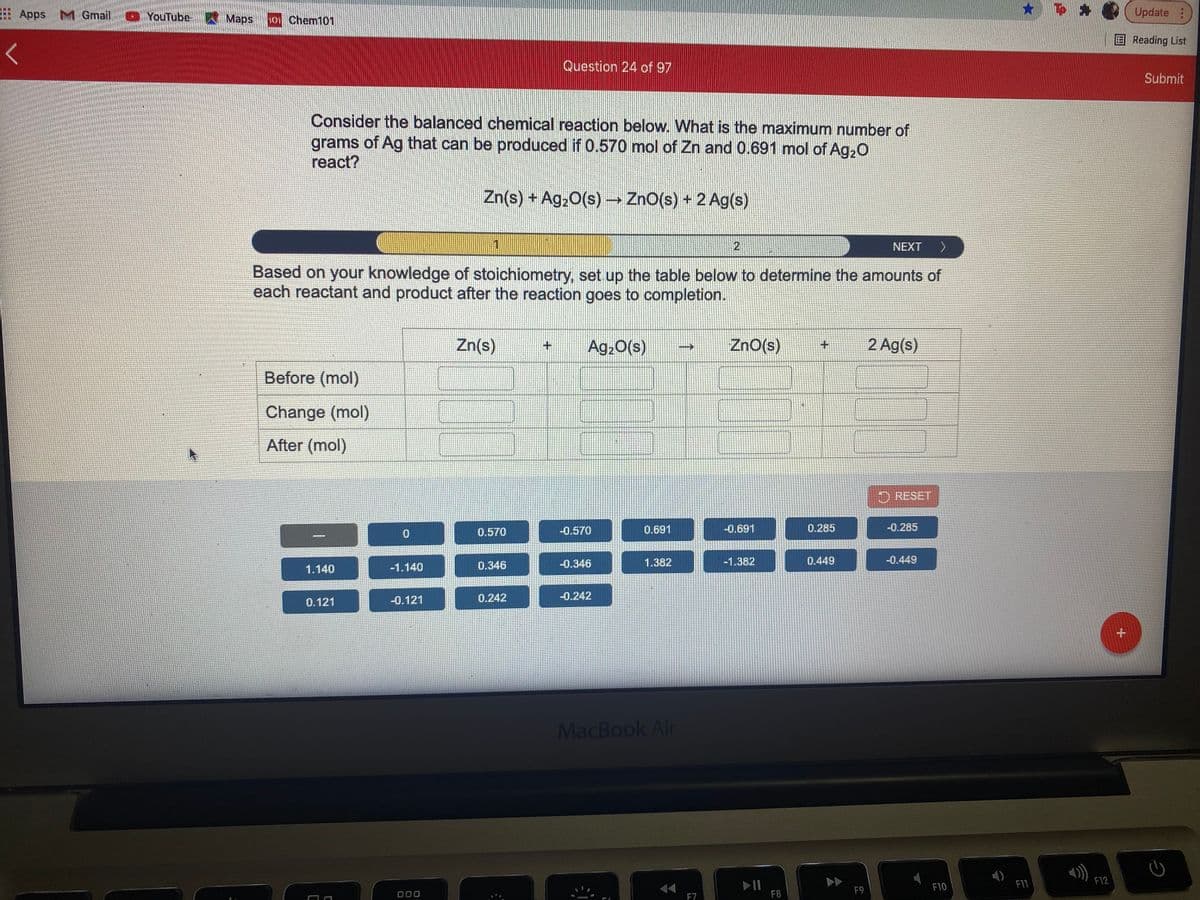
Consider the balanced chemical reaction below. What is the maximum number of grams of Ag that can be produced if 0.570 mol of Zn and 0.691 mol of Ag,0 react? Zn(s) + Ag20(s) ZnO(s) + 2 Ag(s) 2 NEXT > Based on your knowledge of stoichiometry, set up the table below to determine the amounts of each reactant and product after the reaction goes to completion. Zn(s) Ag,0(s) ZnO(s) 2 Ag(s) Before (mol) Change (mol) After (mol) O RESET 0.570 -0.570 0.691 -0.691 0.285 -0.285 -0.346 1.382 -1.382 0.449 -0.449 1.140 -1.140 0.346 0.121 -0.121 0.242 -0.242
Consider the balanced chemical reaction below. What is the maximum number of grams of Ag that can be produced if 0.570 mol of Zn and 0.691 mol of Ag,0 react? Zn(s) + Ag20(s) ZnO(s) + 2 Ag(s) 2 NEXT > Based on your knowledge of stoichiometry, set up the table below to determine the amounts of each reactant and product after the reaction goes to completion. Zn(s) Ag,0(s) ZnO(s) 2 Ag(s) Before (mol) Change (mol) After (mol) O RESET 0.570 -0.570 0.691 -0.691 0.285 -0.285 -0.346 1.382 -1.382 0.449 -0.449 1.140 -1.140 0.346 0.121 -0.121 0.242 -0.242
World of Chemistry, 3rd edition
3rd Edition
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Chapter9: Chemical Quantities
Section: Chapter Questions
Problem 2STP
Related questions
Question
Transcribed Image Text:#Apps
M Gmail YouTube Maps
o1 Chem101
Update :
E Reading List
Question 24 of 97
Submit
Consider the balanced chemical reaction below. What is the maximum number of
grams of Ag that can be produced if 0.570 mol of Zn and 0.691 mol of Ag,0
react?
Zn(s) + Ag,0(s) → ZnO(s) + 2 Ag(s)
1
2
NEXT
へ
Based on your knowledge of stoichiometry, set up the table below to determine the amounts of
each reactant and product after the reaction goes to completion.
Zn(s)
Ag20(s)
ZnO(s)
2 Ag(s)
Before (mol)
Change (mol)
After (mol)
O RESET
0.570
-0.570
0.691
-0.691
0.285
-0.285
0.346
-0.346
1.382
-1.382
0.449
-0.449
1.140
-1.140
0.121
-0.121
0.242
-0.242
MacBook Air
1)
F11
F12
II
F8
F9
F10
O00
F7
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Recommended textbooks for you

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning