Consider the following equilibrium: 2NO, (g) -N,04(g) AG" = - 5.4 kJ Now suppose a reaction vessel is filled with 7.18 atm of dinitrogen tetroxide (N,0.) at 463. °C. Answer the following questions about this system: rise Under these conditions, will the pressure of N,O, tend to rise or fall? 4 O fall Is it possible to reverse this tendency by adding NO,? In other words, if you said the pressure of N,04 will tend to rise, can that yes be changed to a tendency to fall by adding NO,? Similarly, if you said the O no N204 will tend to fall, can that be changed to a tendency to pressure of rise by adding NO,? If you said the tendency can be reversed in the second question, calculate | atm the minimum pressure of NO, needed to reverse it. Round your answer to 2 significant digits.
Consider the following equilibrium: 2NO, (g) -N,04(g) AG" = - 5.4 kJ Now suppose a reaction vessel is filled with 7.18 atm of dinitrogen tetroxide (N,0.) at 463. °C. Answer the following questions about this system: rise Under these conditions, will the pressure of N,O, tend to rise or fall? 4 O fall Is it possible to reverse this tendency by adding NO,? In other words, if you said the pressure of N,04 will tend to rise, can that yes be changed to a tendency to fall by adding NO,? Similarly, if you said the O no N204 will tend to fall, can that be changed to a tendency to pressure of rise by adding NO,? If you said the tendency can be reversed in the second question, calculate | atm the minimum pressure of NO, needed to reverse it. Round your answer to 2 significant digits.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter17: Spontaneity, Entropy, And Free Energy
Section: Chapter Questions
Problem 121CP: If wet silver carbonate is dried in a stream of hot air. the air must have a certain concentration...
Related questions
Question
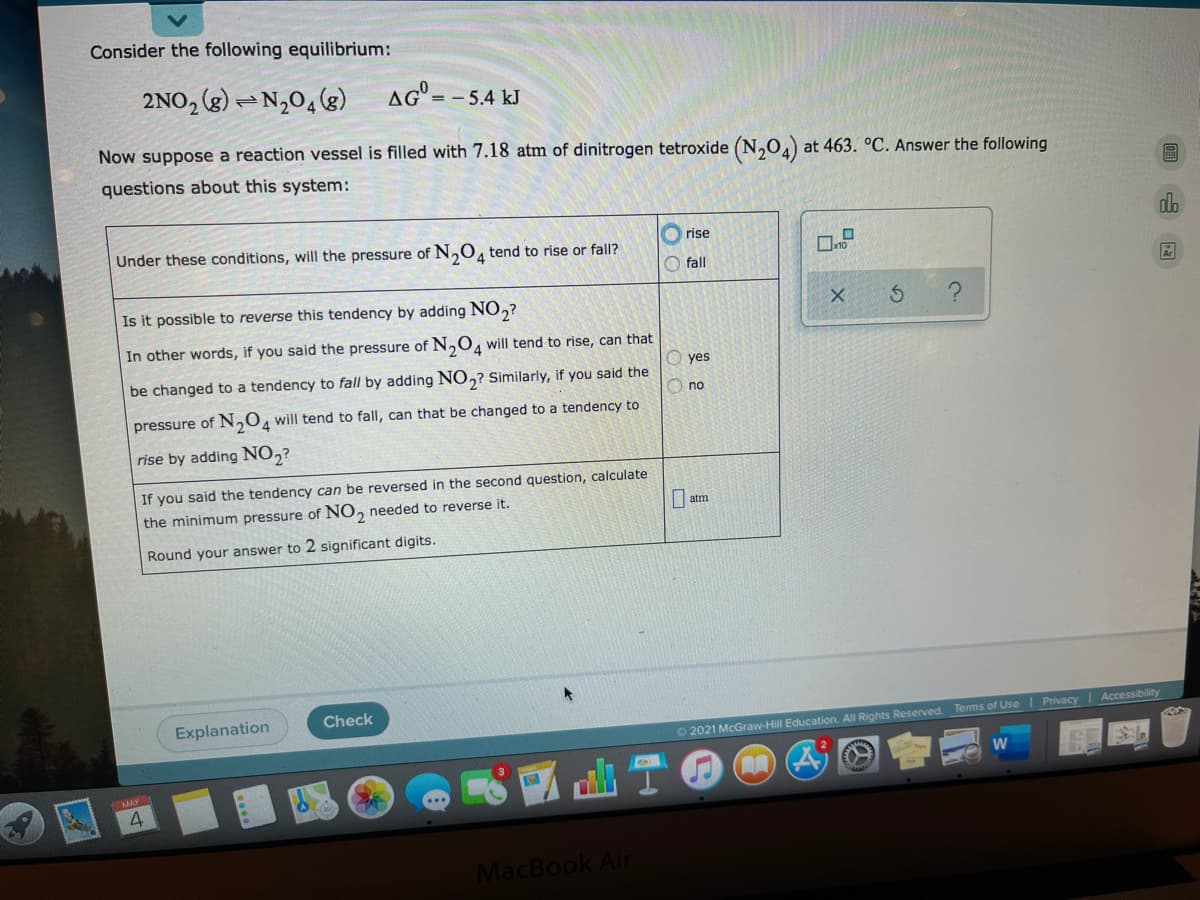
Transcribed Image Text:Consider the following equilibrium:
2NO, (g) - N,04 (g)
=- 5.4 kJ
Now suppose a reaction vessel is filled with 7.18 atm of dinitrogen tetroxide (N,0,) at 463. °C. Answer the following
questions about this system:
Under these conditions, will the pressure of N,0, tend to rise or fall?
rise
O fall
Ar
Is it possible to reverse this tendency by adding NO,?
In other words, if you said the pressure of N,O, will tend to rise, can that
yes
be changed to a tendency to fall by adding NO,? Similarly, if you said the
no
pressure of N,0, will tend to fall, can that be changed to a tendency to
rise by adding NO,?
If you said the tendency can be reversed in the second question, calculate
the minimum pressure of NO, needed to reverse it.
O atm
Round your answer to 2 significant digits.
Explanation
Check
Terms of Use
Privacy Accessibility
O 2021 McGraw-Hill Education. All Rights Reserved
MAY
4.
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax