COH H2SO4 (cat) NaOH HO CH3OH B CH3OH 4. H2SO4 (cat) OCH3 H2SO4 (cat) D+E C H20 H20 NaOH G +H H20 OCH3 H2SO4 (cat) HO. Li= H3O* H30* CHO A в он H20 HO
Q: If equal numbers of molecules of H2 (molar mass=2) , O2 (molar mass=32), and N2 (molar mass=28) are ...
A: Different gas have different velocity as their molecular masses are different.
Q: A chemist prepares a solution of potassium iodide (KI) by measuring out 3.0 x 10 umol of potassium i...
A:
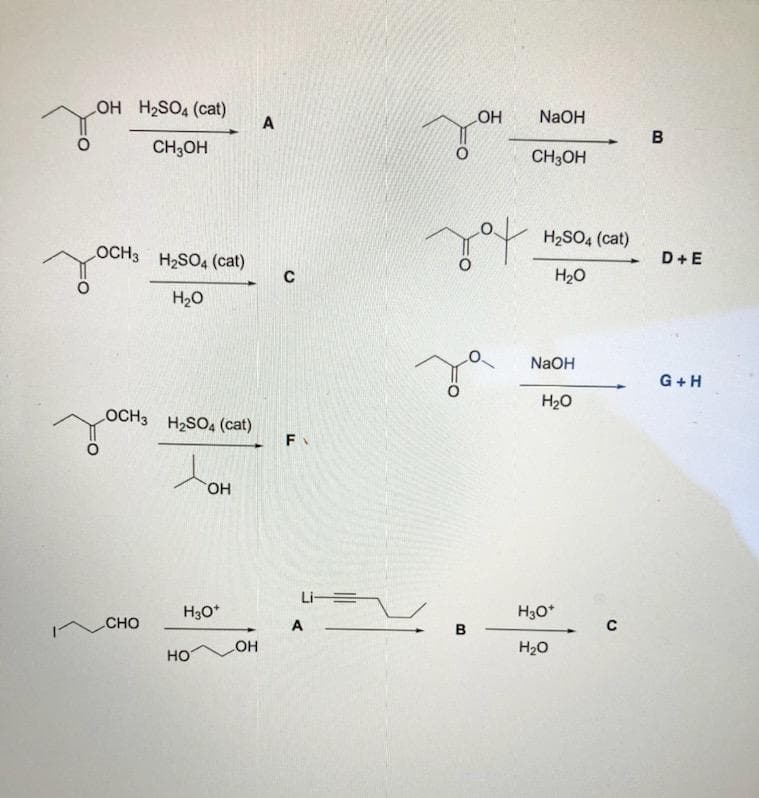
Q: -CH3 PCC, CH,CI, reacting with OH
A:
Q: formulas formed between the given ions. Barium and Chlorine Show solutions. Follow the format in an...
A: a) electronic configuration Ba = 1s2,2s2,2p6,3s2,3p6,3d10,4s2,4p6, 4d10, 5s2 ,5p6,6s2 Cl = 1s2,2s2,...
Q: For each of the following reactions, determine if the AH pxn also represents the AH", Answer with ye...
A: Standard heat of formation is defined as formation of one mole of substances from its constituent el...
Q: 3 ОН NH
A: By following a proper structure one can easily determine the hybridization of an atom.
Q: Determine the pH for the following 0.100 M LiF (Ka = 6.6 x 104) а) b) 3.2x 106 M Ba(ОН)2 2.
A: a. 0.100 M LiF ( Ka = 6.6 * 10-4) b. 3.2 * 10-6 M Ba(OH)2 We have to calculate the pH of the abo...
Q: PROBLEM The iodide ion is oxidized in acid solution to the triiodide ion,I-3, by hydrogen peroxic hy...
A: Given , Reaction : H2O2(aq) +3I-(aq) +2H+(aq) →I3-(aq) +2H2O(I)
Q: Write the empirical formula for at least four ionic compounds that could be formed from the followin...
A: Write empirical formula of the given compounds---
Q: An arctic weather balloon is filled with 25.6 L of helium gas inside a prep shed. The temperature in...
A:
Q: time
A:
Q: The concentration of arsenic in an insectide is determined gravimetrically by precipitating MgNH4AsS...
A:
Q: A 0.025 M solution of compound HX has pH = 2.15 with the given equation below. HX ↔ H+ + X- Which ...
A: Given:: [HX] = 0.025 M pH= 2.15 equation below. HX ↔ H+ + X- Which of the following statements is tr...
Q: For a system using H2SO4, why is the concentration half of the concentration used in the HCl or HNO3...
A: All of the above acid system are strong acid and get completely dissociates in to the solution. In o...
Q: 1. Draw the Product (If applicable) of the Following Reactions: Br HCl
A: Detail mechanistic pathway is given below
Q: Maceration of citrus peels
A:
Q: 1. What is the molar concentration of Aki's disinfectant if she dissolved 5Tbsp. of NACIO (sodium hy...
A: A question based on concentration terms that is to be accomplished.
Q: When 15.0 mL of a 0.5757 M potassium phosphate solution and 0.100 L of 0.7575 M HgNO3 were combined ...
A:
Q: Ammonium perchlorate (NH,CIO,) is a powerful solid rocket fuel, used in the Space Shuttle boosters. ...
A: Ammonium perchlorate decomposes into nitrogen, chlorine, oxygen, and water. The equation for the bal...
Q: -A gas of R= 219 kgm-K undergo a process and results in these changes: AH = %3D 2190 KJ and AU = 145...
A: Given: R = 219 J/Kgm -K ∆H = 2190 KJ ∆U =1458 K Mass = 1.8 Kgm To determine: a) Cv ,Cp and k b) ∆T ...
Q: redox reaction? Oyes O no 2Fe(s) + 3NICI, (aq) 2FEC1, (ag) + 3Ni(s) reducing agent: oxidizing agent:...
A: Reducing agent is a substance that tends to bring about reduction by being oxidized and losing elect...
Q: An average volume of 15.52 mL of HNO3(ag) is required to titrate a 250.00 mL of Ba (OH)2(ag) which h...
A:
Q: What is/are the best reagents (i.e., chemicals for the box below) to carry out the following reactio...
A:
Q: For each reaction in the table below, write the chemical formulae of any reactants that will be oxid...
A: If the oxidation number of an atom decreases which means it is reduced in the reaction. If the oxida...
Q: 2. A gaseous mixture containing 1.5 mol of Ar and 3.5 mol CO2 has a total pressure of 7.5 atm. What ...
A:
Q: On a clear day at sea level, the partial pressure of N₂ in air is 0.78 atm at 25°C. Under these cond...
A:
Q: CH212 Zn-Cu С. EtCuLi PhLi H. MeMgBr D.
A: Organic reactions are those in which organic reactant react to form organic products.
Q: Refer to the following molecule: CH,CH, H. C-Br H,C NO2 How many achiral carbons are there? (Select ...
A:
Q: (a) Calculate the number of moles of iodine which reacted in the titration. (b) Calculate the concen...
A: This is an iodometric reaction because here iodine is titrated which is already produced in the reac...
Q: Methyl acetate is hydrolyzed in approximately 1 mol/L HCl at 298 K. Aliquots of equ plume are remove...
A: Methyl acetate on hydrolysis converted into methanol and acetic acid. It is very slow in pure water ...
Q: Show the steps necessary to synthesize this compound by the Williamson ether synthesis. → t-butyl et...
A: We have to write steps for synthesis of t-butyl ethyl ether.
Q: Each row of the table below describes an aqueous solution at about 25 °C. Complete the table. That i...
A:
Q: 2. Determine the pH for the following 0.100 M LiF (Kа — 6.6 х 104) а) b) 3.2x 10° М Вa(ОН)2
A:
Q: What mass (in grams) of magnesium oxide can be produced from igniting 1.5 g of Mg in Oxygen? (MM of ...
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: 8.14 Predicting the Products of an Addition Reaction
A: Stereochemistry is branch of chemistry in which we deal with arrangement of atoms in three dimension...
Q: e path length
A:
Q: hat volume of
A:
Q: Calculate the volume in milliliters of a 2.18M sodium chloride solution that contains 375. mmol of s...
A:
Q: Problem 2. Consider the following reaction: OH + CO CO2+H The value of the forward rate coefficient ...
A:
Q: For numbers 27 - 30, consider the following: Elements X, Y and Z form a molecular compound XY,Z,, an...
A: Here the given molecule is XY2Z2 in which the central atom contains two lone pair elctrons .We have ...
Q: Ammonium phosphate ((NH4) PO4) is an important ingredient in many fertilizers. It can be made by rea...
A: Consider the given equation is as follows: Reaction: H3PO4 + 3 NH3 → NH43...
Q: A. GIVE THE IUPAC NAME OF TH ОН b) (CH3)3СОН c) CH3CH2CH2OH d) OH H3C CH3
A: Following steps are required for IUPAC naming. 1-Find and name the longest continuous carbon chain t...
Q: ) Write the equation for the reaction between propylamine and HBr. B) Write the reaction for the b...
A: Acid and base react to form salt . The first equation is an example of it. Hydrolysis of amide forms...
Q: Predict the products of the reaction below. That is, complete the right-hand side of the chemical eq...
A:
Q: A chemist prepares a solution of sodium nitrate (NaNO,) by measuring out 270. g of sodium nitrate in...
A:
Q: A major component of gasoline is octane (C3H,g). When liquid octane is burned in air it reacts with ...
A:
Q: A chemist must dilute 0.0757 L of 11.6 M aqueous silver nitrate (AGNO, solution until the concentrat...
A:
Q: 4.91 mL of NaOH solution was required to reach the endpoint when titrated against 0.1005 g of potass...
A: Here 4.91 ml of NaOH solution was required to react with 0.1005 g of KHP dissolved in 50ml water.We ...
Q: calculate the titration curve of a 0.01 m aqueous solution of NH3 with 0.03M HBr. A minimum of five ...
A: Ammonia reacts with HBr as per the following reaction: NH3+HBr↔NH4++Br-
Q: Why is - CN a weaker leaving group than - NHCH3.
A: Weak Bases are the Best Leaving Groups. (Recall that the stronger the acid, the weaker the conjugate...
Step by step
Solved in 2 steps with 2 images

- Solve this bahin hurry up 2abc Show stepo 10minssend help, I'll give you a rate if you send the correct answer. please don't answer like "7.08x10^23" because it is not accepted sometimes or they need whole number w/o x10^23 something. thanks!You are in lab doing an experiment, so far this is your data: Pb(CH3COO)2 (aq) + H2S (aq) → PbS (s) + CH3COOH (aq) 10g Pb (CH3COO)2 + 3g H2S produce 11.56g of led sulfate + 1.44g S 18g Pb (CH3COO)2 + 1.56g H2S produce 11.56g of led sulfate + 8.00g S Based on this data. What type of reaction is it? How many grams of sulfur do you need to completely react 10.00g of Pb. Balanced equation.
- Please help me solve the following question and make sure its correct 100%, thank you sm1. What is the mEq of silver nitrate? 170 0.0170 0.170 17.0 2. How much (mL) silver nitrate is required for the standardization? 4.995 49.950 49.590 45.990 * MW of silver nitrate = 170 g/mol * Please take note that Im looking for the silver nitrateLet me tell you about their task: The students were provided with 5 drinks (sports/soft drinks) and they were asked to determine the glucose concentration in these drinks in the units of g/100mL. (Why these units? Well, once the students have the concentrations in g/100mL they will be able to compare their values with the nutritional values given on the drink bottles’ labels). The samples of the five drinks were all diluted 1/100 (i.e. by a factor of 100). This was an essential step in the method because, without it, the machine used to analyse the glucose concentration (spectrophotometer) would have given an error as the concentration would have been too high for accurate detection. What this means for the students is that the dilution factor will have to be taken into consideration in their calculations (remember the aim is to calculate the concentration in the original drink and not in the diluted drink). The students decided to save time and form five groups where each group was…

