Compare the de Broglie wavelength of a proton moving at 1.30×10' miles per hour (5.81x10 m/s) to that of a golf ball moving at 70.0 miles per hour (31.3 m/s) and an alpha particle with a speed of 3.40×10' miles per hour (1.52×10' m/s).
Compare the de Broglie wavelength of a proton moving at 1.30×10' miles per hour (5.81x10 m/s) to that of a golf ball moving at 70.0 miles per hour (31.3 m/s) and an alpha particle with a speed of 3.40×10' miles per hour (1.52×10' m/s).
Chemistry: Principles and Practice
3rd Edition
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Chapter7: Electronic Structure
Section: Chapter Questions
Problem 7.45QE
Related questions
Question
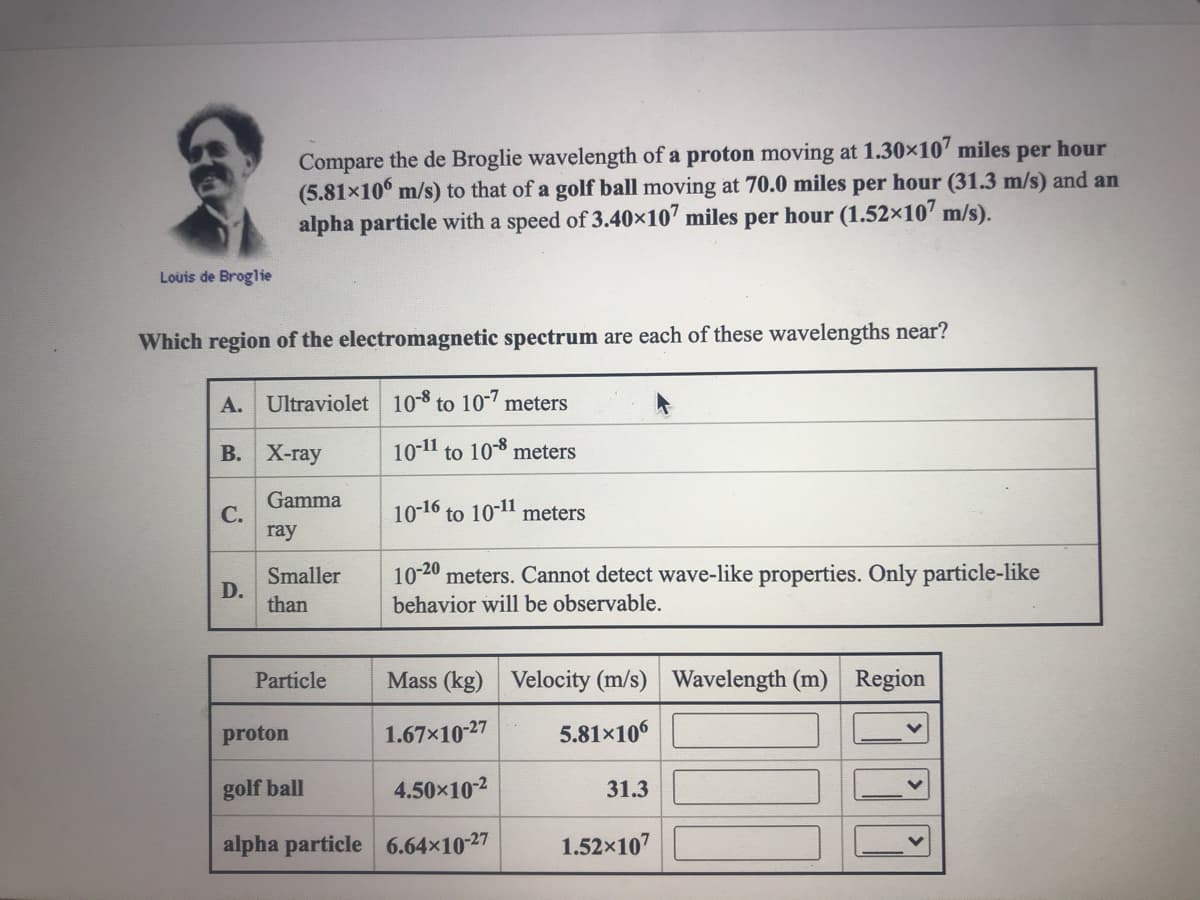
Transcribed Image Text:Compare the de Broglie wavelength of a proton moving at 1.30×10" miles per hour
(5.81x10° m/s) to that of a golf ball moving at 70.0 miles per hour (31.3 m/s) and an
alpha particle with a speed of 3.40×107 miles per hour (1.52×107 m/s).
Louis de Broglie
Which region of the electromagnetic spectrum are each of these wavelengths near?
A. Ultraviolet 10-8 to 107 meters
В. Х-гау
10-11 to 108 meters
Gamma
С.
ray
10-16 to 10-11 meters
10-20 meters. Cannot detect wave-like properties. Only particle-like
Smaller
D.
than
behavior will be observable.
Particle
Mass (kg) Velocity (m/s) Wavelength (m) Region
proton
1.67×10-27
5.81×106
golf ball
4.50x102
31.3
alpha particle 6.64×10-27
1.52x107
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax