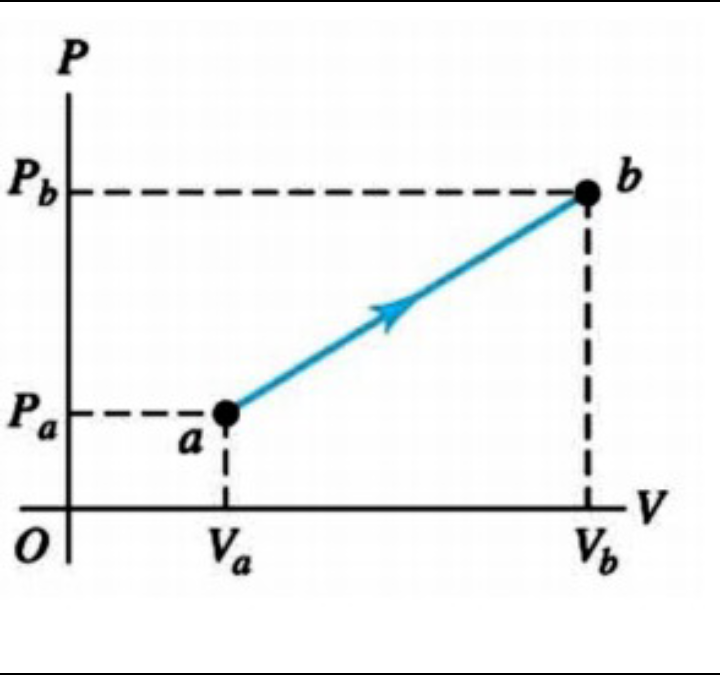
Considering air as an ideal gas, a process can be engineered by which a certain quantity of air change from thermodynamic state a to final state b, as shown in the pressure versus volume diagram. According to the information provided by the diagram and knowing that Va = 0.070 m3,
Considering air as an ideal gas, a process can be engineered by which a certain quantity of air change from thermodynamic state a to final state b, as shown in the pressure versus volume diagram. According to the information provided by the diagram and knowing that Va = 0.070 m3,
Refrigeration and Air Conditioning Technology (MindTap Course List)
8th Edition
ISBN:9781305578296
Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Chapter46: Room Air Conditioners
Section: Chapter Questions
Problem 16RQ
Related questions
Question
Considering air as an ideal gas,
a process can be engineered by which a certain quantity
of air change from
Va = 0.070 m3,
Vb = 0.110 m3
P_a = 1.0 × 10^5 Pa and
P_b = 1,4 × 10^5 Pa. Answer:
(a) The air temperature according to the process will decrease, increase or
will it stay the same?
(b) How much work will be done by air during the process?
Transcribed Image Text:P
P
P.
-A-
Vp
Va
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Refrigeration and Air Conditioning Technology (Mi…
Mechanical Engineering
ISBN:
9781305578296
Author:
John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:
Cengage Learning

Refrigeration and Air Conditioning Technology (Mi…
Mechanical Engineering
ISBN:
9781305578296
Author:
John Tomczyk, Eugene Silberstein, Bill Whitman, Bill Johnson
Publisher:
Cengage Learning