Consider the following reaction where K = 9.52x102 at 350 K. + A reaction mixture was found to contain 3.07x102 moles of CHal9), 3.96x102 moles of CCla(g) and 1.21x102 moles of CH,C{9), in a 1.00 liter container. Is the reaction at equilibrium? If not, what direction must it run in order to reach equilibrium? The reaction quotient, Qo equals The reaction A. must run in the forward direction to reach equilibrium. B. must run in the reverse direction to reach equilibrium. C. is at equilibrium
Consider the following reaction where K = 9.52x102 at 350 K. + A reaction mixture was found to contain 3.07x102 moles of CHal9), 3.96x102 moles of CCla(g) and 1.21x102 moles of CH,C{9), in a 1.00 liter container. Is the reaction at equilibrium? If not, what direction must it run in order to reach equilibrium? The reaction quotient, Qo equals The reaction A. must run in the forward direction to reach equilibrium. B. must run in the reverse direction to reach equilibrium. C. is at equilibrium
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter11: Chemical Kinetics
Section: Chapter Questions
Problem 11.42PAE: Amoxicillin is an antibiotic packaged as a powder. When it is used to treat babies and small...
Related questions
Question
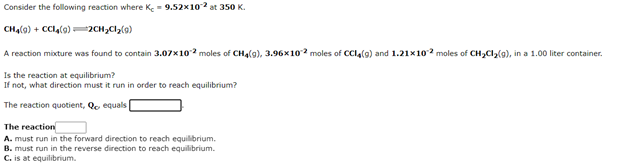
Transcribed Image Text:Consider the following reaction where Ke = 9.52x10 at 350 K.
CHa(9) + CCla(0) =2CH,Cl2(9)
A reaction mixture was found to contain 3.07x102 moles of CHa(g), 3.96x10 moles of CCIA(g) and 1.21x102 moles of CH,Cl2(g), in a 1.00 liter container.
Is the reaction at equilibrium?
If not, what direction must it run in order to reach equilibrium?
The reaction quotient, Qo equals
The reaction
A. must run in the forward direction to reach equilibrium.
B. must run in the reverse direction to reach equilibrium.
C. is at equilibrium.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning