Concentration of Hydrochloric Acid (M) 0.07036 2 2°c Temperature (°C) Titration 3 Titration 1 Titration 2 Volume of Saturated Calcium 10:00ml 38.5 l0:00 ml 10.00'ml 24.7 33.6 8190 Hydroxide (mL) Initial Burette Reading (mL) 17.7 Final Burette Reading (mL) Volume of Hydrochloric Acid (mL) 6.10 7:00 9:0004.91960.00049252 0:0006262. p000229196 lの00049252 10000 6262 0.0429196 0.042952 0:06262 Moles of Hydrochloric Acid (moles ) Moles of Hydroxide (moles) Concentration of Hydroxide (M) Concentration of Calcium Hydroxide b.02146M 743/mol 0:024626M0.03131M 749/mol (М) Formula mass of Ca(OH)2 (g/mol) 749/mal Experimental Solubility of Calcium Hydroxide (g L') 2:3179 1.5889 1.82239 Mean Experimental Solubility of Calcium Hydroxide (g L) Standard Deviation (g L-') RSD (pph) Literature Solubility for calcium Hydroxide' (g L) l ndf 1.5
Concentration of Hydrochloric Acid (M) 0.07036 2 2°c Temperature (°C) Titration 3 Titration 1 Titration 2 Volume of Saturated Calcium 10:00ml 38.5 l0:00 ml 10.00'ml 24.7 33.6 8190 Hydroxide (mL) Initial Burette Reading (mL) 17.7 Final Burette Reading (mL) Volume of Hydrochloric Acid (mL) 6.10 7:00 9:0004.91960.00049252 0:0006262. p000229196 lの00049252 10000 6262 0.0429196 0.042952 0:06262 Moles of Hydrochloric Acid (moles ) Moles of Hydroxide (moles) Concentration of Hydroxide (M) Concentration of Calcium Hydroxide b.02146M 743/mol 0:024626M0.03131M 749/mol (М) Formula mass of Ca(OH)2 (g/mol) 749/mal Experimental Solubility of Calcium Hydroxide (g L') 2:3179 1.5889 1.82239 Mean Experimental Solubility of Calcium Hydroxide (g L) Standard Deviation (g L-') RSD (pph) Literature Solubility for calcium Hydroxide' (g L) l ndf 1.5
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter14: Equilibria In Acid-base Solutions
Section: Chapter Questions
Problem 37QAP: Given three acid-base indicators—methyl orange (end point at pH 4), bromthymol blue (end point at...
Related questions
Question
Calculate :
1)Mean Experimental solubility of calcium hydroxide
2)standard deviation
3)RSD(pph)
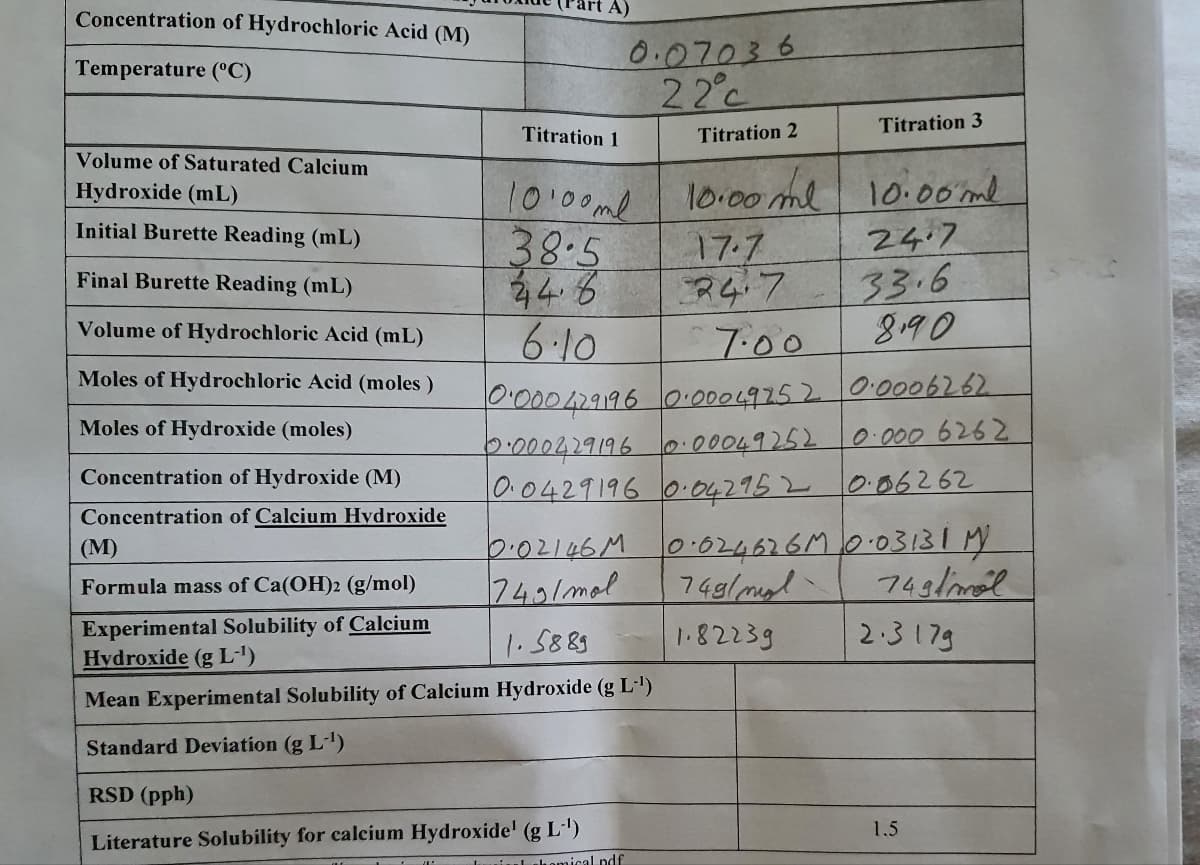
Transcribed Image Text:Concentration of Hydrochloric Acid (M)
0.07036
22°c
Temperature (°C)
Titration 3
Titration 1
Titration 2
Volume of Saturated Calcium
10:00ml
38.5
44.6
6.10
l0:00 ml
17.7
10.00ml
24.7
33.6
890
Hydroxide (mL)
Initial Burette Reading (mL)
Final Burette Reading (mL)
Volume of Hydrochloric Acid (mL)
7:00
9:0004.9196000049752 0:0006262.
2:000429196 0.00049252
0.0429196 0.042952 006262
Moles of Hydrochloric Acid (moles)
Moles of Hydroxide (moles)
0.000 6262
Concentration of Hydroxide (M)
Concentration of Calcium Hvdroxide
0.02146M 0:024626M0.0313IM
74g/msl
(М)
743/mol
749/mal
Formula mass of Ca(OH)2 (g/mol)
Experimental Solubility of Calcium
Hydroxide (g L')
2.317g
1.5889
1.82239
Mean Experimental Solubility of Calcium Hydroxide (g L')
Standard Deviation (g L)
RSD (pph)
1.5
Literature Solubility for calcium Hydroxide' (g L)
uiul homical ndf
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning



Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

