Consider the following voltaic cell (Eigure 1) Figure Fels) Voltmeter Sale bridge IMF IM Ag 1 of 1 Agis) Y What is the change in the cell voltage when the ion concentrations in the cathode half-cell are increased by a factor of 10? Express your answer with the appropriate units. E-E- Value Submit Part C HÅ E-E- * Incorrect; Try Again; 6 attempts remaining Submit Previous Answers Request Answer What is the change in the cell voltage when the ion concentrations in the anode half-cell are increased by a factor of 107 Express your answer with the appropriate units. μÅ + Value V Request Answer 4 TELEVÍ V
Consider the following voltaic cell (Eigure 1) Figure Fels) Voltmeter Sale bridge IMF IM Ag 1 of 1 Agis) Y What is the change in the cell voltage when the ion concentrations in the cathode half-cell are increased by a factor of 10? Express your answer with the appropriate units. E-E- Value Submit Part C HÅ E-E- * Incorrect; Try Again; 6 attempts remaining Submit Previous Answers Request Answer What is the change in the cell voltage when the ion concentrations in the anode half-cell are increased by a factor of 107 Express your answer with the appropriate units. μÅ + Value V Request Answer 4 TELEVÍ V
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 105QAP: Consider a voltaic cell in which the following reaction occurs. Zn(s)+Sn2+(aq)Zn2+(aq)+Sn(s) (a)...
Related questions
Question
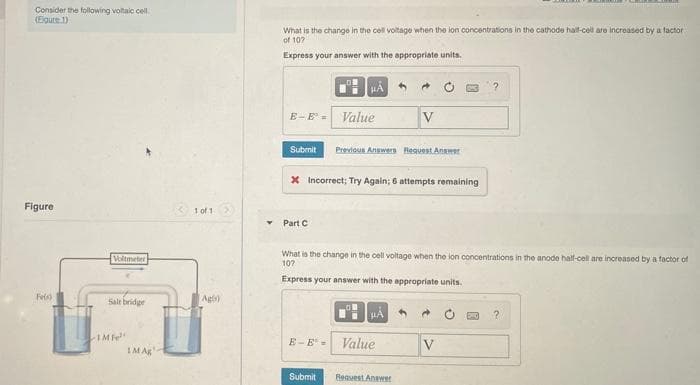
Transcribed Image Text:Consider the following voltaic cell
(Eigure 1)
Figure
Fels)
Voltmeter
Salt bridge
IMF
IM Ag
1 of 1
Agis)
Y
What is the change in the cell voltage when the ion concentrations in the cathode half-cell are increased by a factor
of 10?
Express your answer with the appropriate units.
E-E Value
Submit
Part C
HÅ
E-E-
* Incorrect; Try Again; 6 attempts remaining
Submit
Previous Answers Request Answer
What is the change in the cell voltage when the lon concentrations in the anode half-cell are increased by a factor of
107
Express your answer with the appropriate units.
A
μÅ
V
Value
Request Answer
V
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning