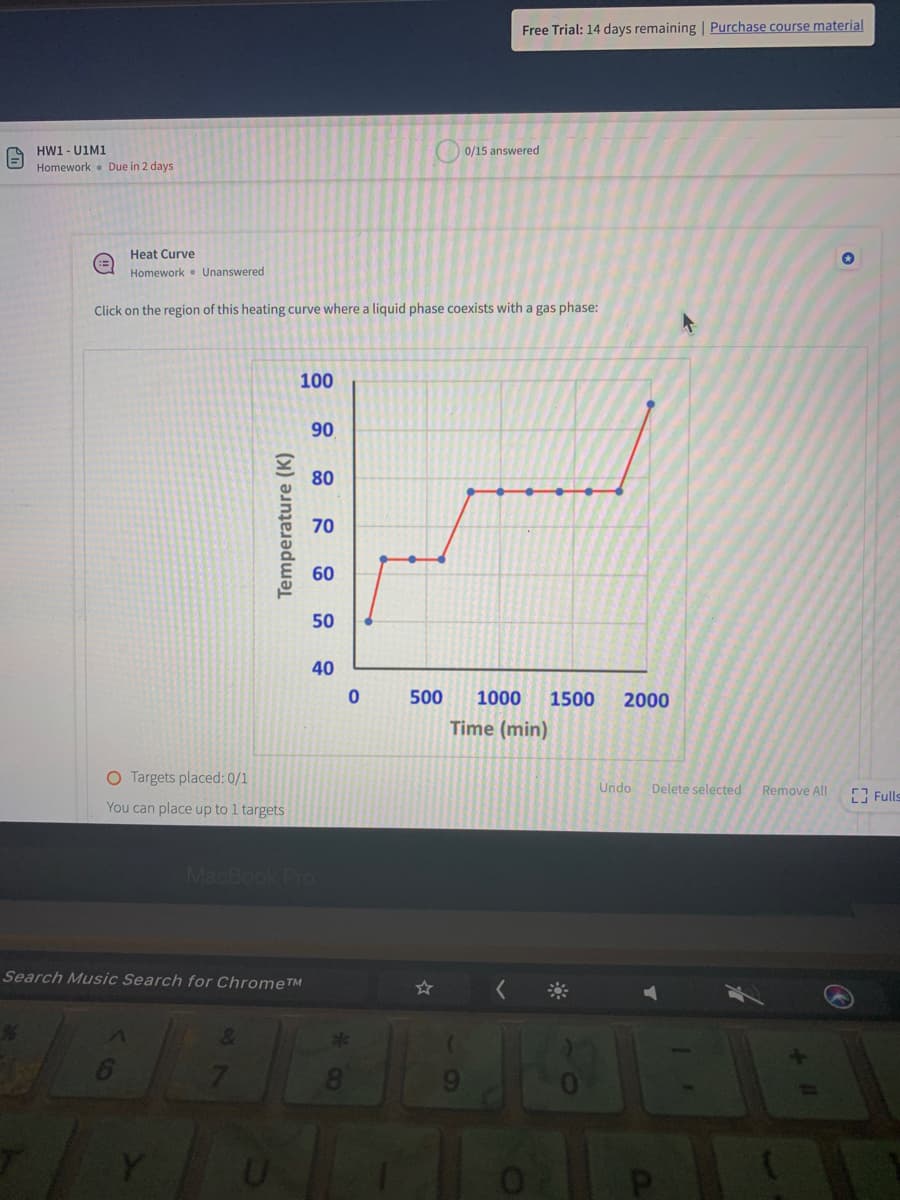
Click on the region of this heating curve where a liquid phase coexists with a gas phase: 100 90 80 70 60 50 40 500 1000 1500 2000 Time (min) Temperature (K)
Click on the region of this heating curve where a liquid phase coexists with a gas phase: 100 90 80 70 60 50 40 500 1000 1500 2000 Time (min) Temperature (K)
Chapter33: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 33.17QAP
Related questions
Question
Transcribed Image Text:Free Trial: 14 days remaining | Purchase course material
HW1 - U1M1
Homework . Due in 2 days
0/15 answered
Heat Curve
Homework • Unanswered
Click on the region of this heating curve where a liquid phase coexists with a gas phase:
100
90
80
70
60
50
40
500
1000
1500
2000
Time (min)
O Targets placed: 0/1
Undo
Delete selected
Remove All
E Fulls
You can place up to 1 targets
MacBook Pro
Search Music Search for ChromeTM
7.
8.
Temperature (K)
Expert Solution
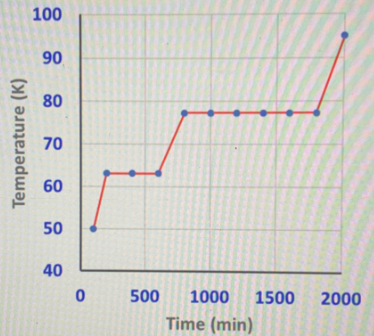
Step 1
The phase diagram given is,
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

