Consider the acid-catalyzed hydration of 3-methyl-1-butene. For each of the four steps, add one or two curved arrows to the Learning reactant side to show the mechanism. Do not delete any pre-drawn bonds, charges, or lone pairs. Remove the last change by using the undo button on the lower left or revert the drawing palette to the original state by selecting the More menu, then select Reset Drawing. Step 1 H CH, CH2 - H3C-C-C+ H. H. ČH3 H. сн, н
Consider the acid-catalyzed hydration of 3-methyl-1-butene. For each of the four steps, add one or two curved arrows to the Learning reactant side to show the mechanism. Do not delete any pre-drawn bonds, charges, or lone pairs. Remove the last change by using the undo button on the lower left or revert the drawing palette to the original state by selecting the More menu, then select Reset Drawing. Step 1 H CH, CH2 - H3C-C-C+ H. H. ČH3 H. сн, н
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter20: Acidity And Pka Of Phenols
Section: Chapter Questions
Problem 11E
Related questions
Question
100%
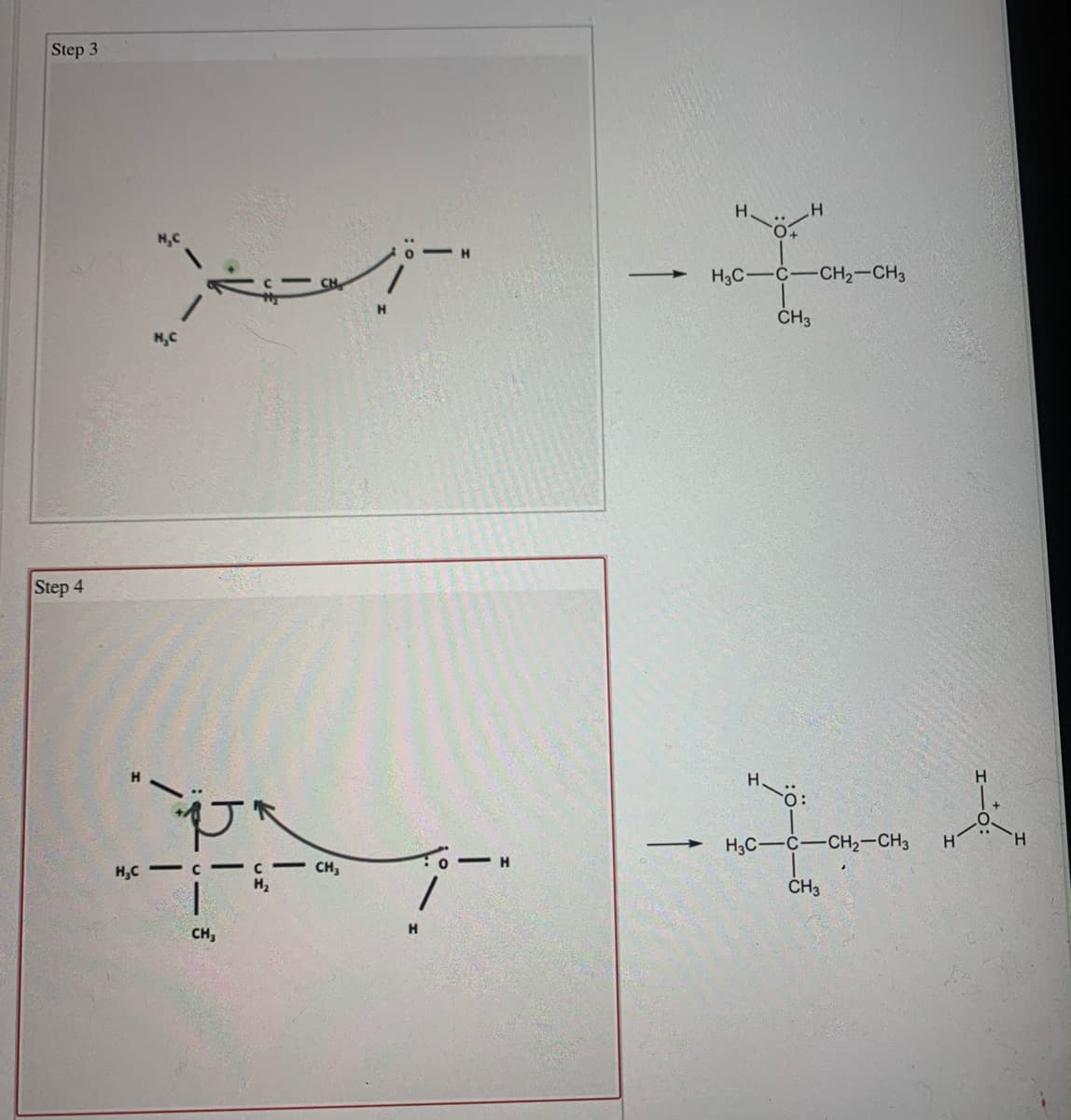
Transcribed Image Text:Step 3
H.
H
H3C-C-CH2-CH3
ČH3
H,C
Step 4
H.
- |-
H.
:-
H.
H3C-C-CH2-CH3
CH,
H,C - C
ČH3
CH,
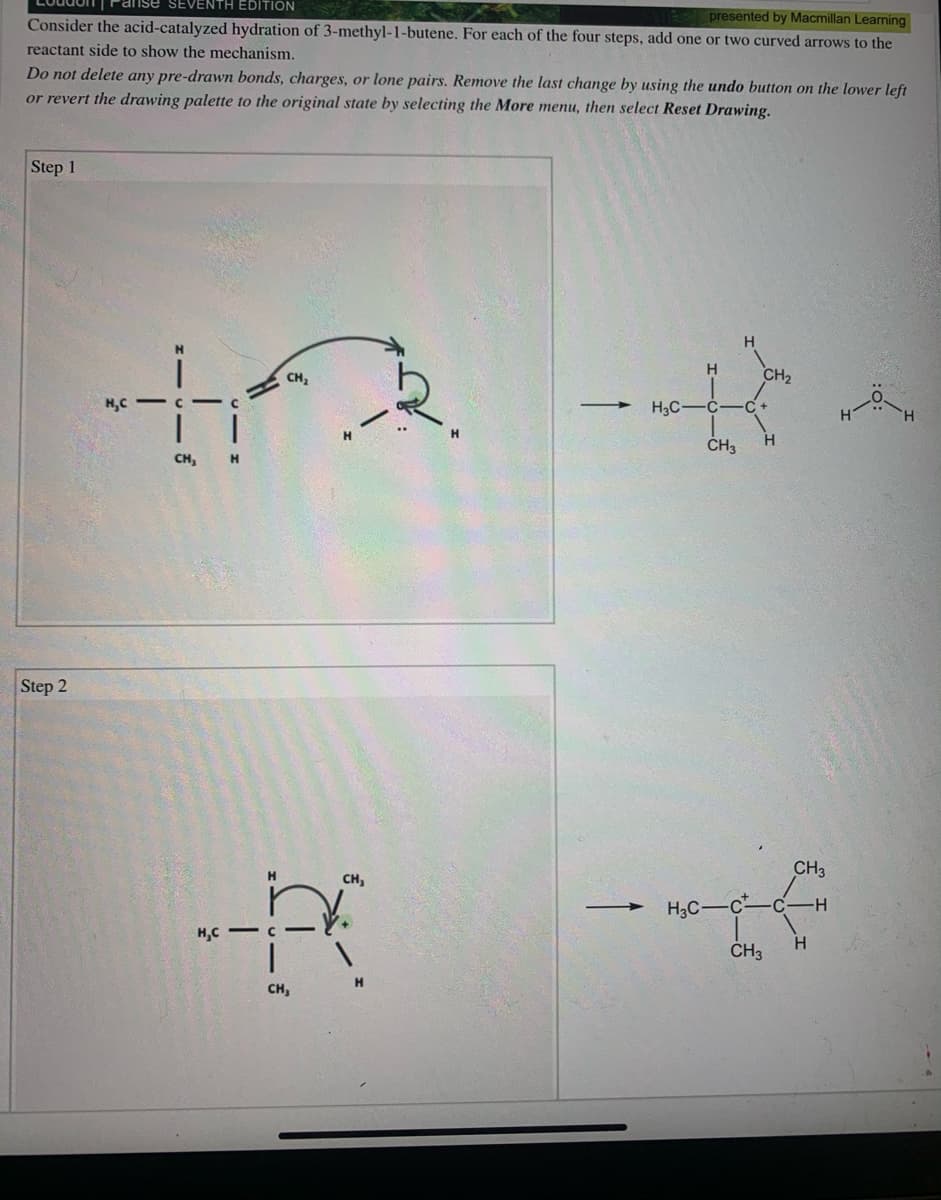
Transcribed Image Text:Loudon| Panse SEVENTH EDITION
presented by Macmillan Learning
Consider the acid-catalyzed hydration of 3-methyl-1-butene. For each of the four steps, add one or two curved arrows to the
reactant side to show the mechanism.
Do not delete any pre-drawn bonds, charges, or lone pairs. Remove the last change by using the undo button on the lower left
or revert the drawing palette to the original state by selecting the More menu, then select Reset Drawing.
Step 1
H.
H
CH2
CH,
H,C
H3C-
-C+
H
CH3
CH,
Step 2
CH3
CH,
H3C-c
-H-
H,C - C
H.
ČH3
H.
CH,
Expert Solution
Step 1
The reaction in which alkene is converted into alcohol by the addition of a water molecule is called a hydration reaction. The reaction is driven by the carbocation formation, hydride shift, and oxonium formation. In this reaction, the more substituted product is the major product. It follows Markovnikoff's rule.
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning