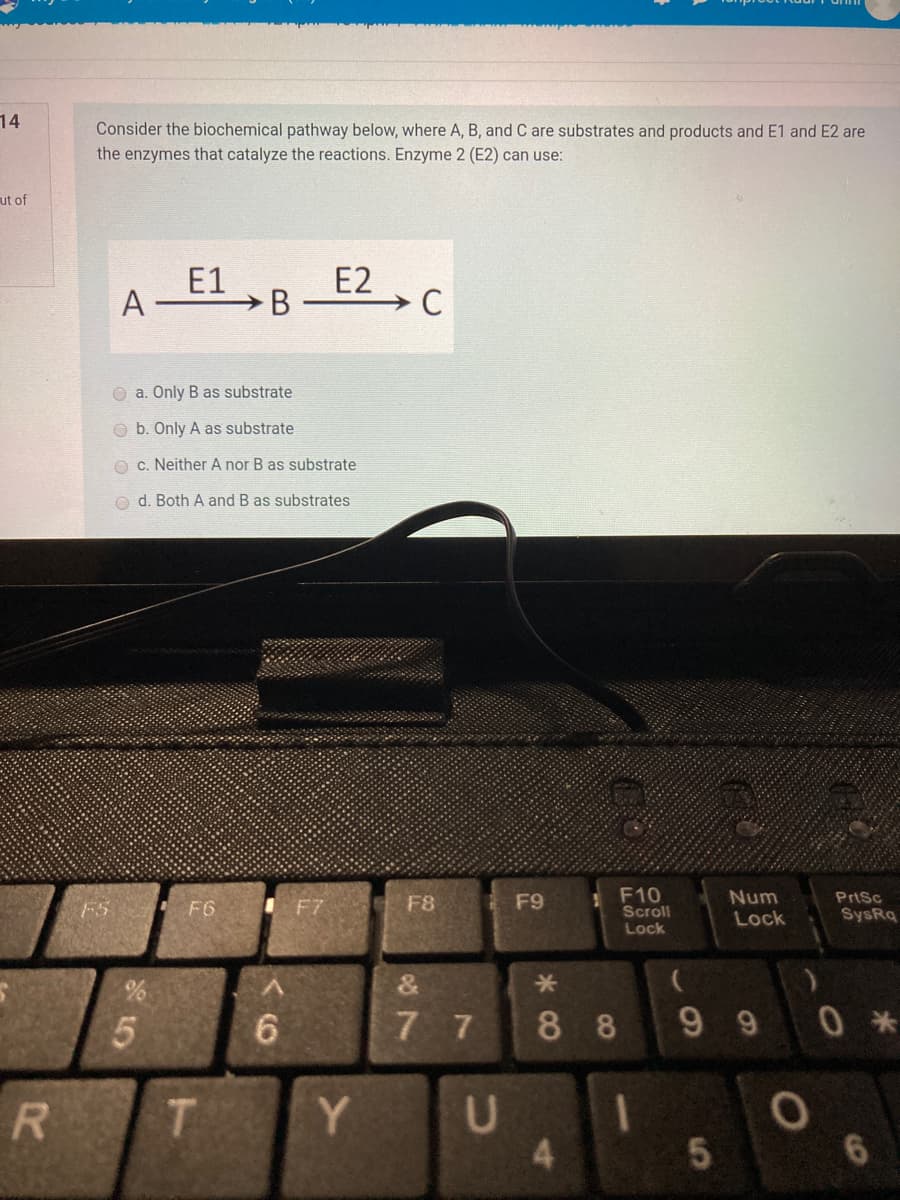
Consider the biochemical pathway below, where A, B, and C are substrates and products and E1 and E2 the enzymes that catalyze the reactions. Enzyme 2 (E2) can use:
Consider the biochemical pathway below, where A, B, and C are substrates and products and E1 and E2 the enzymes that catalyze the reactions. Enzyme 2 (E2) can use:
Biochemistry
9th Edition
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Chapter6: The Behavior Of Proteins: Enzymes
Section: Chapter Questions
Problem 28RE: MATHEMATICAL The enzyme -methylaspartase catalyzes the deamination of -methylaspartate [V. Williams...
Related questions
Question
Transcribed Image Text:14
Consider the biochemical pathway below, where A, B, and C are substrates and products and E1 and E2 are
the enzymes that catalyze the reactions. Enzyme 2 (E2) can use:
ut of
E1
>B
E2
A
O a. Only B as substrate
O b. Only A as substrate
O c. Neither A nor B as substrate
o d. Both A and B as substrates
F10
Scroll
Lock
F5
A F6
F7
F8
F9
Num
PrtSc
Lock
SysRq
&
6.
7 7
8 8
99 0*
R
T.
Y
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305961135
Author:
Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:
Cengage Learning

Biology: The Dynamic Science (MindTap Course List)
Biology
ISBN:
9781305389892
Author:
Peter J. Russell, Paul E. Hertz, Beverly McMillan
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning