Consider the following reaction: Mno, lag) + 5FE2" (ag) + 8H*(aa) Mn2"(ag) + 5Fe Cac) + 4H2Om Initially the reaction is at standard conditions, and 298K. If the volume of the reservoir is 2.0L, what mass of NaOH (neglecting volume changes) is required to give a reduction potential of 0.55V? The reduction potential of MnO4 /Mn²* is 1.51V and that of Fe/Fe* is 0.77V. O 79 g O 40 g O 58 g O 24 g O 93 g
Consider the following reaction: Mno, lag) + 5FE2" (ag) + 8H*(aa) Mn2"(ag) + 5Fe Cac) + 4H2Om Initially the reaction is at standard conditions, and 298K. If the volume of the reservoir is 2.0L, what mass of NaOH (neglecting volume changes) is required to give a reduction potential of 0.55V? The reduction potential of MnO4 /Mn²* is 1.51V and that of Fe/Fe* is 0.77V. O 79 g O 40 g O 58 g O 24 g O 93 g
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 43P
Related questions
Question
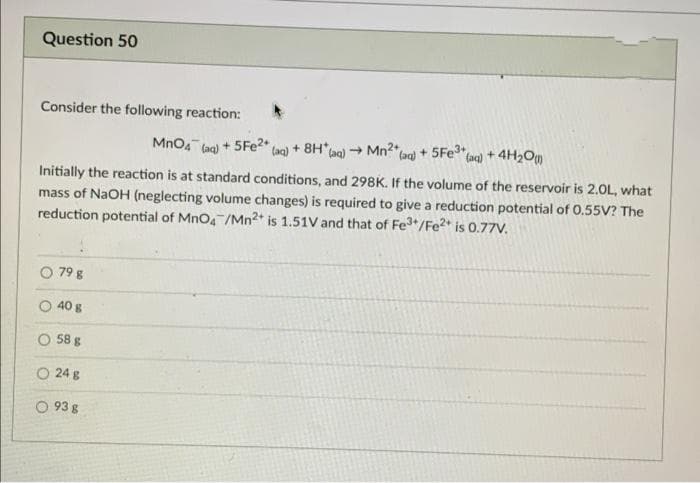
Transcribed Image Text:Question 50
Consider the following reaction:
Mno4 (ag) + 5FE2" (ag) + 8H*Lag) - Mn2"lag) + 5Fe
(ac) + 4H2Om
Initially the reaction is at standard conditions, and 298K. If the volume of the reservoir is 2.0L, what
mass of NaOH (neglecting volume changes) is required to give a reduction potential of 0.55V? The
reduction potential of MnO4 /Mn2* is 1.51V and that of Fe*/Fe?* is 0.77V.
79 g
40 g
O 58 g
O 24 g
O 93 g
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning