Complete and balance the following equations: (a) Mg;N2(s) + H,0(I) (b) C3H;OH(1) + 02(8) (c) MnO2(s) + C(s) (d) AIP(s) + H2O(1) (e) NazS(s) + HCI(aq)
Complete and balance the following equations: (a) Mg;N2(s) + H,0(I) (b) C3H;OH(1) + 02(8) (c) MnO2(s) + C(s) (d) AIP(s) + H2O(1) (e) NazS(s) + HCI(aq)
Chapter18: Introduction To Electrochemistry
Section: Chapter Questions
Problem 18.7QAP
Related questions
Question
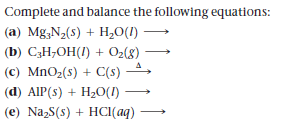
Transcribed Image Text:Complete and balance the following equations:
(a) Mg;N2(s) + H,0(I)
(b) C3H;OH(1) + 02(8)
(c) MnO2(s) + C(s)
(d) AIP(s) + H2O(1)
(e) NazS(s) + HCI(aq)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 5 images

Recommended textbooks for you


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning