Conditions Column I CHOICES Column II Activity of 1. A sample of Sr-90 has Activity of a half-life of 28 years Greater than Less than 100 g Sr-90 1 g Sr-90 Equal to Cannot be determined 2. The radioactive isotope of 4º19K undergoes n/p ratio of Greater than n/p ratio of daughter nuclide Less than electron capture Equal to Cannot be determined parent nuclide After alpha emission 3. Total number of Greater than Less than After electron neutrons of daughter nuclide of 5525M. emission Equal to Cannot be determined
Conditions Column I CHOICES Column II Activity of 1. A sample of Sr-90 has Activity of a half-life of 28 years Greater than Less than 100 g Sr-90 1 g Sr-90 Equal to Cannot be determined 2. The radioactive isotope of 4º19K undergoes n/p ratio of Greater than n/p ratio of daughter nuclide Less than electron capture Equal to Cannot be determined parent nuclide After alpha emission 3. Total number of Greater than Less than After electron neutrons of daughter nuclide of 5525M. emission Equal to Cannot be determined
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter19: Nuclear Chemistry
Section: Chapter Questions
Problem 62CP
Related questions
Question
100%
Answer the following by comparing Column 1 with Column 2 based from condition. Choose from the choices
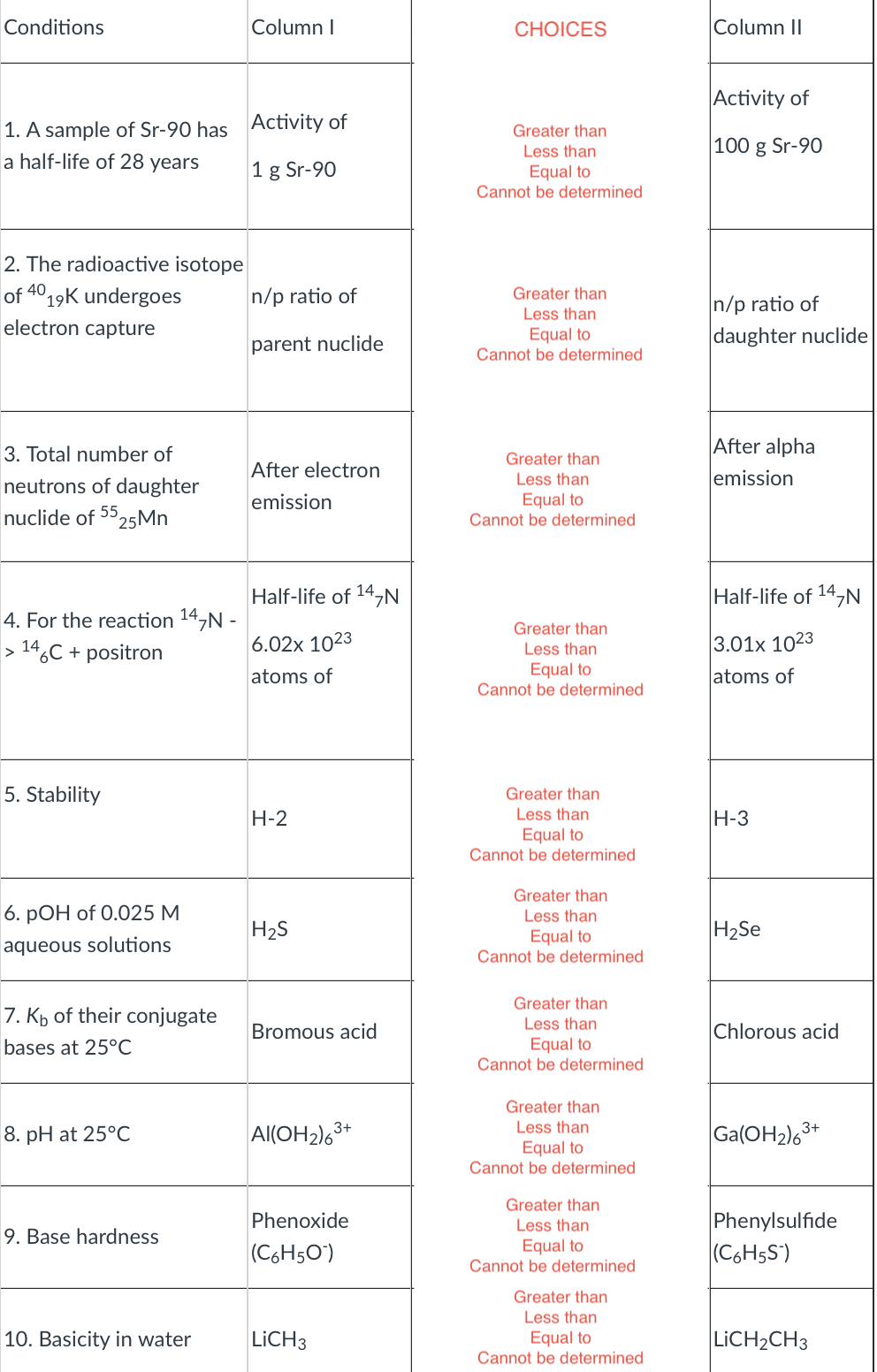
Transcribed Image Text:Conditions
Column I
СHOICES
Column II
Activity of
1. A sample of Sr-90 has Activity of
a half-life of 28 years
Greater than
Less than
100 g Sr-90
1 g Sr-90
Equal to
Cannot be determined
2. The radioactive isotope
of 4019K undergoes
n/p ratio of
Greater than
n/p ratio of
daughter nuclide
Less than
electron capture
parent nuclide
Equal to
Cannot be determined
After alpha
3. Total number of
Greater than
After electron
neutrons of daughter
Less than
emission
Equal to
Cannot be determined
emission
nuclide of 25M.
55
Half-life of 14,N
Half-life of 14,N
4. For the reaction 14,N -
> 14,C + positron
Greater than
6.02x 1023
Less than
3.01x 1023
Equal to
Cannot be determined
atoms of
atoms of
5. Stability
Greater than
H-2
Less than
H-3
Equal to
Cannot be determined
Greater than
6. РОН of 0.025 М
Less than
H2S
H2Se
aqueous solutions
Equal to
Cannot be determined
Greater than
7. Kp of their conjugate
Less than
Bromous acid
Chlorous acid
Equal to
Cannot be determined
bases at 25°C
Greater than
Ga(OH2)63+
8. pH at 25°С
Al(OH2)63+
Less than
Equal to
Cannot be determined
Greater than
Phenylsulfide
(C6H5S')
Phenoxide
Less than
9. Base hardness
(C6H50")
Equal to
Cannot be determined
Greater than
Less than
10. Basicity in water
LICH3
Equal to
Cannot be determined
LICH2CH3
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 7 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning