Q: Is this compound aromatic? Explain why or why not. What if the charge was positive instead of…
A: Conition for compound to be Aromatic. Cyclic, planar, all carbon SP2hybrid, and follow Huckel rule…
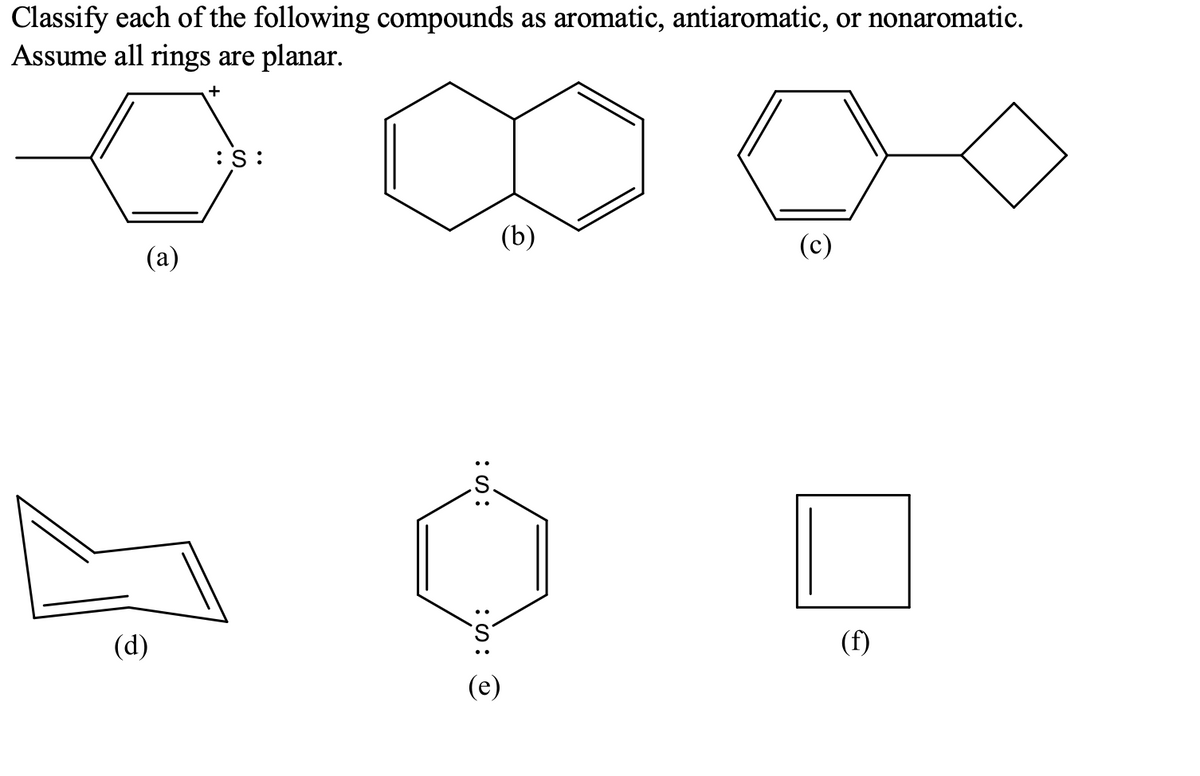
Q: Prove the aromaticity of the molecules given below by discussing them.
A: Since you are posted with multiple sub-parts. As per the rule I am answering the first three…
Q: Identify the molecule below as aromatic or not aromatic (assume planarity and full conjugation of
A: A compound must obey Huckel's rule in order to be aromatic in nature
Q: (b). Compound A a key unit of a prescribed drug for treating sleeplessness (insomnia). -N1 N²…
A:
Q: Some of the following compounds show aromatic properties, and others do not. Predict which ones are…
A: For compound to be aromatic it must follow (4n+2)π huckel's rule. If n =0 : 2π electrons n= 1…
Q: Which of the following anions is aromatic? A) B) C) D) CrO
A:
Q: Borazole, B3N3H6, is an unusually stable cyclic compound. Propose a structure forborazole, and…
A: Structure of borazole: The molecular formula of borazole is B3N3H6. The structure for borazole is…
Q: (b) The following compound (2) is a natural product drug. Using the criteria for aromaticity and…
A:
Q: In the mid-1930s, the German theoretical chemist Erich Hückel developed a rule that dealt with the…
A: The compound given is,
Q: A. Determine the number of electrons in a system of cyclic conjugation (zero if no cyclic…
A:
Q: Which of the tollowing compounds would be considered aromatic?
A:
Q: Which of the following compounds are aromatic based on the principles of the Huckel rule of…
A:
Q: Biphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b)…
A: (a)
Q: Is the unusual dication below aromatic/non-aromatic/anti-aromatic? Anti-aromatic; the overlapping p…
A: Aromatic compound: Those molecules that follow the huckle rule. condition: 1) cyclic molecule 2)…
Q: Discuss the aromaticity of the below compounds? Explain which compound is aromatic, why? II
A:
Q: Cyclopropenones are described as having aromatic character. How would you account for this, given…
A: Yes , Cyclopropenones are aromatic . Because according to huckel rule these follow (4n+2) π e- rule…
Q: Classify the following compound as aromatic, antiaromatic, or nonaromatic. Aromatic Antiaromatic…
A:
Q: A chemist isolated an aromatic compound with molecular formula C6H4Br2. He treated this compound…
A:
Q: Which of the following compounds represents an aromatic heterocycle? A) Compound I II. B) Compound…
A: Aromatic heterocyclic compound is the compound in which one or more carbon in the cycle is replaced…
Q: The following reaction sequence is described as an introductory organic chemistry laboratory…
A: In synthetic organic chemistry, the combination of appropriate reactions under suitable conditions…
Q: e)
A: Huckel's rule: The planar monocyclic conjugated system will be aromatic in character if it contains…
Q: Do you Understand Aromaticity? Determine whether the compounds below are aromatic or not.…
A: Aromatic compounds are must be obeys the Huckel rule which is states that the compounds which are…
Q: Why is the compound shown not considered aromatic ev though it has 3 double bonds in conjugation and…
A: A molecule is aromatic if it is cyclic, planar, completely conjugated compound with 4n+2 pi…
Q: Characterize the following compound as: A. Aromatic B. Antiaromatic C. Nonaromatic D. Not enough…
A: We need to find if the given compounds are aromatic, anti aromatic or non aromatic.
Q: Some of the following compounds show aromatic properties, and others do not. Predict which ones are…
A: Huckel's rule:The planar monocyclic conjugated system will be aromatic in character if it contains…
Q: 8. Classify the following compound as aromatic, antiaromatic, or nonaromatic. Explain your choice.
A: In this question, we will see that what is the class of this given Compound it is Aromatic,…
Q: A
A: Aromaticity is a property of cyclic, planar structures with pi bonds in resonance that increased…
Q: The compound MON-0585 is a non-toxic, biodegradable larvicide that is highly selective against…
A: In organic synthesis, different reagent converts the reactant into target products. These reagents…
Q: Which of the following are consistent with the requirements for aromaticity? I. A system with…
A: Criteria for Aromaticity The molecule is cyclic (a ring of atoms) The molecule is planar (all atoms…
Q: 1.2 Use the criteria for aromaticity to determine if the following compounds are aromatic,…
A:
Q: Discuss the aromaticity of the below compounds? Explain which compound is aromatic, why?
A:
Q: (b) By applying the rules for aromaticity, deduce if compounds E and F, shown below, are either…
A: E) Aromatic F) not aromatic
Q: which of the structures is aromatic, according to Huckle? list the 3 features that make them…
A:
Q: Five-membered aromatic heterocycles with one heteroatom undergo electrophilic substitutions…
A: Electrophilic substitution means electrophile will attack the position where electron density is…
Q: 1. A.Electrons in a cyclic conjugated system.| B.The compound is (a, aa, or na)
A: Aromatic compounds Anti-aromatic compounds Non-aromatic Compounds - cyclic compounds - flat -…
Q: Draw a Frost circle for the cyclopentadienyl anion, which is drawn below. Identify the bonding,…
A:
Q: Explain why electrophilic aromatic substitution in Pyrrole takes place at C-2 positions whereas, in…
A: Pyrrole is more reactive towards electrophilic reagents in comparison to pyridine because of its…
Q: If a molecule contains an aromatic ring, such as a benzene ring, attached to a non-aromatic ring,…
A: A question based on aromaticity, which is to be accomplished.
Q: II IV III
A:
Q: Please explain Frost Circles for aromatic systems and how they can be used to explain the stability…
A: A question to describe about Frost circles for aromatic systems.
Q: For heterocycles containing nitrogen atoms, determine whether nitrogen’s lone pairs are used in the…
A: Introduction- Heterocyclic compounds are the cyclic compounds in which one or more of the ring…
Q: Is the following compound aromatic? Explain the reasons for your answer?
A: Aromatherapy can be decided by checking three basic steps: I) all carbon atoms are sp2 hybridised.…
Q: For the structures shown below, state the number of pi electrons present in the molecule. ОН НО…
A: According to huckel rule of aromaticity for a molecule to be aromatic it should be cyclic planar and…
Q: is this aromatic, anti-aromatic, or non aromatic?
A: The given compound is : Is this aromatic, anti-aromatic, or non aromatic = ?
Q: Cyclopropenones are described as having aromatic character. How would you account for this, given…
A: Cyclopropenone is an organic compound. Its molecular formula is C3H2O. It contains ketone functional…
Q: Electrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the…
A: Given reaction,
Q: Arrange them in the increasing order of stability by applying the rules of aromaticity for the…
A: Rules of aromaticity: 1) The compound must be cyclic and planar. 2) It must be conjugated. 3) It…
Q: Which of the following lone pairs of purine participate in its aromaticity? N. C
A:
please provide explanation
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

- The compound MON-0585 is a non-toxic, biodegradable larvicide that is highly selective against mosquito larvae. Synthesize MON-0585 using either benzene or phenol as a source of the aromatic rings.14. Which of the following compounds is non-aromatic?Which of the following compounds is aromatic? A. only A and B B. only A and C C. only B and C D.only B
- Aromatic heterocyclic chemistry: State whether the following drugs are aromatic heterocycles and if they follow the 4 aromaticity rules (unsaturation, (4n + 2)pi electrons, cyclic and flat/planar), NAME ANY HETEROCYCLES Vildagliptin, Valbenazine, Levetiracetam, (Dolutegravir/Lamivudine)3) In the mid-1930s, the German theoretical chemist Erich Hückel developed a rule that dealt with the aromaticity of various compounds, which became known as the Hückel Rule. Which (parts) of the compounds listed below are aromatic? Justify your answer based on Hückel's rule. You can treat the rings separately or together as you wish.Which of the following are consistent with the requirements for aromaticity?I. A system with delocalized p electrons in a ring.II. 4n p electrons in the ring.III. All the ring atoms must be carbons.IV. (4n + 2) p electrons in the ring. Give the answer why.
- 3) In the mid-1930s, the German theoretical chemist Erich Hückel developed a rule that dealt with the aromaticity of various compounds, which became known as the Hückel Rule. Which (parts) of the compounds listed below are aromatic? Justify your answer based on Hückel's rule. You can treat the rings separately or together as you wish.image iv: colchicine: a highly poisonous alkaloid,obtained from autumn turmeric and used to treat gout.3) In the mid-1930s, the German theoretical chemist Erich Hückel developed a rule that dealt with the aromaticity of various compounds, which became known as the Hückel Rule. Which (parts) of the compounds listed below are aromatic? Justify your answer based on Hückel's rule. You can treat the rings separately or together as you wish. IMAGE IV: colchicine: a highly poisonous alkaloid, obtained from autumn turmeric and used to treat gout.Why isn't this compound aromatic? Doesn't it fit the 4n+2 rule where there is 6 pi electrons? Or is it not planar, which is why it is not aromatic?
- Describe the following molecules as aromatic,antiaromatic,ornon-aromatic. Hint: sometimes resonance structures can reveal hidden aromaticity...or antiaromaticity.Aromatic heterocyclic chemistry: State whether the following drugs are aromatic heterocycles and if they follow the 4 aromaticity rules (unsaturation, (4n + 2)pi electrons, cyclic and flat/planar), NAME ANY HETEROCYCLES. Draw the structure of the core of any aromatic heterocycles in the drug. Riociguat, Imatinib, Clopidogrel, Glatiramer acetate, AxitinibCompound A exhibits a peak in its 1H NMR spectrum at 7.6 ppm, indicating that it is aromatic. (a) How are the carbon atoms of the triple bonds hybridized? (b) In what type of orbitals are the π electrons of the triple bonds contained? (c) How many π electrons are delocalized around the ring in A?

