Concentration Initial Final Volume Mass percent of acetic acid of Heinz volume Volume(mL) (mL) of in original vintager solution vintager(M) (mL) vintager Trial 1 0.0963M 0.25 21.6 21.35 Trial 2 0.0963M 0.4 21.8 21.4 Trial 3 0.0963M 0.3 21.4 21.1 Average concertation of HCI (M)
Concentration Initial Final Volume Mass percent of acetic acid of Heinz volume Volume(mL) (mL) of in original vintager solution vintager(M) (mL) vintager Trial 1 0.0963M 0.25 21.6 21.35 Trial 2 0.0963M 0.4 21.8 21.4 Trial 3 0.0963M 0.3 21.4 21.1 Average concertation of HCI (M)
Chapter5: Equilibrium, Activity And Solving Equations
Section: Chapter Questions
Problem 2P
Related questions
Question
NaOH 0.0963M
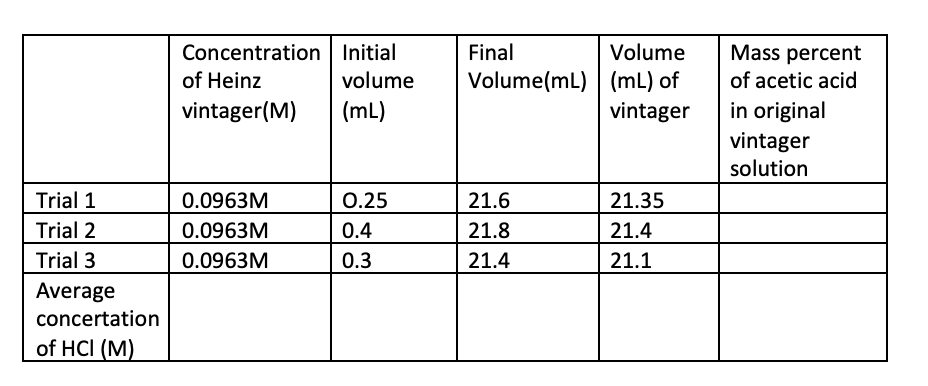
Transcribed Image Text:Concentration | Initial
Final
Volume
Mass percent
of Heinz
volume
Volume(mL) (mL) of
of acetic acid
in original
vintager
solution
vintager(M)
(mL)
vintager
Trial 1
0.0963M
0.25
21.6
21.35
Trial 2
0.0963M
0.4
21.8
21.4
Trial 3
0.0963M
0.3
21.4
21.1
Average
concertation
of HCI (M)

Transcribed Image Text:3. You should include a second table that has information about the concentration of the diluted
vinegar and the concentration (and brand) of the original vinegar solution. Provide one
complete series of sample calculations. (Remember that you diluted the original vinegar by 1:10
before you performed the titration.). The molar mass of acetic acid is 60.05 g/mole and you
should assume that the density of vinegar is 1.00 g/mL. Use this information to calculate the
mass percent of acetic acid in the original vinegar solution.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

