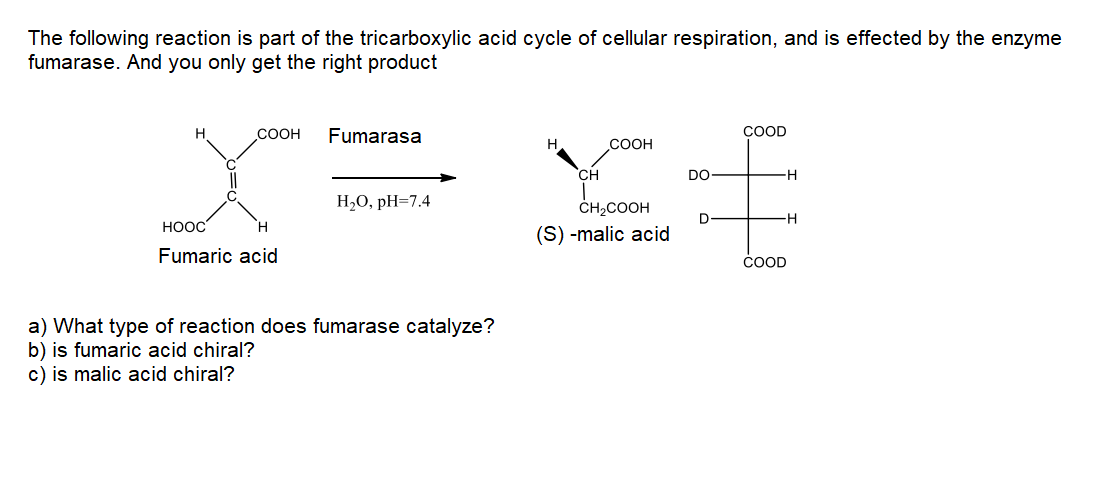
COOH Fumarasa COOD H. COOH CH DO H- H2O, pH=7.4 ČH2COOH D H. НООС H. (S) -malic acid Fumaric acid COOD a) What type of reaction does fumarase catalyze? b) is fumaric acid chiral? c) is malic acid chiral?
Q: A biochemical reaction transfers 60 kJ mol-1(15 kcal mol-1) of energy. What general process most…
A: Biochemical Reaction : It is the transformation of one molecule to a different molecule inside a…
Q: Consider the following dipeptides: TY SW HF LC LR How many of the dipeptides in this group that…
A: Amino acids are monomers that make up peptides. Amino acids found in proteins are α-amino acids.…
Q: Potassium superoxide, KO2, is used in rebreathing masks to generate oxygen according to the reaction…
A: A chemical reaction is a process by which a bunch of molecules called the reactants are converted…
Q: For each of the following reactions, give a balanced net-ionic equation. Sulfate Ion a) MgSO4 +…
A: Note: As per guidelines we answer the first question. Kindly resubmit the questions to be answered.…
Q: I. Predict the outcome of the following reactions. If positive write what will you observe (color,…
A: Introduction 1.) This is the reaction to Bial's orcinol test for the detection of pentoses. A…
Q: Substrate Concentration (mol L1) Velocity (mM min-1) 2.500 0.588 1.000 0.500 0.714 0.417…
A: According to Michaelis-Menten Kinetics, when the rate or velocity of an enzyme catalyzed reaction…
Q: Define the following terms:a. allyl groupb. epoxidec. SAM d. PAPSe. phase I reaction
A: In chemical structure, the group of atoms that replace the hydrogen atoms on the parent hydrocarbon…
Q: Enzyme A, B, and C having the following properties are to be separated by various chromatography at…
A: The chromatographic process is generally helping a solvent that destroys an adsorbed substance from…
Q: Give 5 examples of molybdenum complexes. State their physical and chemical properties and their…
A: The atomic number of molybdenum is 42 and it is represented by the symbol 'Mo'. On the earth,…
Q: Name colour Reactions okAmino Acidd t puoteine.
A: The presence of amino acids can be detected using some specific reagent known as colour reactions…
Q: The pl of alkaline phosphatase is 4.5; the pl of the DEAE cellulose is 10.5. We used a buffer of pH…
A: Ion exchange chromatography is a chromatographic separation techniques based on the charge of the…
Q: Consider the following dipeptides: PC HE TG IR FL How many of the dipeptides in this group that…
A: Dipeptides are compounds which are made by the combination of two amino acids. Both the amino acids…
Q: What is the definition of hydrolosis reaction?
A: In human body, digestion and glucose metabolism are examples of hydrolysis reaction. Solubilization…
Q: For a Michaelis Menten reaction the following rate constants were observed: k1 =7x 10/M-sec k1=1x…
A: The correct answer is option, d. 0.0003M.
Q: In the electrolysis of aqueous sodium bromide, there are two possible anodic reactions: *2H2O(l)…
A: Water is reduced at the cathode in the electrolysis of NaBr. This occurs because water is more…
Q: What is the [CH3COO−]/[CH3COOH] ratio inan acetate buffer at pH 4.00?
A: A buffer is a mixture of weak acid/weak base and its conjugate base/acid. It is an aqueous solution.…
Q: Please balance the following half-based reactions: SbH3 → Sb (acidic solution) BrO3- → Br2 (acidic…
A:
Q: For the series of reactions below, what is the overall reaction and the value of ∆Go’(kJ/mol) for…
A: Consider a set of reactions 1 and 2; Reaction 1 : a→b have a ∆G0' = y Reaction 2 : c→d have a ∆G0' =…
Q: Is starch soluble in water? How might that be helpful in determining the rate of reactivity? How…
A: Starch is a white, odorless, and tasteless substance. It is made up of glucose molecules which…
Q: Give full reaction for the synthesis of Diclofenac sodium
A: Diclofenac Sodium is the sodium salt of diclofenac, a benzene acetic acid derivate and NSAID with…
Q: two chemical structures of aldehyde or ketone
A:
Q: Consider the following dipeptides: AC HE TG LR VK How many of the dipeptides in this group that…
A: An ion-exchange column is a chromatography method to separate molecules based on charge. An anion…
Q: Calculate the equilibrium concentration of H2O for the following esterification reaction performed…
A:
Q: What is oriental flush? Mention the cause of oriental flush. A. What type of molecule will be the…
A: Alcohols are organic compounds. They have one or more hydroxy groups bonded on the parent chain of…
Q: Suppose you have extracted an alkaloid from natural sources how will you establish the structure of…
A: Alkaloid are one of the major classes of phytochemicals, i.e. plant secondary metabolite along with…
Q: -8 K, for HCO3 ` = 2.38 × 10 Calculate Ka for the conjugate acid ( H¿CO;) For the reaction :…
A: Conjugate acid-base pairs are those that differ from each other by a proton or H+ ion. For example,…
Q: Determine whether or not reaction is a redox reaction. For each redox reaction, identify the…
A: Redox reaction is considered as a process, during which one element is must be reduced or oxidized…
Q: The reaction quotient is Q=1.6×10-26 Part B What pH is needed to produce this value of Q if the…
A: The reaction quotient tests the relative quantities of compounds and reactants present at a certain…
Q: Consider the following reaction: Glucose-1-phosphate → Glucose-6-phosphate ΔG° = −7.1 kJ/mol What is…
A: Equilibrium constant is the reaction quotient of a chemical reaction, at the stage at which the…
Q: Why is picric acid used for burns? Explain the principle involved
A: Cells are the basis of life. Cells give rise to tissues and organs. Organs that work towards the…
Q: According to this standard curve for chlorophyll dissolved in ethanol, what is the concentration of…
A: the concentration of betacyanin = 3 micromolar
Q: CH, o-CH, CH, 0- 0. CH, OH ATP ADP O. H HO H. HO. H HỌ OH phdrutk 1 H HỌ OH H Frucione& phosphate…
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: NH HO-P-o Br, in H,O OH он ОН Ba(ОН),
A: ATP→cAMP(cyclic AMP) Adenosine triphosphate reacts with barium in presence of water or with barium…
Q: What is the name of reagent used? (b) What is your observation if any unreacted starting material is…
A: Ans. Aspirin is a common drug, used as an anti-inflammatory and in the diagnosis of minor aches,…
Q: Consider the proteins in the table below. Protein M.W. Isoelectric point (pl) Alcohol dehydrogenase…
A: Proteins are polypeptide sequences that are folded into secondary and tertiary structures based on…
Q: Give the relative rates of reaction of the four carboxylic acid derivatives below with aqueous…
A: Introduction: Most reactive = b 2nd most reactive = a 3rd most reactive = c Least reactive = d
Q: Give a complete and well descriptive definition of the following: 1.1 Fractional saturation…
A: Hi! Thank you for your question. As you have posted multiple subparts and have not mentioned which…
Q: If catalase is tested with the biuret reagent, what would be the ideal result? Why? If catalase is…
A: Hi, thank you for posting the question on Bartleby. As per the guidelines, we can answer only one…
Q: Explain why the blue color of starch-iodine complex disappears upon heating?
A: The iodine-starch is a chemical reaction that is used to test for the presence of starch or for…
Q: Among acetic acid, formic acid, lactic acid, benzoic acid, oxalic acid, and succinic acid. Which…
A: Carboxylic acids are organic compounds that have a carboxyl (-COOH) group. They possess carbon (C),…
Q: What will be the complete hydrolysis products of the given structure below? CH₂-0-C-C17H37…
A: Given structure is a phosphatidylcholine (PC) It belongs to the class of phospholipids that has…
Q: Explain the process of polyacrylamide reaction, and what are the two reagents are crucial for this…
A: Polyacrylamide is defined as a mesh like matrix that is suitable for the separation of proteins of…
Q: I Write a balanced equation for each of the following two reactions that you will use in this…
A: The substances undergoing reactions are called reactants and are written on the left side of the…
Q: The conversion of glucose-1-phosphate to glucose-6-phosphate by the enzyme phosphoglucomutase has a…
A: According to standard free energy change ∆G0 = -RT ln Keq where ∆G0 =standard free energy change…
Q: Calculate the overall ΔG° (report up to two decimal places) for the net reaction (see attached…
A: Standard free energy change is the Gibb's free energy at 273k temperature and 1 atm pressure ∆G°=…
Q: H2N NH3 HO, H,O H2N. NH2 СООН COOH glutamic acid glutamine why does the reaction not proceed?
A: Amino acids are units that synthesize proteins. Amino acids are synthesized by ribosomes. The…
Q: Calculate the boiling points of a 8.50 m aqueous solution of fructose. Boiling point constants can…
A: Formula for boiling point: ∆Tb=Kbmwhere,∆Tb= change in boiling pointkb= boiling point constantm=…
Please be clear in your writing
Step by step
Solved in 2 steps

- Both prokaryotic and eukaryotic organisms carry out some form of glycolysis. How does ha fact support or not support the assertion that glycolysis is one of the oldest metabolic pathways?Based on your knowledge of the structure of NAD+ and an assumption that coenzyme dissociation is the rate limiting step of the alcohol dehydrogenase mechanism, hypothesize why a N249W mutation at the coenzyme binding site would increase the rate of catalysis.which of the following glycolytic reactions is an oxidation? a) G3P to 1,3-BPG b) PEP to Pyr c) lactic acid fermentation under anaerobic conditions d) condensation of acetate with OAA e) succinate decarboxylase reaction
- The enzyme glutamine synthetase catalyzes the following reaction: glutamate + ATP + NH3 → glutamine + ADP + phosphate Which of the following statements about this reaction is correct? he reaction is only exergonic if enzyme is added b. The reaction is endergonic whether or not enzyme is added c. The reaction is exergonic whether or not enzyme is added d. The rate of oxygen production will be unaffected e. The reaction is only endergonic if enzyme is addedDoes the complete oxidation of tridecanoic acid (C13:0) make more ATP than the complete oxidation of 9-tetradecenoic acid (C14:1, Δ9) inside human body? Show your calculation.The pyruvate →lactate reaction in animals is reversible, but the pyruvate → ethanol fermentation in yeast is irreversible. Explain.
- The enzyme glutamine synthetase catalyzes the following reaction: glutamate + ATP + NH3 → glutamine + ADP + phosphate Which of the following statements about this reaction is correct? a. The reaction is endergonic whether or not enzyme is added b. The reaction is only endergonic if enzyme is added c. The reaction is only exergonic if enzyme is added d. The rate of oxygen production will be unaffected e. The reaction is exergonic whether or not enzyme is addedAssume that you have a solution of 0.1 M glucose 6-phosphate. To this solution you add the enzyme phosphoglucomutase, which catalyzes the reaction: glucose-6-P ⇔ glucose-1-P ∆G°′ = +1.7 kJ/mol (a) Does this reaction proceed at all as written at 25 °C, and if so, what are the final concentrations of glucose 6-P and glucose 1-P? (b) What effect would omitting the enzyme have on the reaction. Be specific. (c) Under what cellular conditions, if any, would this reaction continuously produce glucose 1-P at a high rate?FAD is a coenzyme for dehydrogenation.(a) When a molecule is dehydrogenated, is FAD oxidizedor reduced?(b) Is FAD an oxidizing agent or a reducing agent?(c) What type of substrate is FAD associated with, and whatis the type of product molecule after dehydrogenation?(d) What is the form of FAD after dehydrogenation?(e) Use the curved-arrow symbolism to write a generalequation for a reaction involving FAD.
- ATP synthase, hexokinase, and isocitrate dehydrogenase are important enzymes in aerobic respiration. Please provide a description for each enzyme containing the following information: (1) the metabolic stage in aerobic respiration they are involved in, (2) the enzyme class where they belong, and (3) the chemical eaction they catalyze.Which reaction in the citric acid cycle is most analogous to the oxidative decarboxylation of 6- phosphogluconate to ribulose 5-phosphate? What kind of enzymebound intermediate is formed in both reactions?Under standard conditions, is the oxidation of ubiquinol (Coenzyme Q) by O2 sufficiently exergonic to drive the synthesis of ATP? If yes, how many ATP can be synthesized assuming 100% efficiency?



