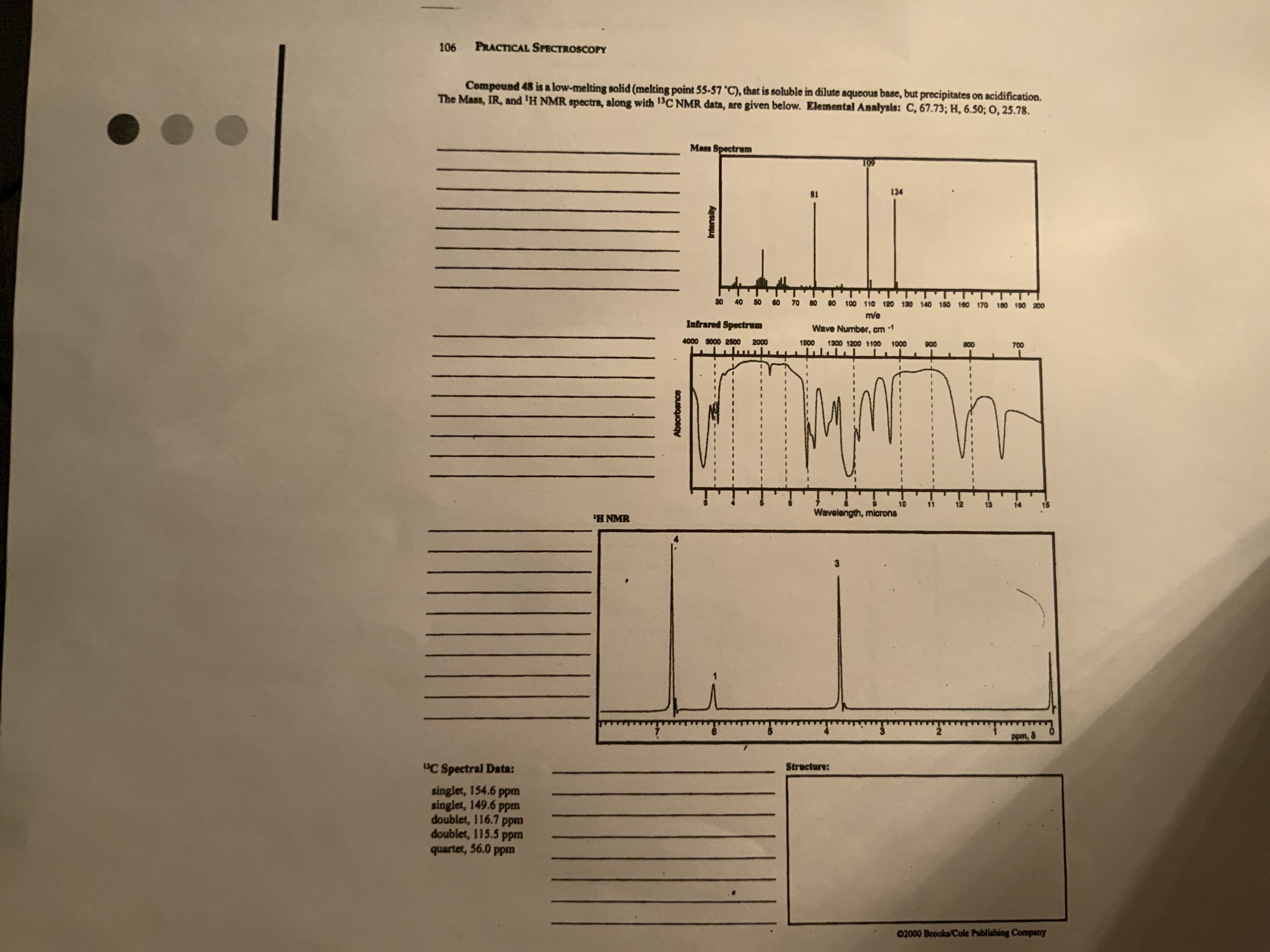
Compound 48 is a low-melting solid (melting point 55-57 °C), that is soluble in dilute aqueous base, but precipitates on acidification. The Mass, IR, and 'H NMR spectra, along with 13C NMR data, are given below. Elemental Analysis: C, 67.73; H, 6.5o; O, 25.78.
Q: Compound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be…
A:
Q: The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of…
A:
Q: The method of assay for sulfonamides is? Kjeldahl method Diazotization method Gravimetric…
A: The solution is given below -
Q: An optical core righyende N, when completely hydolysed yields two moles of myrsic acids (140) an one…
A: Triglycerides are tri esters formed when a glycerol molecule is bonded to three fatty acids with…
Q: An organic compound (A) was heated with a solution of NaOH. The resulting solution was cooled,…
A: To form a precipitate, the chloride ion (Cl-)needs to detach. This Cl-, once free, interacts with…
Q: Explain and describe objective for preparation of methyl orange?
A: Methyl orange is a pH indicator. It has two distinct forms. Red in acidic medium and yellow in basic…
Q: Propose a methodology for the synthesis in the laboratory of AICI3*6H2O from shot or aluminum foil,…
A: Metals are the substances that have a tendency to lose electrons. Hence, metals have a tendency to…
Q: The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3…
A: Given, salt A benzene diazonium chloride is dissolved in 10cm3 of water, cooled and added drop by…
Q: In preparation of p-Nitroaniline, Starting material is 50g of p-nitroacetanilide, 22ml of HCI,…
A: A numerical problem based on quantitative analysis, which is to be accomplished.
Q: What are the volume (uL), moles, and/or weight (mg) of each reagent and solvent in: 50 mg of the…
A:
Q: The UV spectrum of this compound shows no maximum above 250 nm. In the mass spectrum, notice that…
A: Mass spectrum: at M+ 214 the structure is 1,4 dibromobutane. at 55 M-2Br-H having 2 cation at 1,4…
Q: 1g of phenylamine yielded 1.2g of compound A. Calculate the percentage yield of the reaction.
A: When phenyl amine (aniline) reacts with sodium nitrite and hydrochloric acid, then benzene diazonium…
Q: A 0.703 g sample of a commercial detergent was red-calcined to destroy its components. organic.…
A:
Q: The typical oxidation state of aluminum is III, and the oxidation state I is fairly rare. Al `N-N a)…
A:
Q: 1. The following molecule has a molecular formula of C,H10O. The IR and 'H-NMR spectra of the…
A: Given
Q: Propose structure(s) for the compound and give the physical properties, chemical properties, and…
A: The degree of unsaturation for the given compound, having a formula of C9H10O, is 5. Thus, one…
Q: spectru moleculor formula and use the formula to calculate units of ursaturation Assume that the…
A: From this we will do the calculation of the molecular formula of the compound who gives this…
Q: shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of…
A: The aniline at low temperature reacts with the sodium nitrite in acidic medium forms the diazo…
Q: Compound Z with molecular formula C3H9N reacts with C6H5SO2Cl to give a solid, insoluble in alkali.…
A: Given,Compound Z with molecular formula C3H9N reacts with C6H5SO2Cl to give a solid, insoluble in…
Q: The question has been attached as a picture. Please answer this question as every other question I…
A: Please note that ethyloxirane mentioned in the question has a structureSo the name in the brackets…
Q: prepare a flow sheet for the preparation of benzoic acid from a) bromobenzene and magnesium and b)…
A: From grignard reagent this can be done .
Q: -0.500 g sample consisting of NazCO3, NaHCO3, and inert materials was dissolved in sufficient vater…
A: Soln
Q: The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of…
A: Phenyl amine is treated with sodium nitrate in HCl to give compound A(C6H5N2Cl) salt. Then Compound…
Q: Why was it necessary to obtain a constant melting point for B?
A:
Q: prepare glyoxal from acetylene and acetylene from tetrahalide and carbon tetrachloride from methane…
A: The given conversion can proceed as follows:
Q: Explain and describe the conclusion for preparation of p-Iodonitrobenzene.
A: We have to explain and describe the conclusion for preparation of p-Iodonitrobenzene or 4-…
Q: Suggest a retrosynthetic analysis and synthesis for the moloCuloc given in thec e gUection V ou
A: When ethyl benzoate hydrolysis under alkaline condition followed by acidification gives benzoic acid…
Q: In preparation of p-Nitroaniline, Starting material is 50g of p-nitroacetanilide, 22ml of HCI, 20g…
A: Given: mass of p-nitroacetanilide used = 50 g Mass of p-nitroaniline obtained = 20 g
Q: (a) What is the general name given to the reaction between phenylamine and sodium nitrite and…
A: Structure of phenylamine Chemical formula for Sodium nitrite is NaNO2. Structure of…
Q: Please find the open chemical structure of the compound. Please indicate your comments on IR, 1H-NMR…
A: The analysis suggest the presence of hydroxyl and amine group attached on aromatic ring.
Q: 2. Compound P, C4H9Br reacts with aqueous solution of sodium hydroxide to form compound Q, C4H10O.…
A: Organic compounds are of different types. They are classified into saturated, unsaturated and…
Q: Compound K, L and M are three isomers with the molecular formula C5H10O. Compound K cannot be…
A: All the details are explained in handwritten solution.
Q: Indicate the Materials needed for the Synthesis of Terephthalic Acid from Benzene. Chemicals /…
A: Chemicals/Reagents CAS or Catalogue from a supplier Benzene, anhydrous, 99.8% 71-43-2…
Q: A compound E C7H12O gives a crystalline precipitate with 2,4-dinitrophenylhydrazine. It gives a…
A:
Q: CH CI CI 30 -75
A: A ring compound having conjugated system and planner and content (4n+2) number of Pi electron is…
Q: 39), Thionyl chaloride, sOcz orying ageet in meay syotetic chamistry experimepts isi used as a very…
A:
Q: The reaction between Pl3, PSCl3 and Zn powder gives P3ls as one of the products. The sołution state…
A: This Question is based upon P31 NMR.
Q: Discuss the reactivity of organolithiums and why their standardisation is recommended?
A: organolithium compounds are hihgly reactive, they are more reactive compare to organomagnesium…
Q: ethyl mercaptan concentration in a mixture was determined by shaking a 2.0430-g sample with 50.00 mL…
A:
Q: (i) Give the structure of the product formed when an acidified solution of compound A is reacted…
A:
Q: Compound EE, C4H8O shows a positive result (formation of yellow precipitate) when reacted with…
A: This is a question from inorganic reaction.
Q: A ketone reacts with hydrazine to form hydrazone. The reduction of hydrazone under basic conditions…
A:
Q: Why is the reaction mixture extracted with sodium bicarbonate? Give an equation and explain its…
A:
Trending now
This is a popular solution!
Step by step
Solved in 7 steps with 5 images

- The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Questions: (i) Give the name and suggest the colour of the precipitate B. Hence explain why it is necessary to recrystallize B several times. (ii) Why was it necessary to obtain a constant melting point for B?The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. 1g of phenylamine yielded 1.2g of compound A. Calculate the percentage yield of the reaction.The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Why was it necessary to obtain a constant melting point for B?
- The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Give the structure of the product formed when an acidified solution of compound A is reacted with (i)Naphthalen-2-ol (2-naphthol) (ii) Sodium cyanideThe following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Question: Answer both (i) and (ii) below (i) Give the structure of the product formed when an acidified solution of compound A is reacted with Naphthalen-2-ol (2-naphthol) and Sodium cyanide separately (ii) 1g of phenylamine yielded 1.2g of compound A. Calculate the percentage yield of the reaction.The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. Write the equation for the reaction between compound A and phenol Give the name and suggest the colour of the precipitate B. Why is it necessary to recrystallize B several times?
- The following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. (a) What is the general name given to the reaction between phenylamine and sodium nitrite and explain why it's necessary to carry out the reaction at low temperatures. (b) Write the equation for the reaction between compound A and phenolThe following is an experimental account of the preparation of compound B from Phenylamine. 2cm3 of phenylamine are dissolved in 10cm3 of 0.5M hydrochloric acid in a test tube and cooled in an ice bath. 5cm3 of 0.2M sodium nitrate solution cooled to about 5o are added to the solution above and shaken. A salt A, C6H5N2Cl is formed, collected and dried. 1g of salt A is dissolved in 10cm3 of water, cooled and added drop by drop to a cold solution of 0.3g phenol in 5cm3 of 0.1M sodium hydroxide solution. B is precipitated, filtered, dried and the melting point determined. B is further crystallized twice and the melting point taken each time and found to be constant. What is the general name given to the reaction between phenylamine and sodium nitrite and explain why it's necessary to carry out the reaction at low temperatures? Write the equation for the reaction between compound A and phenol Write the equation for the reaction between phenylamine and sodium nitrite.The cyclobutenyl dichloride below reacts with the powerful Lewis acid antimony pentafluoride in liquid SO2 at -75o to give a pale yellow solution that exhibits one singlet at 3.68 ppm in its 1H-NMR spectrum. The species in solution has been deduced to be the salt C8H12X2 where X is an hexahaloantimonate anion. Draw the cation part of the salt C8H12X2
- The reaction of Compound B with O3, followed by Me2S gave two products, C and D (Scheme 1). The mass spectrums of products C and D are shown below. Identify the structures of products C and D. Provide explanation for your answer (based on mass spectrometry interpretation).Compound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentaneCompound K, L and M are three isomers with the molecular formula C5H10O.Compound K cannot be oxidized, while compound M and L can be oxidized.Oxidation of compound L with hot acidified potassium permanganate,KMnO4 solution yields 2-methylbutanoic acid. When treated with Iodoformreagent, yellow precipitation only occurs in compound K. Fehling test onlyyielded positive results for compound L and negative for compound M andK. Compound M is then reacted with hydrogen chloride, HCl produceschlorocyclopentane (i) Draw the structural formula of compounds K, L and M.



