Composition (at% Zn) 100 2200 20 40 60 80 1200 2000 atL Liquid 1000 1800 B+L H1600 800 1400뜨 8+L -1200 600 1000 800 400 te 600 %3D 200 400 100 20 40 60 80 (Cu) Composition (wt% Zn) (Zn) Temperature ('C) Temperature (*F)
Q: Material Gold Copper Aluminum Stainless Steel Water Ethyl Alcohol Ice α (°C-¹) 1.65×10-5 2.3x10-5…
A:
Q: Complete the table. a. 1245 kg 1.245 x 10° g 1.245 x 10° mg b. 515 km dm cm c. 122.355 s ms ks d.…
A: Write the conversion of Kilometer into decameter. Write the conversion of Kilometer into…
Q: pectin was studied. With this purpose, a 75°Brix concentrated industrial juice was assayed, and by…
A: The data given for the juice was plotted and then curve fitting was done to obtain the best curve…
Q: 32. Three liquid substances of the same mass and have an S.G. of 1.50, 0.885 and 1.2 were being…
A:
Q: Calculate the volumetric analysis for Nitrogen in % given the following gravimetric analysis.…
A: Write the % of nitrogen by gravimetric analysis. %Ng=75.53%
Q: Conductivity Concentration of KCL vs Conductiv 160 140 120 100 80 60 40 20 0.1 0.2 0.3 0.4 0.5…
A:
Q: mixture. a. 8.33°C b. 3.83 °C c. 5.83 °C d. 10.33 °C
A: solution is given as under: I have solved the problem but none of the answers are matching. The…
Q: Thermistors are semiconductors used for measuring temperature. The electri- cal resistance of a…
A:
Q: The empty cylinder of a soil-coring tool weighs 47.27 g, and holds a total volume of 220 cm3. The…
A: Given data: Mc= 47.27 gVt = 220 cm3Average particle density=2.65 g/cm3 Need to determine the…
Q: Q2: From Figure (1) Find the Equation of Liquidous line (AE) ,(EB) and prove the eutectic pint…
A: Liquidous line AE :-which separate the phase of liquid line to solid+liquidi.e.L⇌@650°Cαsolid +LLine…
Q: The performance of an element can be written as p = fi(F).f:(G)fs(M). F, G and M in the performance…
A: Following is the answer to the above question
Q: A certain radioactive material known to decay at a rate proportional to the amount present. If…
A: dN/dt = kt is integrated to arrive the solution. No = 50 mg
Q: The pipe size of a pneumatic conveyer to convey plastic pellets with bulk density of 30 lb/ft2 using…
A: solution 1:- correct option is A As per general rule the pneumatic conveying system can carry 2…
Q: An perfect gas of a specific composition At 10 g/mol, the molecular weight and specific heat ratio…
A:
Q: The following table provides compressive strength of an alloy as a function of concentration of some…
A: Write the given data. Concentration (x) 5 7.5 10 12.5 15 Compressive strength f(x) 26.4 29.8…
Q: Temperature = Temperature = Composition (at% Sn) 20 40 60 80 100 327 °C 600 300 Liquid 500 Liquid a…
A:
Q: Constants I Periodic Table A cylindrical pipe has inner radius R1 and outer radius R2 . The interior…
A: The cross-section of the pipe is drawn as
Q: 20. Three liquid substances of the same mass and have an S.G. of 1.50, 0.835 and 1.2 were being…
A:
Q: Air, assumed to be an ideal gas with k = 1.40, fl ows isentropicallythrough a nozzle. At section 1,…
A: Given data: The temperature at the section 2 is, T2 = -50 0 The ratio of specific heat is, k =…
Q: The density matrix of a pure quantum state ly) is defined as p = 1)land the density matrix of the…
A:
Q: The kinetics of some phase transformations obey the Avrami relationship. Using the fraction…
A:
Q: QUESTION 5 Given the Volumetric Analysis of a Natural Gas: CH4 = 67.3% Nitrogen = 9.6% 0₂ = 2.7%…
A:
Q: Q8. The performance of an element can be written as p = fi(F).f2(G)f3(M). F, G and M in the…
A:
Q: Strain Stress (Мра) 0.002 220 STRESS VS. STRAIN 0.003 340 450 0.004 360 400 0.005 400 350 0.006 420…
A:
Q: 1. The phase diagram of an alloy compromised of components A and B is shown below. For this problem,…
A:
Q: An aluminum plate measuring 15 cm long, 10 cm wide and 1 cm thick was cooled in air. At the time of…
A: Given: Dimension of aluminium plate l=15 cm b-10 cm h=1 cm T∞=230C Aluminum data: density = 2702…
Q: mix two fluids together, and I use the same mass of each fluid. They are completely miscible; in…
A: Composition is the collection of mole fractions that represents a system's molar chemical…
Q: 1- The shape of phase diagram draw by lever rule is * Liquidus-A'bB' B' Liquid a L+S d Solidus- A'dB…
A: Given phase diagram of composition A and B
Q: A generic property is being transported through a fluid of constant cross-sectional area at…
A: The data given is, Concentration of property at point 1, Γ1 = 0.015/unit volume Concentration of…
Q: The following table shows data collected to measure the thermal conductivity of steel specimen of 30…
A: Thermal Conductivity: It is nothing but the heat carrying capacity or ability to transfer of heat.…
Q: A pan with initial temperature -25C is put in a room with temperature 25C. After half an hour, the…
A: Given: The initial temperature of the object, To = - 25 degree The room temperature, Ti = 25 degree…
Q: 33) (USCS units) What's the increase in length of a steel bar whose length 100ft, if the bar is…
A:
Q: The equipartition theorem of energy in classical statistical mechanics says that the contribution to…
A: sol:Given data:1)Mixture of gasesIdeal, Classical and Diatomic2)MoleculesNα3)Specific…
Q: Q1) The solid phase of a point (in the two-phase region) in the Bi-Sb alloy contains 80 at% of Sb.…
A:
Q: The results of a temperature jump test are shown below. At one hour, the temperature was suddenly…
A: Given: Initial temperature→T1=950 K Final temperature→T2=1000 K The equation for calculating…
Q: A 327°C 600 300 290°C Liquid liquidus 500 Solidus 232°C F a +L 400 200 185°C +L 183°C 61.9 18.3…
A: given; at temperature 1810c overall composition (wt % Sn) (W1)=80andat temperature 1850c overall…
Q: Ceramics Metals 1000 Composites - Tungsten Iron/Steel Titanium SiC Al,0, Si,N. Zro2 CFRC 100…
A: Material Material is a mixture of substances which makes a object. Substances are made by molecules.…
Q: M4.17 Asteel shaft has a starting diameter= 15.00 mm. It is to be inserted into a hole in an…
A: Refer Table 4.1, “Volumetric properties in U.S. customary units for selected engineering materials”,…
Q: M4.18 (Sl units) A bridge to be fabricated of steel girders is designed to be 500 m long and 12 m…
A: Given:
Q: QUESTION 1 Use the Pb-Sn diagram below to answer the following questions. 350 327°C Liquid, L 300…
A: For solution refer below images.
Q: In the Fig. 2 below, let Ki = K2 = K and ti t=t. %3! T- Fig. 2 (a) Let Tr 0 °C and T= 200 °C. Solve…
A: According to the information we have heat flow of the system, where…
Q: A forensic chemist is given a white solid that is suspected of being pure cocaine (C17H21NO4 , molar…
A:
Q: term dT/dx Where T is temperature and x is length, in Fourier law conduction, is called Temperature…
A: The Fourier Law suggests that , heat transfer is directly proportional to the temperature gradient.
Q: Consider an alloy containing 30 wt% Sb. If the alloy is held at 400 oC, how much of the alloy is in…
A:
Q: Which of the following is FALSE regarding the continuum assumption of fluids? Select one: a.…
A: 1. According to the continuum theory, the average value of any fluid quality inside the REV tends…
Q: M4.16 Determine the increase in length of a steel bar whose length 100 in, if the bar is heated from…
A: Refer Table 4.1, “Volumetric properties in U.S. customary units for selected engineering materials”,…
Q: Suppose that you have measured the strain aging kinetics of a high-strength low-alloy (HSLA) steel…
A: As we know that for high strength alloy(HSLA) steel,the stranging kinetics -given data-…
Q: Composition (ars C) 10 15 20 1538C Lever Rule: 1493°C 400 Wa = 2500 R+S 1394C 200 1147C Wp R+S 2.14…
A: Answer is attached below.
Q: Complete the following table for refrigerant-134a. Show your work and explain how you got your…
A: From row 1 in the table, the given pressure, P = 80 psia.The enthalpy is given as, h = 78…
Q: ) Assuming the effluent from the pulp and paper plant has a daily discharge of 15 m3/s, calculate…
A: Given: pulp and paper plant discharge is 15 m3/s. BOD5 concentration of 10 mg/L, and a TSS of 20…
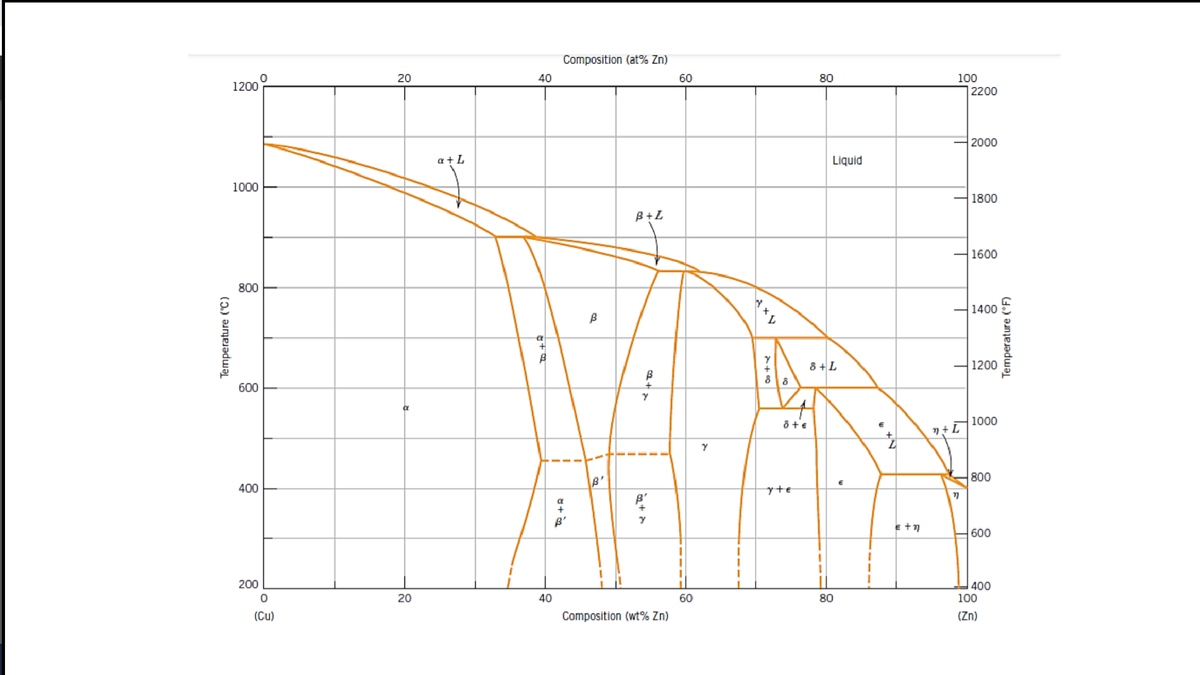
Calculate the invariant reactions for Cu-Zn
|
Reaction (eutectic eutectoid peritectic peritectoide) |
Temperature | Transformation |
Step by step
Solved in 2 steps with 3 images

- 7 Advanced Thermodynamics With compulsory diagrams or graphs. 1)a) What are the methods to destroy state of superconductivity? Discuss in details with diagrams. 1)b) Discuss with year wise progress made in superconductivity research and application till date.In an electrochemical cell, a metal anode lost 0.227 g while a total volume of 0.08149 L of hydrogen gas was produced. The temperature was 298 K and the barometric pressure was 745.4 mm Hg. At 298 K, the vapor pressure of water is 23.8 mm Hg. How many moles of hydrogen gas were collected?1. Energy consumption in a country from 2001 to 2011 increased from 1041.3 million to 1526.1 million BOE (barrels of oil equivalent). If the high calorific value (HHV) of the oil is 141,000 kJ/gallon what is the increase in energy consumption over 10 years in Joule units. What is the rate of energy consumption per year? 2. Based on data on the rate of energy consumption per year from question no. 1, if the growth rate energy 2.5% per year, calculate the doubling time (td) and what is the approximate rate of energy growth in 2021.
- A forensic chemist is given a white solid that is suspected of being pure cocaine (C17H21NO4 , molar mass = 303.35 g/mol). She dissolves 1.22 ± 0.01 g of the solid in 15.60 ± 0.01 g benzene. The freezing point is lowered by 1.32 ± 0.04°C.a. What is the molar mass of the substance? Assuming that the percent uncertainty in the calculated molar mass is the same as the percent uncertainty in the temperature change, calculate the uncertainty in the molar mass.b. Could the chemist unequivocally state that the substance is cocaine? For example, is the uncertainty small enough to distinguish cocaine from codeine (C18H21NO3, molar mass = 299.36 g/mol)?c. Assuming that the absolute uncertainties in the measurements of temperature and mass remain unchanged, how could the chemist improve the precision of her results?A core was mounted in a gas permeameter to measure permeability. Determine the liquid permeability (mD) of the core using gas (in oilfield units). The laboratory data are as follows: Diameter of the core = 3.78 cm Length of the core = 7.63 cm Gas viscosity = 0.0148 cP Flow rate = 9.07 cm3/s P1 = 1.439 atm P2 = 1.086 atmWhich of the following is FALSE regarding the continuum assumption of fluids? Select one: a. Uncertainties within the molecular properties of the fluid will be neglected b. Limit concepts can be applied on fluids c. Fluids with same composition may have different properties d. The fluid will be analyzed in the macroscopic level Which of the following does NOT describe a difference between liquids and gases? Select one: a. Liquids form a free surface while gases do not b. Liquids have lower bulk modulus of elasticity than gases c. Specific gravities of liquids are higher than gases d. Viscosity of liquids decreases as temperature increases while on gases, viscosity increases with temperature
- 5.2 Evaluate the Prandtl number from the following data: , .You can use 19.6 x 10^-6 as the experimental result Brass is an alloy that can consist of copper and zinc, where zinc has a coefficient of linear expansion that is between23.0to32.5in units of10−6(∘C)−1. Use the experimental results from the second procedure to determine the range of percent of coppper and the range of percent of zinc in the unknown rod. (Please type answer no write by hend)You are given a sample of polypropylene and wish to find out the volume % crystallinity. You know that the density of 100% crystalline polypropylene (ρc) is 0.946 g/cm3, and the density of 100% amorphous polypropylene (ρa) is 0.855 g/cm3. Through your careful measurements, you determine the density of the sample (ρs) is 0.93 g/cm3. Use the following equation to determine the volume % crystallinity (ν) of your sample. Enter your answer with 1 decimal place. Answer is 82.4 Another sample of polypropylene has a 68 volume percent crystallinity. What is the density of the sample? Enter your answer is g/cm3. ( I need help with this second question. The answer is .917, i just can't get to that.) Hint: use the values and equation from the previous problem.
- You are given a sample of polypropylene and wish to find out the volume % crystallinity. You know that the density of 100% crystalline polypropylene (ρc) is 0.946 g/cm3, and the density of 100% amorphous polypropylene (ρa) is 0.855 g/cm3. Through your careful measurements, you determine the density of the sample (ρs) is 0.93 g/cm3. Use the following equation to determine the volume % crystallinity (ν) of your sample. Enter your answer with 1 decimal place. Another sample of polypropylene has a 68 volume percent crystallinity. What is the density of the sample? Enter your answer is g/cm3. I can answer the first part of the question. I just don't know how to do the second question?a) The number of kilocalories in food is determined by calorimetry techniques in which the food is burned and the amount of heat transfer is measured. How many kilocalories per gram are there in a 5.00-g peanut if the energy from burning it is transferred to 0.500 kg of water held in a 0.100-kg aluminum cup, causing a 54.9C temperature increase? (b) Compare your answer to labeling information found on a package of peanuts and comment on whether the values are consistent.I mix two fluids together, and I use the same mass of each fluid. They are completely miscible; in fact, the two chemicals are isomers. What is the composition of the mixture on a molar basis? Give a numerical answer and a written explanation of how you arrived at it, but do not use any equations.

