Consider the reaction: N2O4 (g) ⟷ 2 NO2 (g) KP = 0.36 (at 100°C) The reaction above is carried out at the same temperature but the vessel contains only the reactant, PN2O4 = 0.0550 atm and none of the product. Complete an ICE diagram to find the equilibrium partial pressures for N2O4 and NO2.
Consider the reaction: N2O4 (g) ⟷ 2 NO2 (g) KP = 0.36 (at 100°C) The reaction above is carried out at the same temperature but the vessel contains only the reactant, PN2O4 = 0.0550 atm and none of the product. Complete an ICE diagram to find the equilibrium partial pressures for N2O4 and NO2.
Chemistry & Chemical Reactivity
9th Edition
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter18: Principles Of Chemical Reactivity: Entropy And Free Energy
Section: Chapter Questions
Problem 72SCQ: Consider the formation of NO(g) from its elements. N2(g) + O2(g) 2 NO(g) (a) Calculate Kp at 25 C....
Related questions
Question
100%
Consider the reaction:
N2O4 (g) ⟷ 2 NO2 (g) KP = 0.36 (at 100°C)
The reaction above is carried out at the same temperature but the vessel contains only the reactant, PN2O4
= 0.0550
atm and none of the product. Complete an ICE diagram to find the equilibrium partial pressures for N2O4 and NO2.
Expert Solution
Step 1
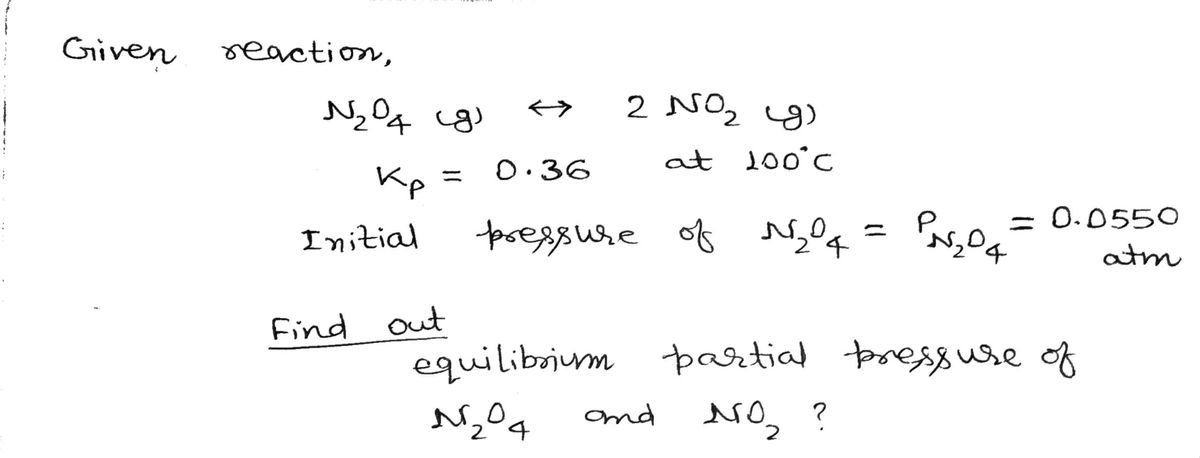
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
I got 0.03844 when I used the quadratic by hand...did I make a mistake somewhere?
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning