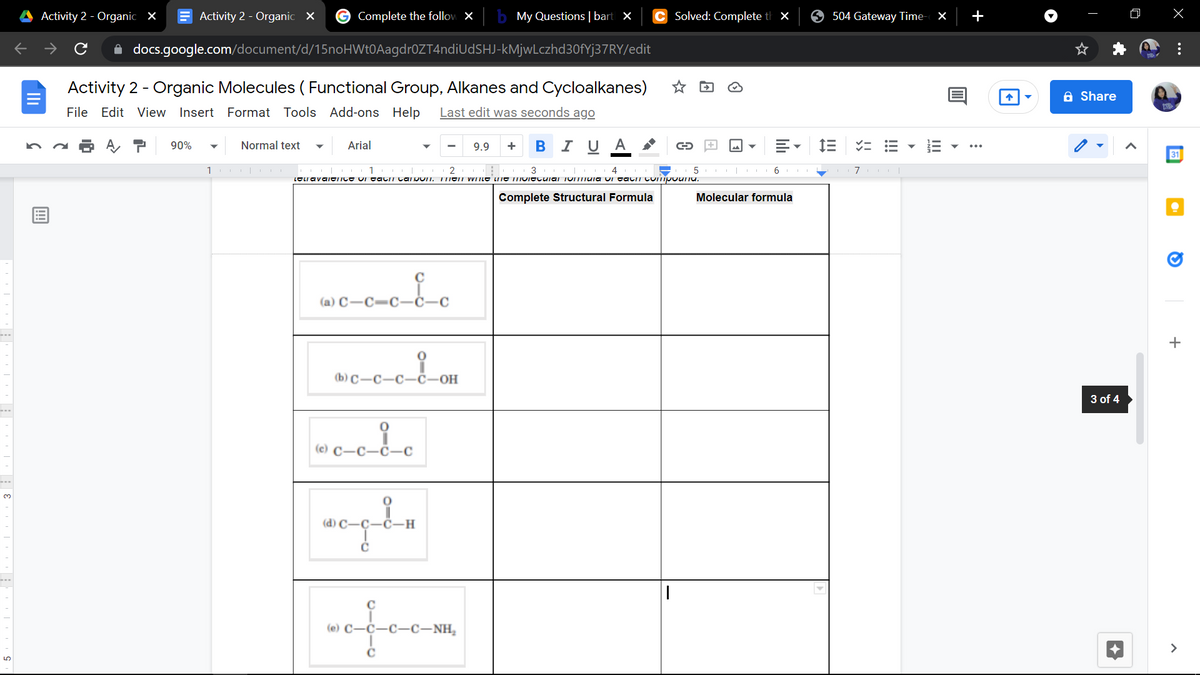
Complete Structural Formula Molecular formula (a) C-C=c (b) C-C-C-č-OH c-c- -c-c-NH,
Q: нно HHO Draw a diagram to show how the monomers to the right could be joined to form a polymer and…
A:
Q: Give the name and structure of at least two examples of each of the following: heterocyclic amino…
A: Hi! As you have posted multiple questions, I will be answering the first 3 questions for you. If you…
Q: Functional Group Name Structural Diagram (draw all bonds) Found where in the body??? Hydroxyl H -N H…
A: Functional groups can be described as the groups of molecules that get linked to organic molecules…
Q: In the following monosaccharide hemiacetal, identify the anomeric carbon atom. Identify the…
A: Monosaccharides are compounds that cannot be further hydrolyzed into still smaller molecules. These…
Q: The name of the structure labelled X is:
A: The trachea is also known as the windpipe. It is a hollow tube that joins the larynx and lungs.…
Q: K Name and match structure J with its function. [HINT: The structure name is a single word]
A: These structures as mentioned in the hint are of the Echinodermata phylum. These are the group of…
Q: Functional group as indicated by letters A,B,C and D?
A: Tamiflu is a drug made from the starting molecule shikimic acid. Tamiflu is a prodrug usually used…
Q: Why does the structure H-C=C-H fail to make sense chemically?
A: A hydrocarbon is an organic compound that is composed of hydrogen and carbon atoms. The four classes…
Q: Draw a structural formula for each compound. (a) trans-2-Methyl-3-hexene (b) 2-Methyl-3-hexyne (c)…
A: Structural formula for each compound are given in the next step,
Q: Draw skeletal structure for the following structures: a. (25, 3R)-dichloropentane b. (25, 3R, 4R)-2…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: ELEMENTS PRESENT EXAMPLES FUNCTIONAL GROUP BIOMOLECULES FOUND 1. 2. 3. СООН 4. Monosaccharide…
A: The method of electrophoresis is used to separate biomolecules based on particle charge, particle…
Q: All of the following have terpene structure, except Cholecalciferol Squalene Carotenoids O…
A: Terpenes are hydrocarbons composed of 5 carbon atoms attached to 8 hydrogen atoms. Terpenes consists…
Q: IUPAC name for the structure given. (DO NOT write "cis or trans") * H
A: Since this is a structure having a -COOH functional group and rest all is a CH backbone thus it…
Q: In the structure shown, which arrow is pointing to a peptide bond? OH LOH NH2 H OH HO-P=0 H. `A ÓH…
A: Peptide bond: It is a covalent bond used to join two amino acids to form a peptide chain. Generally,…
Q: Draw the possible structural formulae of compounds P, Q, R, S, T and U. Label the peptide bond(s)…
A: Amino acids are organic compounds that contain an alpha-amino and an alpha-carboxylic acids group.…
Q: Molecule Amino Acid (Glycine R=H) Glycerol a-glucose Molecular Formula Variable C2H5NO2 Structural…
A: Biomolecules are carbon-based organic compounds produced by living organisms. They are primarily…
Q: Match the molecular geometry to its name. 1. Tetrahedral 2. Linear 3. Octahedral SUSMIT
A: Molecular geometry is the 3-D plan of the atoms that comprise a molecule. It incorporates the…
Q: Draw the structure of the wax composed of stearic acid and a straight-chained alcohol with 30 carbon…
A: Waxes are simple lipids composed of fatty acids (saturated and unsaturated) which are esterified to…
Q: using the expanded structure of Levothyroxine. Determine the molecular geometry of each central…
A: To determine the geometry of the carbon atoms in Levothyroxine , we would draw the structure and see…
Q: Construct the primary structure of the following oligopeptides, preferably in their zigzag formulas.
A: Amino acids Proteins are the polymers of nitrogenous compounds called amino acids. Each amino acid…
Q: How many H2O particle were produced by creating the following How many peptide bonds were produced…
A: Introduction: Amino acids are joined together by a condensation reaction between carboxylic and…
Q: Instructions: Differences between Cis-fatty acid VS. Trans fatty acids Use white bond…
A: Unsaturated fatty acids are in configuration that is called cis, but partially hydrogenation can…
Q: Associated Structure Name Elements Involved Acid/base/neutral (pH 7) Charge at pH 7 Macromolecule…
A: A biomolecule is basically a carbon-based organic compound. It is produced by living organisms.…
Q: Functional Group Name Structural Diagram (draw all bonds) Found where in the body??? 7. 8.…
A: Introduction:- Alcohol is dispersed throughout the body's fluids, exposing most tissues, including…
Q: Draw the complete structural formula of arachidonic acid (Table 23.1) in a way that shows the cis…
A: Arachidonic acid is a polyunsaturated fatty acid that was synthesized from dietary linoleic acid and…
Q: 7. general structure of a TAG 8. structure of glycerol 9. structure of a sterol
A: You have asked question with multiple subparts. I will answer 1st three subparts. Triacylglycerol -…
Q: Given the structure below: CH,OH но он ÓH HỎ Which of the following choices corresponds to the…
A: The Fischer form shows the linear form of the monosaccharide while the pyranose is the…
Q: In the structure shown, which arrow is pointing to a phosphoester bond? OH HO NH2 H ОН HO-P=0 `A ÓH…
A: DNA is the hereditary material in almost all organisms except some viruses. The basic unit of DNA is…
Q: pindolol
A: In normal medicine tests in man and animals, beta-adregenic blocking agents attenuates increases in…
Q: Name the following molecular: CH3-CH2-CH2-CH2-CH2-CH-OH
A: An alcohol is an organic compound with an aliphatic carbon atom bearing the hydroxyl (OH) functional…
Q: te the structure for a generic triacylglyceride at pH 7.4.
A: The simplest lipids which are derived from fatty acids are the triacylglycerols which are also known…
Q: H HN-C-C-OH H-C-CH, CH2 CH3 The following short passage is about the compound with chemical…
A: Protein is a polymer formed from amino acid joined by peptide bond. There are twenty standard amino…
Q: In the space provided, draw a Haworth projection for the beta anomer of the following monosaccharide…
A: Haworth projection: It is used for cyclic sugars ( pyranoses and furanoses) It clearly indicates…
Q: Which of the functional groups listed in shown Table can function as hydrogen bond donors? As…
A: Atoms can neither be created nor be destroyed. Specific groupings of atoms within molecules which…
Q: Differences between Cis-fatty acid VS. Trans fatty acids
A: ANS: Unsaturated fatty acids have one or more double bonds. Each double bond may be in a cis or…
Q: The chemical structure of ethane (C2H6) is provided below. Which of the following isomers is ethane…
A: Option (5) none of the above is correct answer. (1) Ethane only have one form. It has too few…
Q: HH H H H C-C- Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η Η H H H-C-O C- онн C-C- H H H H H H H CH, H H…
A: There are many macromolecules present in the living organism. They include carbohydrates, lipids,…
Q: For the complete name of the Hawort structure given, (use the structure given fischer projection) *…
A:
Q: Compounds A and B was discovered by a group of scientists. Compound A is a dipeptide, optically…
A: Proteins are polymers of amino acids. In protein amino acids are joined together by peptide bonds.…
Q: Functional Groups: MATCHING 1. sulfhydryl 2. carboxyl 3. carbonyl 4. phosphate 5. amino а.- ОН b. -…
A: Macromolecular:- A molecule containing a very large number of atoms, such as a protein, nucleic acid…
Q: condensed structural formula for glycerol tricaprylate (tricaprylin).
A: The condensed structural formula of a molecule is somewhat identical to its normal structural…
Q: Jsing the picture provided, match the correct name of each of the functional groups highligthed in…
A: The correct name of each of the functional groups highlighted in blue is given below:
Q: structural feature of terpenes?
A: Terpenes are organic compounds that are made up of isoprene subunits (C5H8)n. These are unsaturated…
Q: Structural formula for Cutte Alkene With doulde bond an that Shaws lis isomerismm,
A: Ans. In cis isomerism, functional groups lie on the same face of the carbon chain. The molecule…
Q: CH2OH CH2OH H H OH H H H OH H OH H ОН H OH H OH What is the molecular formula of Maltose?
A: Carbohydrates are polyhydroxy compounds, composed of carbons, hydrogen, and an oxygen atom. It is…
Q: Please draw all four bonds at chiral centers. Use wedge and dash bonds at chiral centers only. Do…
A: Enantiomers are the compounds that are mirror image to each other but are not superimposable .
Q: cis, cis-9,12-OCTAdecadiENoic acid Enumerate the number of carbons and unsaturations (double bonds)…
A: Linoleic acid is also known as Ocatadecadienoic acid is a polyunsaturated essential fatty acid that…
Q: 5. Give the molecular formula of the functional group that is missing. H H,N-C-? R a. NH+ b. CH3 с.…
A: First, we will analyze the given structure and decide which can be the functional group. There is an…
Q: Construct the primary structure of the following oligopeptides, preferably in their zigzag formulas.…
A: Proteins or peptide is the sequence of amino acids which is joined together by peptide bond…
Q: For the first part, draw a Fischer projection formula for the enantiomer of each of the following…
A: Fischer projection is a method of that represents the 3-D structures of molecules on paper,…
How to complete this?
Step by step
Solved in 2 steps with 1 images

- Answer should be 107 ATP. Can u help me to explain to me, please? I stuggled.BIOMOLECULES - Please answer the questions properly. - Multiple choice 1. Which of the following best describes the relationship between a reactant molecule and an enzyme? A. it is a permanent mutual alteration of structure B. it is a temporary association C it is an association stabilized by a covalent bond D. it is one in which the enzyme is changed permanently 2. Which of the following best describes an apoenzyme? A. it is a complete, biologically active conjugated enzyme B. it is a prosthetic group C. it is a protein portion of an enzyme D. it is a non-protein groupQuantitative Estimation of Amino Acids by Ninhydrin http://vlab.amrita.edu/?sub=3&brch=63&sim=156&cnt=2 can u help me with question 2 of the assignment questions Based on the experimental data provided, estimate the amount of amino acid in the given unknown solution by Ninhydrin method. SI No. Volume of standard amino acid solution (ml) Amount of amino acid (µg) OD at 570nm 1 Blank 0 2 0.2 0.12 3 0.4 0.25 4 0.6 0.45 5 0.8 0.55 6 1.0 100 0.68 7 Unknown (0.5ml) 0.41
- VII - BIOMOLECULES Instructions : - Answer the question properly. - Please do not copy here in Bartleby - Explain why did you choose that answer in 3-5 sentences.topic about : precipitation of proteins by heavy metal salts 3. Will the precipitate formed after treating egg white with strong acid dissolve or not in addition of excess acid? Explain why. ____________________________________________________________________ ____________________________________________________________________Need help, please. Answer choices are provided below for drop-down questions. 1. The overall charge of this protein at pH 7 is Drop-down answer choices are [positive, negative, or neutral] 2. In the first dimension of 2-D PAGE, this protein's isoelectric focusing point is Drop-down answer choices are [between 4 and 6, between 6 and 8, or between 8 and 10] 5. A mole of the above protein weighs ____ g. For 1 mL of a 5 mM solution of the above protein, you would need to weigh out ____ Drop-down answer choices are [g, mg, mcg, ng, or pg] (number and unit, at least one non-zero integer to the left of the decimal point, don't round)
- Separation of Amino Acids by Thin Layer Chromatography Lab Questions 1. Describe in detail Thin layer chromatographic experiment. Example: the theory behind it, how youwould prepare the materials to spot on the plate with different mobile amino acids and unknown andhow TLC Plate is developed and the reasoning behind which solvent/ solvent mixture should be used,along how to correctly identify of the unknown. 2. Calculate the Rf value if a solute travelled 5 cm from the base spot and the solvent front is 10 cmfrom the origin? 3. In a TLC experiment using a 70:30 mixture of Petroleum ether and ethyl acetate, a student noted thedevelopment of spots in the origin, what can you suggest about this observation?PLEASE HELP 1. How many chirality centers does ribose have? Identify them.1. The following question tests your lab skills as well as basic knowledge understanding: (1). In Quant Lab, primary standard is defined as _______________________________________________________ . (2). In Quant Lab, secondary standard is defined as ______________________________________________________. (3). In your Lab9-Vinegar Titration, the perceived primary standard material is__________________________________. (4). In Lab9, the perceived secondary standard substance is _________________________________________________. (5). In your Lab9, the balanced titration reaction is _____________________________. It belongs to ___________ titration. At your titration equivalence(end) point, its pH equaled to ~ ___ (rough value). The pH indicator used in the NaOH solution standardization was _______________ while the pH indicator employed in the vinegar determination was _____________ ____________________ In your Lab9 report, you drew the derivative titration curve to help…
- BIOMOLECULES - Please answer the questions properly. - Multiple choice 1. Chymotrypsin is a digestive enzyme that breaks down proteins. From the following attributes, which one does not characterize chymotrypsin? A. A thiol nucleophile B. Performs proteolysis C. A tetrahedral intermediate D. A catalytic triad of serine, histidine, and aspartate 2. Evaluate the secondary structure of proteins. Which of the following differentiates alpha structures to those beta structures? ? A. alpha structures are helices and beta structures are pleated sheets B. alpha structures are primary and beta structures are secondary C. alpha structures are parallel and beta structures are antiparallel D. alpha structures are L and beta structures are DPlease ASAP. Thankyou. Question 6 Assuming the atomic radii are as follows: K+ > Na+ > Mg2+ and the electronegativities are as follows: Mg2+ >>Na+ >> K+ Which hydrated ion is most unlikely to pass through a small pore in the cellular membrane? K+ Na+ Mg2+ not enough informationSeparation of Amino Acids by Thin Layer Chromatography Lab Questions 7. Why must a pencil be used for drawing the line and spotting? 8. How will the Rf value of a compound be affected if the developing solvent is allowed to run off thetop of the TLC plate (e.g. the TLC plate is allowed to remain in the developing chamber after thesolvent front has reached the top of the plate.)? Explain. Be specific. 9. Describe what an iodine vapor chamber is and explain how does it work? (Hint: Backgroundinformation – showing spots up chemically talks about two methods using Ninhydrin and Iodinecrystals)



