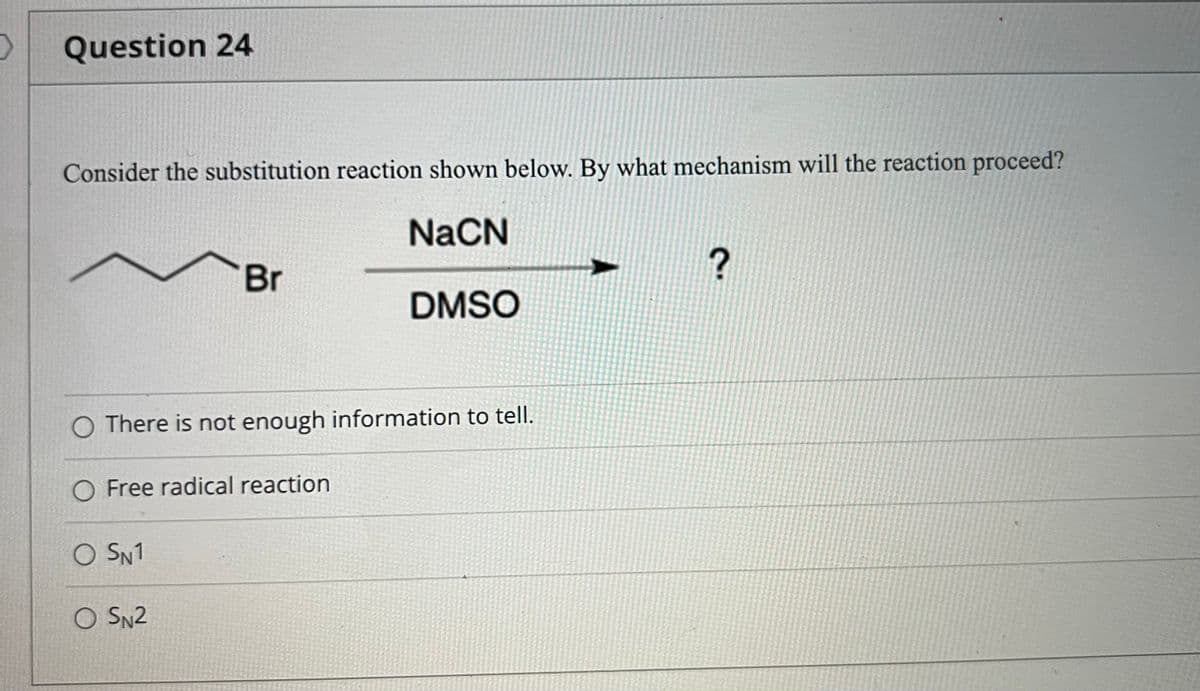
Consider the substitution reaction shown below. By what mechanism will the reaction proceed? NaCN Br DMSO There is not enough information to tell. Free radical reaction SN1 O SN2
Q: A diprotic acid, H₂A, has Ka1 = 3.4 × 10⁻⁴ and Ka2 = 6.7 × 10⁻⁹. What is the pH of a 0.23 M solution...
A: The PH of diprotic acid can be calculated as follows
Q: I. Observing pH Changes in Water and Buffer Solutions upon Addition of HCI and NaOH Solutions soluti...
A:
Q: Calculate the pH of a 0.87 M methylamine solution.
A: pH of solution is defined as negative logarithm of hydrogen ions.
Q: 2. A 1000 cc flask is filled with 4.00 grams of hydrogen gas and 8.27 ounces of bromine gas at 448°C...
A: A 1000 cc flask is filled with 4.00 g of hydrogen gas and 8.27 ounces of bromine gas at 448◦ C. The ...
Q: Br
A: -->Functional group is ketone.
Q: Compound A is an alcohol that undergoes oxidation to produce compound B. Compound B is a ketone that...
A:
Q: Assign the correct stereochemical description to the chiral center in this molecule: CH,CH3 "Н H;C B...
A: Every chiral carbon has specific configuration.these configuration are either R or S.
Q: For the reactions below write out the overall reaction and the equilibrium constant expressions for ...
A: Chemical equilibrium is defined as the stage at which both reactant and product are in equilibrium.
Q: Data is collected for the gas phase reaction 2 A + B + 3 C → Products at 470 K. If the reaction is z...
A: Reaction: 2 A + B + 3 C → Products reaction is zero order in A reaction is second order in B reac...
Q: Calculate the pH of a 0.31 M sodium formate solution (HCOONa). Kb for HCOO− = 5.9 × 10−11
A: Given :- [HCOONa] = 0.31 M Kb for HCOO- = 5.9 × 10−11 To calculate :- pH of solution
Q: Fill up the empty boxes with the correct chemical structures.
A: Interpretation- We have to fill the empty boxes with the correct chemical structures - Introduc...
Q: How much heat is removed from the condensation of water at 25 °C? Given: S° H2O (1) = 69.9 J/mol·K a...
A: In condensation gas become liquid ∆S = Sproduct - Sreactant ∆S = 69.9 J/ mol K - 188.7 J/ mol K ∆S...
Q: An electron in a hydrogen atom is excited from the n = 1 ground state to the n = 4 excited state. Cl...
A:
Q: Be sure to answer all parts. The reaction 2A B is second order with a rate constant of 51.0/M-min at...
A: Using summary of second order kinetic in chemical kinetics.
Q: Part B If the volume of the original sample in Part A(P = 412 torr , V1 = 10.0 L ) changes to 64.0 L...
A: Given, Initially Pressure (P1) = 412 torr Volume (V1) = 10.0 L Later Pressure (P2) = ? Volume (V2) ...
Q: What volume of a 0.470M HCl reacted with enough Zn metal to produce 1.50 g ZnC12 based on the follow...
A: Given mass of ZnCl2 = 1.50 gram Molar mass of ZnCl2 = 136.39 g/mol
Q: Galvanic Ecell vut s Anode Reduction potrntial Voits Cathode (u/s) -Znrs): Fecss- Zncoi
A: 1) Cu-Zn Cell Reaction : Zn(s)+Cu2+(1.0 M)→Zn2+(1.0 M)+Cu(s) ...
Q: What is the enantiomeric excess (ee) of a mixture of enantiomers that is 45% the (-)-isomer and 55% ...
A: Given- + isomer is 55% - isomer is 45%
Q: Calculate for the theoretical and percent yield. Please show detailed calculations
A: To Calculate for the theoretical and percent yield.,
Q: What is the pH of the solution obtained by mixing 65.00 mL of 0.220 MM HCl and 60.00 mL of 0.125 ...
A:
Q: A solution of HCN was prepared in 2.5 L of water. The pH of this solution at 25°C was determined to...
A: We have to calculate the moles of HCN.
Q: for hypochlorous acid, HClO, is 3.0×10-8. Calculate the pH after 40.0 mL of 0.100 M NaOH have been a...
A: Ka(HClO) = 3.0 × 10^-8 Volume of NaOH = 40.0 ml Molarity of NaOH = 0.100 M Volume of HClO = 40.0ml M...
Q: excess reactant
A:
Q: . For the following consecutive first order reactions K =2.0s- K =2.0s →Y the time (in seconds) requ...
A: Here it is given a first order consecutive reaction.we will find the time required for Y to reach i...
Q: The equilibrium constant K, for the reaction HCN(aq) =H*(aq) + CN (aq) is 4.9 x 10-10 at 25 °C. Calc...
A: The equilibrium concentration can be calculated as follows
Q: Isobutyric acid has a 5 in H2O solvent 1/3 of its solubility is diethyl ether. Imagine 4.0 g of 1BA ...
A:
Q: Calculate the mass of 0.11 moles of Cu(NO3)2. grams Give your answer to 2 decimal spaces.
A: Given that , No. Of moles of Cu(NO3)2 = 0.11 moles
Q: For a precipitation reaction to be useful in a gravimetric analysis, the product of the reaction mus...
A: Gravimetric analysis is a method of quantitative chemical analysis in which the constituent is conv...
Q: Classify the relationship between the two structures shown below: and Br Br Br Br Identical Enantiom...
A: Isomer are compounds which have same empirical formula If configuration are same at every carbon cen...
Q: The internuclear distance between two closest Kr atoms in solid krypton is about 4.0 Å. The polariza...
A: We have find out boiling point of Krypton.
Q: Calculate the most probable radius, rmp, at which an electron will be found when it occupies a 1s or...
A:
Q: When 6 g of granulated Zn is added to a solution of 2 M HCl in a beaker at room temperature, hydroge...
A: When 6 g of granulated Zn is added to a solution of 2 M HCl in a beaker at room temperature, hydroge...
Q: Evaluate the mean radius of a 3s orbital by integration.
A: To evaluate: mean radius of a 3s orbital by integration
Q: Give clear handwritten final major product only
A:
Q: Reaction Rates acetone 12 HCI Run 1 3.74x10 L mol1 0.320 M 0.0000960 M 0.0800 M sec 1 Run 2 7.33x10'...
A: The formula used is :- r1r2= k[A]1a[B]1bK[A]2a[B]2b
Q: Complete the following reaction by drawing the structure of the major product(s) expected.
A: The given reaction is of oxidation of alkene to alcohol in presence of water and H2SO4.
Q: Answer the following questions: 1. Why do liquids assume different shapes on the surface of the wax...
A: As per the guideline, since you have asked multiple questions, we have solved the first question for...
Q: Which of the following glycosidic linkage is found in maltose? * A. Glucose (a1-2B) Fructose B. Gl...
A: Glycosidic linkage is the linkage between two monosaccharide units through oxygen atoms with the los...
Q: IH NMR spectrum of methyl-m-nitrobenzoate 'H nmr, 8: 8.86 (t, IH), 842(dd, IH), 8.37 (dd, IH), 7.67 ...
A: There are two types of equivalency-Chemical and magnetic. Chemically non-equivalent hydrogens show...
Q: Solve it asap
A: Suppose a solution contains 0.28 M Pb2+ and 0.50 M Al3+ . Calculate the pH range that would allow Al...
Q: 2,3-Dimethylfumaric acid has a molecular formula C6H8O4. It undergoes oxidative cleavage to form two...
A: GIVEN:- 2,3-Dimethylfumaric acid has a molecular formula C6H8O4. It undergoes oxidative cleavage to ...
Q: 5. Solve the Q given the following : 200-g sample of an unknown substance changed, temp from 40°C to...
A: Given : Mass = m = 200 gram Final temperature = T1 = 25°c Initial temperature = T2 = 40°c ...
Q: Using the table of standard formation enthalpies that you'll find under the ALEKS Data tab, calculat...
A: Solution -
Q: 1. Reaction Rates Cupric Oxide is leached in an acidic solution which results in the formation of Cu...
A: Leaching is a process of extracting a metal from its ore by dissolving it in a certain liquid. The c...
Q: e two structures sho
A:
Q: What is the molarity of a solution that contains 0.180 moles KOH in 0.350 L of solution?
A: Given : 1 . Moles of KOH (Solute) = 0.180 moles 2. Volume of solution = 0.350 L To calculate : Mol...
Q: Under certain conditions the rate of this reaction is zero order in hydrogen iodide with a rate cons...
A: Integrated rate law for a zero order reaction is, [A]t = -kt + [A]o [A]o = Initial concentrati...
Q: A diprotic acid has a pKa1 = 3.10 and pka2 = 6.50. What is the pH of a 0.10 M solution of this acid ...
A:
Q: QUESTION 5 An unhealthy by-product of combustion is carbon monoxide which occurs in incomplete combu...
A: Please note- As per our company guidelines we are supposed to answer only one question. Kindly repos...
Q: The solubility of Pb(OH)2 is 78 mg/500. mL. Calculate the Ksp for lead (II) hydroxide
A: We have to calculate the Ksp for given salt.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Pls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2OThe Products are from substitution and elimination reaction mechanism. explain the reaction mechanism. I got the product in substitution as CH2OCH3, i dont understand the shift in double bonds in other product. Also could you specify which ones are products from substitution and elimination...The Products are from substitution and elimination reaction mechanism.explain the reaction mechanism. I got the product in substitution as CH2OCH3, i dont understand the shift in double bonds in other product. Also could you specify which ones are products from substitution and elimination...
- A mixture propane and bromine is exposed to sunlight. The reaction equation is given below. ??3??2??3 + ??2 + ?? → ? + ? a. State the type of the reaction above. b. What is the function of sunlight in the reaction? c. Draw the structure of the monosubstituted products, A and B. identify the major products. d. Write the mechanism for the formation of the major product. e. What would happen if the mixture is kept in the dark at room temperature?Please make reactants and etc clear thank youWrite five different EAS reactions at the top of the page.
- Hello, can somebody please help me with this organic chemistry question? question: Draw the mechanism of a hydro-halogenation reaction using cyclohexene. Show all the steps .Explain your reason for the answer1.All the following parameters would affect the rate and kinetics of the reaction exceptA.Solvent mediumB.Reaction temperatureC.Reaction timeD.Geometry and structure of the alkyl halideE.pH1 example for organic elimination reaction show: a. the overall reaction (reactants --> products) b. the reaction mechanism (indicate intermediate product) c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?) d. overall description of the reaction eg., radical substitution or SR
- What's the Reaction mechanism for formation of L1 example for organic rearrangement reaction show: a. the overall reaction (reactants --> products) b. the reaction mechanism (indicate intermediate product) c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?) d. overall description of the reaction eg., radical substitution or SR1 example for organic substitution reaction show: a. the overall reaction (reactants --> products) b. the reaction mechanism (indicate intermediate product) c. indicate which is the reactive species or intermediate in the reaction (radical? electrophile? nucleophile?) d. overall description of the reaction eg., radical substitution or SR

