Consider the following reaction at 293 K Feaqy+ SCNaq)→ FeSCN (aq) Initially, 0.924 moles of FeNO3 and 0.924 moles of KSCN are added to a 1 L solution. Equilibrium is established and the concentration of FESCN²" is found to be 0.074M. What is the concentration of Fe* and SCN¯ ions in solution at equilibrium? Report your answer to only three decimal places. Hint: the moles of both species should be equal.
Consider the following reaction at 293 K Feaqy+ SCNaq)→ FeSCN (aq) Initially, 0.924 moles of FeNO3 and 0.924 moles of KSCN are added to a 1 L solution. Equilibrium is established and the concentration of FESCN²" is found to be 0.074M. What is the concentration of Fe* and SCN¯ ions in solution at equilibrium? Report your answer to only three decimal places. Hint: the moles of both species should be equal.
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter12: Gaseous Chemical Equilibrium
Section: Chapter Questions
Problem 47QAP: For the following reactions, predict whether the pressure of the reactants or products increases or...
Related questions
Question
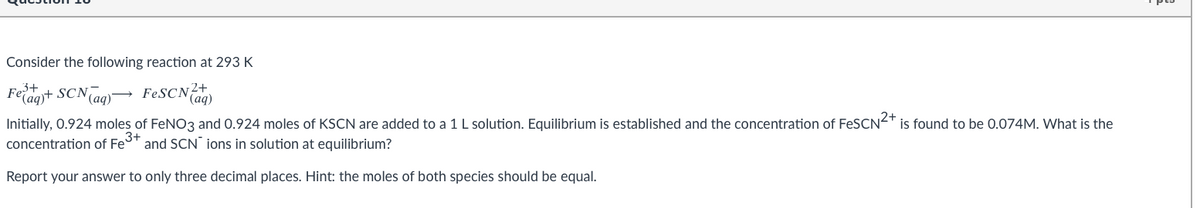
Transcribed Image Text:Consider the following reaction at 293 K
Feragyt SCNTao)→ FESCN?+
(aq)
(aq)
Initially, 0.924 moles of FeN03 and 0.924 moles of KSCN are added to a 1 L solution. Equilibrium is established and the concentration of FeSCN<T is found to be 0.074M. What is the
concentration of Fe" and SCN ions in solution at equilibrium?
Report your answer to only three decimal places. Hint: the moles of both species should be equal.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning