reacts faster in an SN1 reaction?
A:
Q: Rank the attached carbocations in order of increasing stability?
A: The order of stability of carbocations is: Tertiary carbocation > secondary carbocation >…
Q: a.) Classify the following carbocations (1º, 2º, or 3º). b.) Encircle the carbocation/s that can…
A:
Q: Which compound in each pair undergoes a faster SN2 reaction? а. or b. Br or CI Br
A: The bimolecular nucleophilic substitution reaction is said to be SN2 reaction that means the rate of…
Q: Which is the major product? a. A b. Neither product would likely form c. B
A: According to Saytzeff's rule (also known as Zaitsev's rule), during dehydration, more substituted…
Q: Label the α and β carbons in each alkyl halide. Draw all possible elimination products formed when…
A: The carbon to which the functional group is attached is called the alpha carbon. The next carbon to…
Q: Q#1 Draw all possible constitutional isomers formed by DEHYDROHALOGENATION of each alkyl halide. Br…
A:
Q: Rank the following alkenes in order of increasing stability:
A: The stability of alkene depends upon the degree of substitution and due to the hyperconjugation…
Q: Draw enol tautomer(s) for each compound. Ignore stereoisomers.
A: a) Please fine below the enol tautomer for compound 1
Q: Which compounds (B–F) are identical to A? (b) Which compounds (B–F) represent an isomer of A?
A: Given:
Q: Which compound in each pair undergoes a faster SN2 reaction?
A: SN2 is a reaction in which attack takes place from back side, so less hindrance means faster or…
Q: Which compound in each pair undergoes a faster SN2 reaction?
A: SN2 is a reaction in which attack takes place from back side, so less hindrance means faster or…
Q: Arrange carbocation in order of increasing stability
A: More the hyperconjugation, more is the stability of the carbocation.
Q: (b) (а)
A:
Q: Draw all stereoisomers formed when each alkene is treated with mCPBA
A:
Q: What alkenes are formed from each alkyl halide by an E1 reaction? Use the Zaitsev rule to predict…
A: By using the Zaitsev's rule the major product can be predicted as it states that the more…
Q: Write the IUPAC name and, where possible, the common name of each compound. Show stereochemistry…
A:
Q: Draw the given carbocation in orbital phase. CH2 CH2 (b) (a) (d) (c) H.
A: Correct answer is (b). Benzyl carbocation is resonance stabilized. Correct resonance structure is…
Q: A is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. (a) Label each alkene that…
A: Given data : A is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. Each alkene…
Q: Drag and drop the reagents into the right places. CI Cl2 CI- hv HNO3 H2SO4 FeCl3
A:
Q: Use any reagents necessary to complete the folowing transformations. a. b. C. d. OH
A: Applying concept of organic synthesis and reagent.
Q: Draw all stereoisomers formed in each reaction. а. Br2 b. Cl2 H2O
A: Answer of the question given below,
Q: Classify each substituent as electron donating or electron withdrawing.
A: Electron donating group: The group that increases the electron density on the ring. Electron density…
Q: ng carbocations
A:
Q: Match each structure with its correct IUPAC name. A) B) C) D)
A: Concept- In IUPAC, first check the longest carbon chain and then do the numbering. make sure the…
Q: Consider the energy diagram shown below when answer questions A- В C D FG E Progress of Reaction (a)…
A: The given energy diagram is shown below :
Q: 8. Choose the most stable alkene from each group: a. b.
A: Note: According to our guidelines we are supposed to answer only first question
Q: Draw the product of attached thermal electrocyclic ring closure.
A: An electrocyclic response is a reversible response that can include ring closure or ring opening. An…
Q: Which is the least stable carbocation? A. В. С. D. Select one: D
A: Correct answer is (B) Carbocation B is least stable because it is not stabilized by resonance and…
Q: Why is a substituion (carbocation) more stable for a (OH) more than a teritary substituion?
A: Tertiary carbocation is a more stable carbocation thats why OH substitution is faster for tertiary…
Q: (c) With increasing nucleophilicity (1: least nucleophilic; 3: most nucleophilic) ОН CHO.
A: Given nucleophilicity order
Q: Consider the reaction scheme below NBS hv a. Draw the major regioisomer resulting from…
A:
Q: Draw enol tautomer(s) for each compound. Ignore stereoisomers.
A: a. Those compounds which the same molecular formula but differ in the position of protons and…
Q: CI NaOH F G DMSO (Transition State) (Product)
A: Substitution reaction: When one of the substituents will be replaced by an incoming group then that…
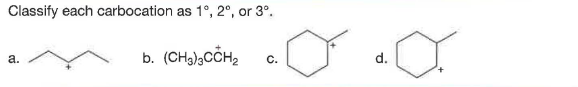
Q: Classify each carbocation as 1°, 2°, or 3°.
A: Primary or 1o carbocation is the carbon in which the positive charge is attached to the carbon which…
Q: Classify each solvent as protic or aprotic.
A: A solvent can be categorised on the basis of polarity of the bond. If it has a polar bond, that is,…
Q: C. Which substituent has higher priority? CEN CH3
A:
Q: Label each heterocycle as aromatic, antiaromatic, or not aromatic.
A: The aromaticity of a molecule is governed by the following factors: Cyclic Planar Delocalization of…
Q: Draw a stepwise mechanism for the attached reaction.
A: The reaction forms a carbocation intermediate, which undergoes rearrangement to give alkene as the…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Draw the enol form of each keto tautomer in parts (a) and (b), and the keto form of each enol tautomer in parts (c) and (d).A is a toxin produced by the poisonous seaweed Chlorodesmis fastigiata. (a) Label each alkene that exhibits stereoisomerism as E or Z. (b) Draw a stereoisomer of A that has all Z double bonds.Draw enol tautomer(s) for each compound. Ignore stereoisomers.

