Constants Penodic Wile E. Coyote is up to no good again. he's been cooking up a secret plan to get the Roadrunner in his underground lab. His brilliant plan this time was to use a compressed, toxic gas. He compressed the gas from an initial volume of 5.75 L to a final volume of 1.20 L by an external pressure of 1.42 bar During the compression, the gas released 128 J of heat. Part A What is the total work (in Joules) associated with the compression of the gas? Sign is important! Express your answer with three significant figures. ? w = J Submit Request Answer
Constants Penodic Wile E. Coyote is up to no good again. he's been cooking up a secret plan to get the Roadrunner in his underground lab. His brilliant plan this time was to use a compressed, toxic gas. He compressed the gas from an initial volume of 5.75 L to a final volume of 1.20 L by an external pressure of 1.42 bar During the compression, the gas released 128 J of heat. Part A What is the total work (in Joules) associated with the compression of the gas? Sign is important! Express your answer with three significant figures. ? w = J Submit Request Answer
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter4: Introduction To Gases
Section: Chapter Questions
Problem 67E: The compression ratio in an automobile engine is the ratio of the gas pressure at the end of the...
Related questions
Question
2
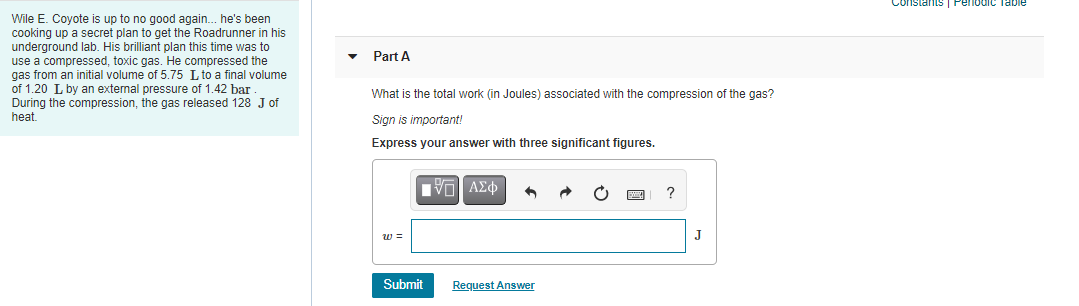
Transcribed Image Text:Constants
Pelodic Table
Wile E. Coyote is up to no good again. he's been
cooking up a secret plan to get the Roadrunner in his
underground lab. His brilliant plan this time was to
use a compressed, toxic gas. He compressed the
gas from an initial volume of 5.75 L to a final volume
of 1.20 L by an external pressure of 1.42 bar
During the compression, the gas released 128 J of
heat.
Part A
What is the total work (in Joules) associated with the compression of the gas?
Sign is important!
Express your answer with three significant figures.
?
w =
J
Submit
Request Answer
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning