Q: The mass ratio of sodium to fluorine in sodium fluoride is 1.21:1. A sample of sodium fluoride…
A: Number of moles is calculated by dividing given mass with molefluorinecular mass. In the given…
Q: alculate ASn for the balanced chemical equation: ASixn ΑΣΦ -6.56-104 2H₂S(g) + 302(g) → 2H₂O(g)…
A:
Q: Mary is trying to determine if some household substances are acids or bases. She collects the…
A:
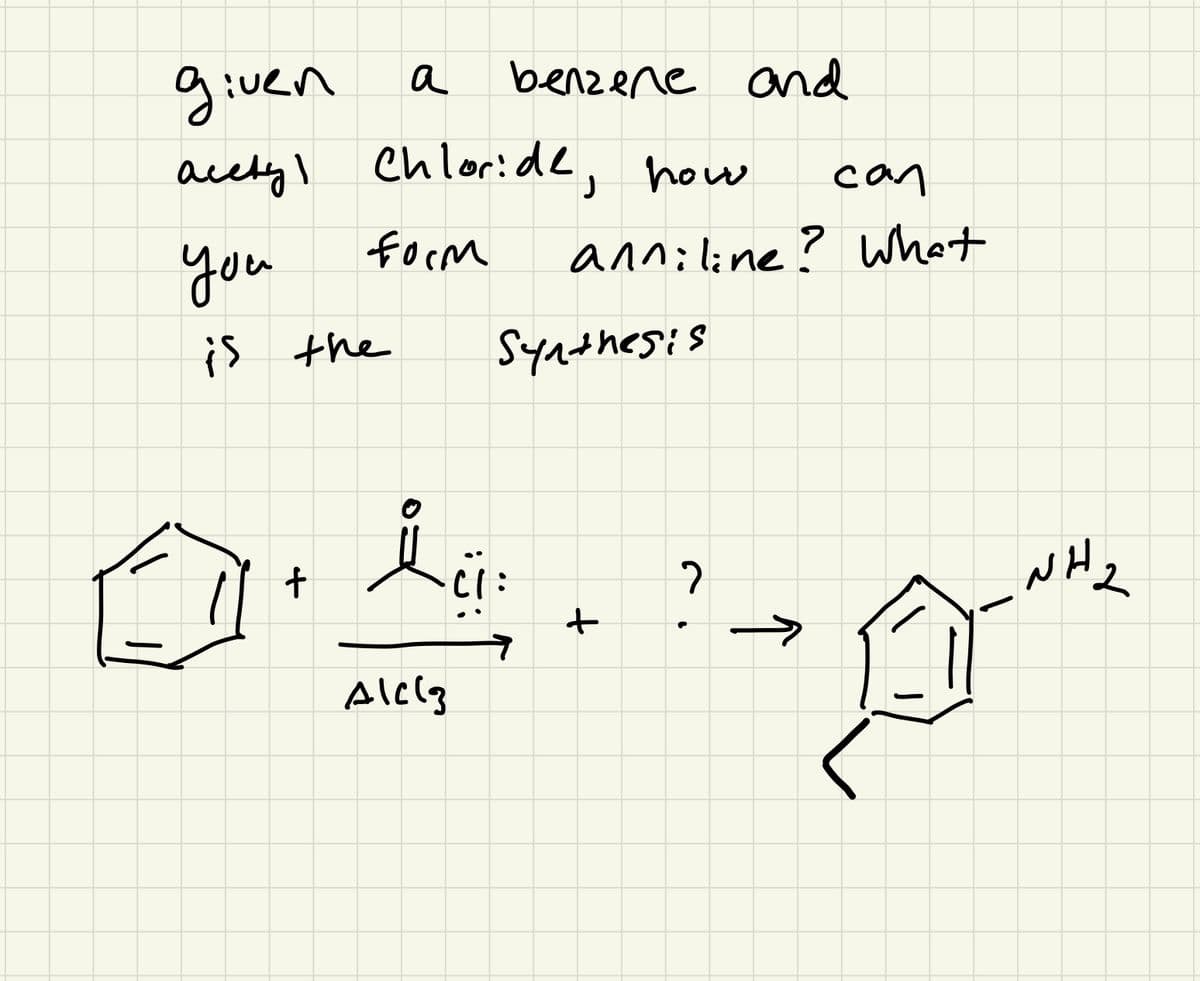
Q: 5. Provide a step-by-step reaction mechanism for the following transformation. You must show all…
A: Ketals are prepared by reacting a ketone with alcohol in the presence of an acid catalyst. In this…
Q: Draw the product of this series of reactions. 1. Br₂ 2. 2 equivalents of NaNH₂, 3. Na/NH3 (1) 4.…
A: Bromination is reaction in which Bromine is being added to a Reactant to give compound with Bromine…
Q: Draw the organic and inorganic products for the following acid/base reaction. H "-" H + 2 products…
A: Bronsted-Lowry acid-base theory: Acids are substances that gives H+ ions and base are substances…
Q: For the following reaction. ONa a. List all expected organic and covalent products. b. Indicate what…
A:
Q: Identify the spectator ions in the following complete ionic equation. Ba²+ (aq) +2 I (aq) + 2 Na+…
A: To solve this problem we have to identify the spectators ions from the given complete ionic equation…
Q: Decide whether each chemical reaction in the table below is an oxidation-reduction ("redox")…
A: A reaction in which oxidation & reduction occurs simultaneously are called Redox reaction.…
Q: When 18.0 mL of a 4.00 x 10-4 M potassium hydroxide solution is combined with 25.0 mL of a 9.71 x…
A: Solubility product quotient, Qsp is the product of the concentrations of the products, each raised…
Q: Consider the decomposition of lead(IV) oxide: PbO₂ (s) PbO(s) + O₂(g) At a certain temperature the…
A: Pressure exerted by individual gases in mixture of gases is known as partial pressure of that gases.
Q: Rank the organic molecules in order of increasing acidity.
A: According to Arrhenius concept, an acid is a substance i.e. when dissolved in water gives H+ ions .…
Q: Calculate the volume of 2.31 M NaI that would be needed to precipitate all of the Hg+2 ion from 175…
A:
Q: Light fuel oil with an average chemical composition of C10H18 is burned with oxygen in a bomb…
A: This problem is based on Thermodynamics. The solution is given below.
Q: The initial concentration of NOCl in the second-order reaction 2NOCl→2NO+Cl2 is 0.878M. After…
A: The rate of the reaction is directly proportional to the concentration of the reactants. So a…
Q: redict the oxidative product(s) of 2-methyl-6-octyn-2-ene after undergoing ozonolysis with O3, H₂O2.
A: Ozonolysis of the alkynes or alkenes produces the carbonyl compounds. It produces the ozonide…
Q: A mixture of gases has 0.3000 mol of CO2, 0.3190 mol of SO2 and 0.3500 mol of water vapor. The total…
A: Given that, a gas mixture having 0.3000 mol of CO2, 0.3190 mol of SO2, 0.3500 mol of water vapor.…
Q: Can you please explain how to go about assigning chirality priority from 1-4?
A: Since, According to Cahn-Ingold-Prelog rule- 1) More atomic number having more priority.2) If first…
Q: Write the reaction in water for H2CO3? Be careful to use the proper arrow. If none, write N.R.
A: An acid is a substance which when dissolve in water give H+ ions. For weak acid there exist an…
Q: Insert the correct number of atoms from each element on the reactant side of the equation
A:
Q: What is the correct value of the equilibrium constant Kc for the reaction: 2 NH3(g) N₂(g) + 3 H₂(g)…
A: Since,Equilibrium constant or Kc is the ratio of the equilibrium concentrations of product over…
Q: A chemical reaction involves the reactants A and B, and the product C. A+B → C Provide your answer…
A:
Q: Predict the products that are formed when diphenyl C O 1) Excess MeMgBr 2) H3O+
A: Diphenyl carbonate react with excee grignard reagent to give tertiary alcohol
Q: Calculate the change in pH when 7.00 mL of 0.100 M HCl(aq) is added to 100.0 mL of a buffer solution…
A: To solve this problem, we need to first determine the initial pH of the buffer solution. The buffer…
Q: A certain solvent has a normal freezing point of 2.65 ° C, and a freezing point depression…
A:
Q: hich of the following compounds has an activating group? A. Bromobenzene B. Benzoic acid OC. None in…
A:
Q: Does a reaction occur when aqueous solutions of nickel (II) nitrate and cobalt(II) sulfate are…
A: net ionic equation is a chemical equation where those ions undergoing chemical changes during the…
Q: If we start the reaction below with 16.78 g of Ag2S (molar mass of 247.80 g/mol) and 6.780 g HCI…
A:
Q: solution initial components (check all that apply) initial type acidic basic neutral acidic basic…
A: Given, Based on pH scale, The solutions which have pH greater than 7 ( pH>7 ) are basic…
Q: In the net ionic equation for the following, what are the spectator ions? NH4NO3(aq) + KCl(aq) →…
A: Steps to write net ionic equation is 1) First write the balanced equation 2) Write total ionic…
Q: What is the volume of 27 mol of CH4 gas at STP?
A: The question is based on the concept of ideal gas equation. we need to calculate volume of given…
Q: Given the following information: 1.6 g of an unknown monoprotic acid (HA) required 50.80 mL of a…
A:
Q: If a buffer solution is 0.180 M in a weak base (K₁ = 4.9 x 10-5) and 0.510 M in its conjugate acid,…
A:
Q: What is the conjugate base of [Fe (H2O)6]^3+ (aq
A: According to Bronsted and Lowry concept an Acid is a substance that has the tendency to donate a…
Q: A 25.00 mL sample of NaOH is titrated with 21.35 mL of 0.6736 M H2SO4. What is the Molarity of the…
A:
Q: Draw the neutral organic product formed by the reaction of hydroxylamine hydrochloride with acetone.
A: Reaction between acetone and hydroxylamine hydrochloride produces the condensation product oxime…
Q: Complete the curved arrow pushing mechanism of the reaction of butanal in ethylene glycol and…
A: Carbonyl compounds reacts with ethylene glycol to give a cyclic ether as the product.
Q: The equilibrium constant, Ka, for a monopratic acid (propionic acid) is 1.3 ×10^-5. Which of the…
A:
Q: In the IR spectrum of the product, which signals are due to the carbonyl and the alkene?
A: To find out the signal for the carbonyl and the alkene in the given IR spectrum of the product.
Q: What is the pH of a 0.750 M solution of NaCN (Ka of HCN is 4.9 × 10-10)?
A: pH is the negative logarithm of hydrogen ion concentration. It can be given as pH=-log [H+] NaCN…
Q: Consider the decomposition reaction: XY2(s) X(g) + 2 Y(g) where X and Y are some chemical groups. At…
A: In the equilibrium constant expression, the concentration of pure solids and liquids are considered…
Q: Two students are given the starting material benzoic acid and are asked to prepare benzaldehyde. The…
A: LiAlH4 is stronger and non-selective reducing agent which reduces almost all the functional group…
Q: Medical grade Heliox, used in cases of respiratory distress, may contain 20.0 mole % oxygen (0₂) and…
A:
Q: HEMWORK When a calcium chloride solution is mixed with a lead(II) nitrate solution, a precipitate…
A: Steps to write a net ionic equation and complete ionic equation for a molecular reaction :…
Q: Classify the unbalanced reactions below. You will have to select the best answer if more than one…
A: Precipitation reaction: The reaction in which an insoluble solid is obtained as one of the product.…
Q: Suppose 2.89 g of zinc iodide is dissolved in 350. mL of a 46.0 m M aqueous solution of potassium…
A:
Q: 2 O 11 C-H benzaldehyde + CH₂CCH3 acetone NaOH (E.E)-1,5-diphenyl-1,4-pentadiene-3-one…
A:
Q: How many atoms are present in (2x10^0) moles of an element?
A:
Q: What is the most likely product resulting from the reaction shown? Н 0= OH НО OH NaBH4 ? од
A: NaBH4 is a good reducing agent. It can reduce aldehydes , ketones , acid chlorides and anhydrides .…
Q: Which compound below is predicted to be the most acidic? OH OH да вовн OH 0 OH
A: carboxylic acid stronger than phenol Due to the resonance in carboxylic acids, the negative charge…
Step by step
Solved in 8 steps with 7 images

- Chlorhexidine gluconate is available in different products in concentrations of 4% w/vand 0.12% w/v. H ow many milliliters of the more dilute product may be prepared fromeach fluidounce of the more concentrated product?Draw the organic product(s) formed when CH3CH2CH2OH is treated withfollowing reagent : [1] TsCl, pyridine; [2] NaSHWhen some sugars dissolve in water they spontaneously undergo changes in optical rotation called mutarrotation. The Mutarrotation of D-glucopyranose is catalyzed by acid and bases. 2-Hydroxypyridine is a more effective catalyst than phenol and pyridine for this reaction because: a.Both oxygen and N in 2-hydroxypyridine act as bases increasing the rapid interconversion of sugar b. The OH of 2-hydroxypyridine serves as the base while the current N as the acid. c. 2-hydroxypyridine acts both as a base to remove the proton from the hydroxyl group in the hemiacetal and as an acid to provide a proton to the oxygen in the hemiacetal. d.Phenol and pyridine are very expensive.
- d-Glucuronic acid is found widely in plants and animals. One of its functions is to detoxify poisonous HO-containing compoundsby reacting with them in the liver to form glucuronides. Glucuronides are water soluble and therefore readily excreted. Afteringestion of a poison such as turpentine or phenol, the glucuronides of these compounds are found in the urine. Draw thestructure of the a- and b-glucuronides formed by the reaction of b-d-glucuronic acid and phenol.Which of these reagent(s) will not react with HOCH2CH2CH2COOH? A) NaCN in ethanol B) C2H5OH in the presence of an acid catalyst. C) (CH3CO)2O D) Concentrated H2SO4What product is formed when each compound is treated first with LDA in THF solution at low temperature, followed by CH3CH2I?
- What prochirality is correct?please explain why this happends, im having trouble in this section. Part A Chymotrypsin, an enzyme that hydrolyzes peptide bonds in proteins, functions in the small intestine at an optimum pH of 7.7 to 8.0. How is the rate of the chymotrypsin-catalyzed reaction affected by each of the following conditions?Draw the products formed when CH3COCH2CH2CH = CH2 is treated with each reagent:(a) LiAlH4, then H2O;(b) NaBH4 in CH3OH;(c) H2 (1 equiv), Pd-C;(d) H2 (excess), Pd-C;(e) NaBH4 (excess) in CH3OH;(f) NaBD4 in CH3OH.

