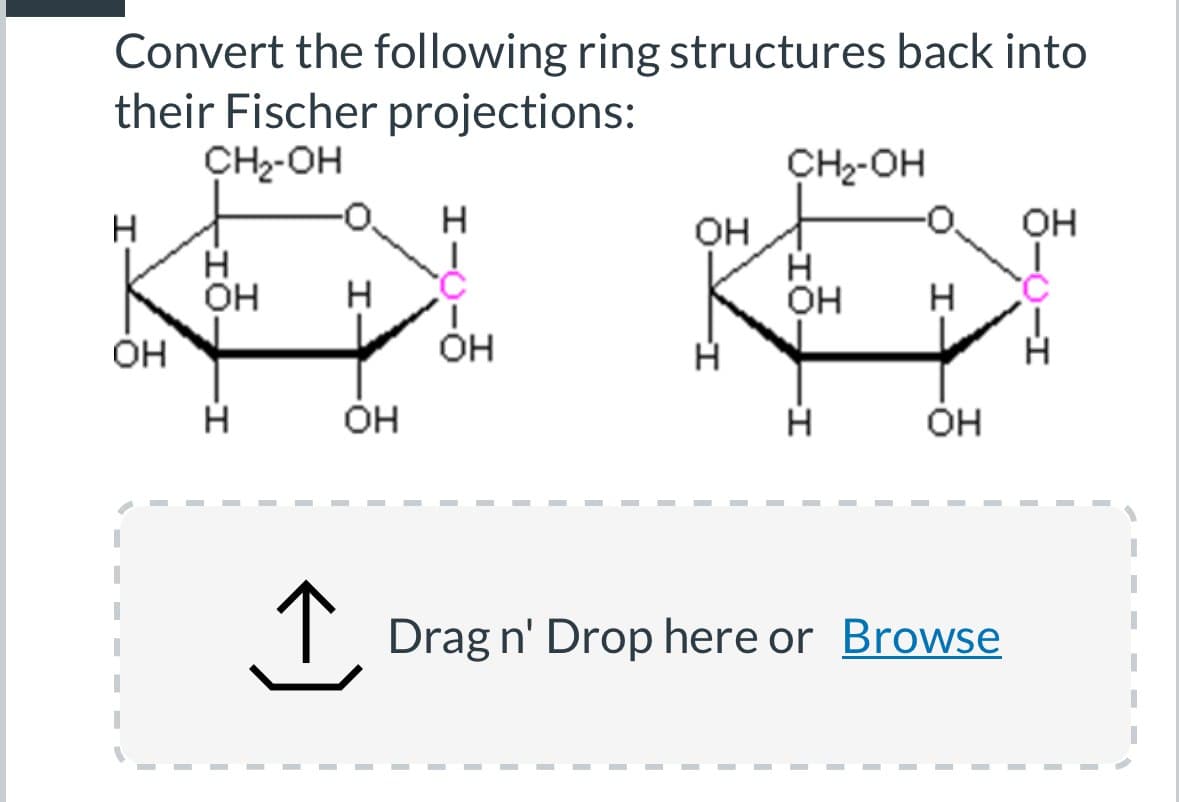
Convert the following ring structures back into their Fischer projections: CH₂-OH CH₂-OH H H OH OH OH OH -1. H H H OH H Он OH I- Н エー -I Н OH Н OH ↑ Drag n' Drop here or Browse I
Q: The table below shows the equilibrium % of product C in a mixture of A, B and C at a temperature of…
A: Le Chatelier's principle suggests that when a system at equilibrium is disturbed by changing factors…
Q: An isotropic amorphous vitreous substance (meaning that it has the same refractive index regardless…
A: The term "isotropic amorphous vitreous substance" applies to materials exhibiting uniform properties…
Q: Draw a structural formula for the product of the reaction shown. CH3 H3CC CN L H H CH3 • Use the…
A: The given reaction is Diels alder reaction. The Diels alder reaction is defined as, Diene in…
Q: What is the structure of the unknown? please determine it by analyzing the spectroscopic data given.…
A: The objective of the question is to determine the structure of an unknown compound using the…
Q: Include all lone pairs and charges as appropriate. Ignore stereochemistry. Ignore inorganic…
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 2合 + 1 <➡1 b. Then, use the sim to check your answer and make any corrections. Prediction Simulation…
A: When the reactants involved in a reaction are taken in non-stoichiometric amounts and allowed to…
Q: The concentration at which micelles first form in solution is known as critical micelle…
A: 1. **Conductivity Measurement**: - As surfactant concentration increases, the solution's…
Q: Predict the major product for the following reaction. EtO. OEt Br- x Br 1) NaOEt 2) H₂O, heat
A:
Q: 65.0 mL of 0.575 M HNO, is titrated by 0.450 M KOH. Calculate the pH of the acid solution before any…
A:
Q: Question 1 (1 point) An electrochemical cell is set up with a Cu(s)|Cu(NO3)2(aq) electrode (E° red =…
A: The objective of the question is to identify the false statements about the operation of an…
Q: Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the…
A: Acidic salts are the substances that produce ions in water upon hydrolysis. Similarly, if they…
Q: Part 1 of 3 N, AS, P O<O<O Part 2 of 3 Sn, I, Rb O<O<O Part 3 of 3 Ca, O, P X
A: 1)The order of ionization energy for nitrogen (N), phosphorus (P), and arsenic (As) is N > P >…
Q: 0.0850 M F (from 0.0850 NaF) Ka for HF=7.20 x 10-4 [Solve First] Kb for F =
A: Step 1: Step 2: Step 3: Step 4:
Q: For the chemical reaction shown on the left, please select all the statements that correctly…
A: Peptides: Two or more amino acids join together to form chains called peptides. In this the amino…
Q: Draw: trans-4-chloro-5-fluorocyclohexyne
A: Step 1: Step 2: Step 3: Step 4:
Q: www-awu.aleks.com/alekscgi/x/Isl.exe/10_u-IgNslkr7j8P3jH-IQUHIQg6bJxmeSyVPHOEB1plef9xyC5Ca9QIIXeFeRg…
A: Higher the atomic number or collective atomic mass, higher will be its priority. Thus priority of…
Q: List carbon-carbon bond formation reactions to synthesize 1-bromohexane from starting materials…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: dont provide handwriting solution...
A: Step 1:- When , we have a solution of CuSO4 , Copper(II) sulfate (CuSO4) dissociates completely in…
Q: Calculating the solubility of an ionic compound when a common ion is... Calculate the solubility at…
A: Step 1:Given that :Ksp = 1.77 × 10-10 [AgNO3] = 0.0090 MWe need to find the solubility of AgCl in(A)…
Q: None
A: Ans part 1 Given Mass of UO2 = 2.26 kg= 2260 g Write the balanced equation UO2 (g) + 4 HF (g) →…
Q: 9. Determine the rate law and the value of k for the following reaction using the data provided.…
A: Given a general chemical equation: A+B→productsRate has a general formulaRatios of 2 different runs…
Q: QUESTION 38 What is the missing chemical, A, in the following reaction? HN HCI + A+ N
A: It is an organic reaction that consists of an amino alkylation of an acidic proton placed next to…
Q: Provide the products of the following acid-base reactions: 요오 ii OH OH OMe e OH OH
A:
Q: None
A: Thank You.
Q: 7. Draw the product of the following reaction explain if there is any stereochemistry. NaOH -HBr Br
A: Step 1: Step 2:In the 1st step there is formation of 3° carbocation by elimination of Br- as leaving…
Q: Curve 3 best represents which species? Ne at 600 K Ne at 450 K Ar at 600 K
A: We need to find which curve is for which gas in the graph.
Q: Chapter 17: Voltaic C B. Effect of Concentration on Cell Po 1. Complex ion formation: - Potential,…
A: The objective of this question is to calculate the residual concentration of free ions in…
Q: Continue and confirm I got the one already there correct and provide answer for the rest
A: all are given here. 1-6 all are correct. 7. ISOBUTANE8. MCPBA9. p-Toluenesulfonic acid10. 1,2-DME11.…
Q: Show the proton and carbon NMR peaks. This can be the NMR graph OR a list of peaks labeled 1,2,3 or…
A: Step 1: In the given molecule, there are 11 (eleven) hydrogens that give the H-NMR signals.The…
Q: Payalben
A: Explanation of Oxidation and Reduction in the Reactions:In each of the reactions you provided, we…
Q: :&:&;;$($$;$
A:
Q: Which of the following statements is False? OA. The thermodynamic system is all the area outside of…
A: Given statements:A. The thermodynamic system is all the area outside of where a reaction takes…
Q: -37.1 Celsius to Fahrenheit round to 3 significant digits
A: Celsius serves as the standard measurement for temperature in both practical and scientific…
Q: Write the common (not systematic) name of each organic molecule. Hint: your answer should have more…
A: Rule to write common name of ketone : In common name of ketones , write name of the alkyl groups…
Q: molecule = cocoa butter find its structure and just deprotonate it to stimulate the soap reaction;…
A: The objective of this question is to predict the structure of cocoa butter’s molecules’…
Q: 2. For the following reaction scheme a. Propose an efficient synthesis for the transformation of…
A: a. To synthesize 3-methylbutan-2-one from 3-methyl-1-butene, one efficient method involves a…
Q: Which of the following species will NOT form an alkaline solution in water? A. LICNOr b. KCIO2c. Nal…
A: Four options are givenIt is required to determine which one doesnot form an alkanline solution in…
Q: Here are sketches of four electron orbitals: O A B C D O yes Are any of them s orbitals? 0,0,... no…
A: Answer:Orbital is the region around the nucleus where probability of finding an electron is maximum.…
Q: 8. Our friend carbon. Recall that all organic molecules, by definition, contain carbon. Carbon is a…
A: The objective of the question is to-Write the chemical formula of Methane.Draw the ball and stick…
Q: I 3. All radioactive decay processes follow first-order kine - ics. What does this mean? What…
A: The statement that all radioactive decay processes follow first-order kinetics means that the rate…
Q: How much lead(II) chloride will precipitate if 185 mL of 0.021 Msodium chloride is added to 235 mL…
A: Step 1:To find the amount of lead (II) chloride that will precipitate, we need to know the limitting…
Q: :&:&;(&($$;&:&:&&:&:$:;&;&&:&:&;&
A:
Q: 1. What is the chemical formula for each of the following compounds? (3 point × 4) a. Octane b.…
A: The objective of the question is to determine the chemical formulas for the given compounds and to…
Q: dont provide handwriting solution ...
A: The objective of this question is to calculate the change in internal energy (ΔU) of the reaction…
Q: Can you include the species please
A: Step 1:KC3H5O3 is potassium lactate. It will dissociate to potassium ions (K+ ) and lactate ions (…
Q: None
A: Correct Option - Maltose In the context of maltose, the alpha (1→4) glycosidic bond is formed…
Q: Please provide appropriate reagents to achieve the conversion of B into α,ẞ-unsaturated ester C over…
A: Given is organic synthesis reaction.Here, we need to find out reagents for conversion of B to C.
Q: 2. What affect, in any, would this error have on the molarity of acid in part two? Would you…
A: The calculation for the average molarity of NaOH, 0.1137 M is correct from the Data table:…
Q: Draw the product formed by the reaction of pyruvate with lactate dehydrogenase during lactic acid…
A: Detailed explanation: Let's revisit the image and break down the reaction step by step:Starting…
Q: None
A: The given pH of the solution is 10.15. To calculate the pOH of the solution, we need to know that…
Step by step
Solved in 2 steps with 1 images

- Lithocholic acid is an A–B cis steroid found in human bile. Draw lithocholic acid showing chair conformations, as in Figure 27-11, and tell whether the hydroxyl group at C3 is axial or equatorial.Can you help me sort these into Chiral and Achiral categories?Based on the image. What is 1. The anti-conformer of hexane in Newman projection. 2. The equivalent structure of LAM in sawhorse representation. 3.
- Draw all possible configurational stereoisomers for the given molecule below. Designate the R/S on the stereogenic centers and E/Z on the C=CDraw the Fischer, the Haworth, and the most stable chair conformation (equatorial vs axial) of β-D-mannose. Please number your carbons. Please draw it by hand and not by the internet through screen shot. Please explain. Thank you!Draw (2S, 3R)-2-bromo-3-chloropentane on the Fischer projection shown below.
- Draw each compound in its most symmetric conformation, star (*) any asymmetric carbon atoms, and draw any mirror planes.Label any meso compounds. You may use Fischer projections if you prefer.(a) (2R,3S)-2-bromo-3-chlorobutaneFor each pair, indicate whether they are constitutional isomers, enantiomers, diastereoisomers, or identical molecules. A BDEF(a) (S)-2-chlorobutane, draw any enantiomer.
- In a fischer projection, is the molecule r or s, and what is the highest priority?Locate the stereogenic centers in the attached Newman projection and labeleach center as R or S.Draw all possible stereoisomers for the molecule in Fischer projection and label the srelationship between each of the isomer.



