Chapter14: Chromatography
Section: Chapter Questions
Problem 9P
Related questions
Question
3
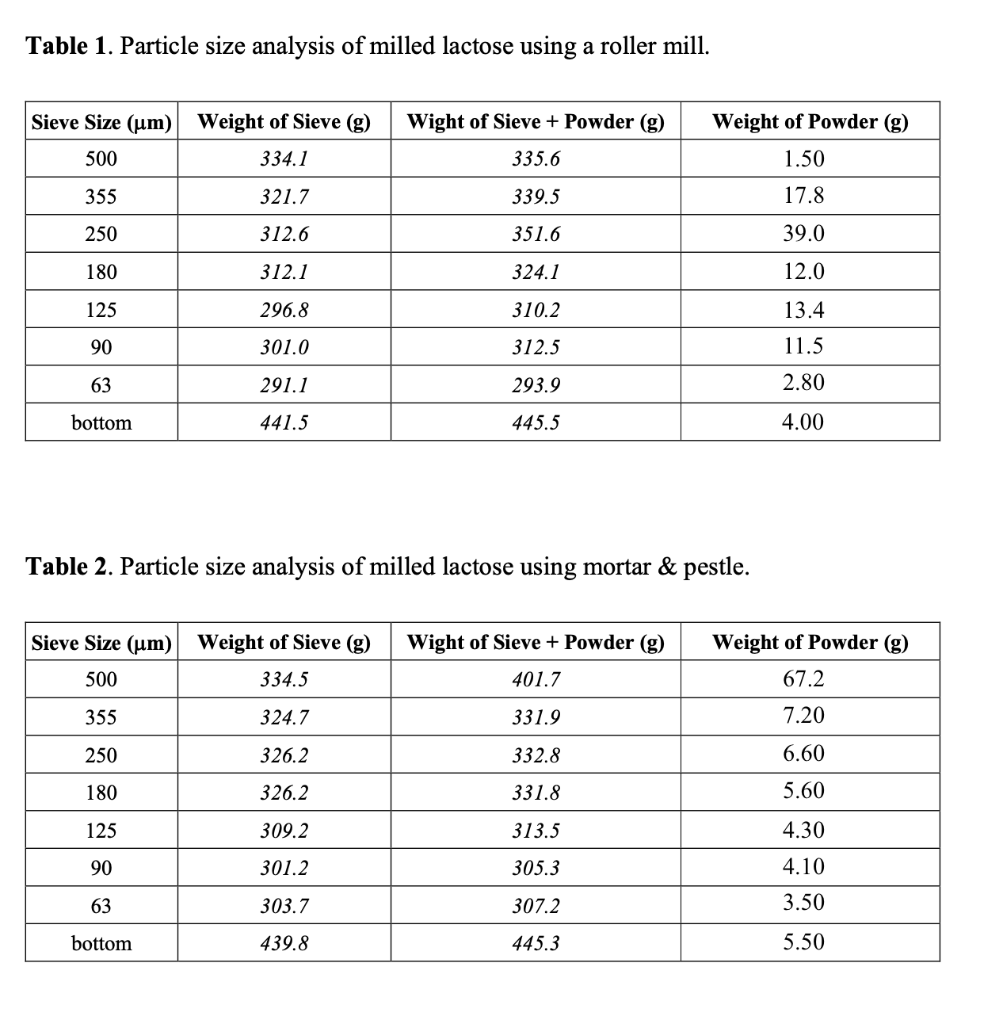
Transcribed Image Text:Table 1. Particle size analysis of milled lactose using a roller mill.
Sieve Size (um) Weight of Sieve (g)
Wight of Sieve + Powder (g)
Weight of Powder (g)
500
334.1
335.6
1.50
355
321.7
339.5
17.8
250
312.6
351.6
39.0
180
312.1
324.1
12.0
125
296.8
310.2
13.4
90
301.0
312.5
11.5
63
291.1
293.9
2.80
bottom
441.5
445.5
4.00
Table 2. Particle size analysis of milled lactose using mortar & pestle.
Sieve Size (um) Weight of Sieve (g)
Wight of Sieve + Powder (g)
Weight of Powder (g)
500
334.5
401.7
67.2
355
324.7
331.9
7.20
250
326.2
332.8
6.60
180
326.2
331.8
5.60
125
309.2
313.5
4.30
90
301.2
305.3
4.10
63
303.7
307.2
3.50
bottom
439.8
445.3
5.50

Transcribed Image Text:6. Comment on the strengths and limitations of particle sizing using sieve analysis. Suggest
an alternative sizing technique and justify.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning