Chapter3: Organic Compounds: Alkanes And Their Stereochemistry
Section3.SE: Something Extra
Problem 46AP: Draw the most stable conformation of pentane, using wedges and dashes to represent bonds coming out...
Related questions
Question
please answer them
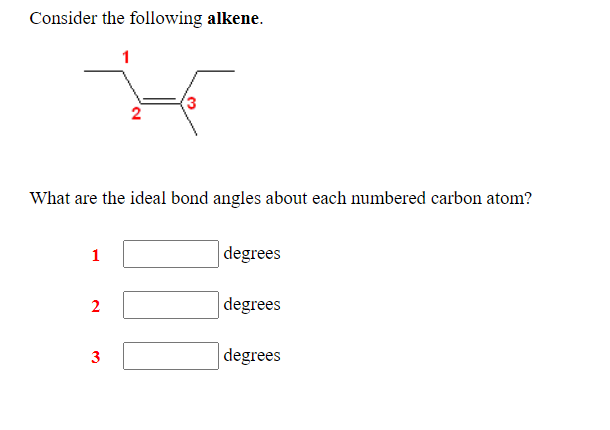
Transcribed Image Text:Consider the following alkene.
1
(3
What are the ideal bond angles about each numbered carbon atom?
1
degrees
|degrees
3
degrees
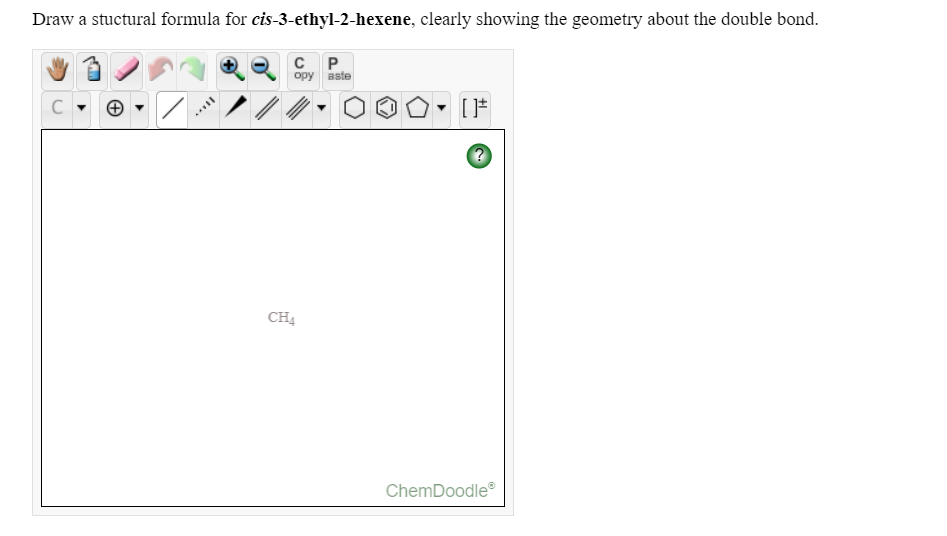
Transcribed Image Text:Draw a stuctural formula for cis-3-ethyl-2-hexene, clearly showing the geometry about the double bond.
C P
opy aste
C
CH4
ChemDoodle
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

