Q: Check the box under each structure in the table that is an enantiomer of the molecule shown below.…
A: Given are organic compounds.Enantiomers are isomers that are non-superimposable mirror images of…
Q: Table 18.2 K, Values for Some Monoprotic Acids at 25°C Name (Formula) lodic acid (HIO) Chlorous acid…
A: buffer is a solution which resists any change in pH on adding a small amount of acid or base. it is…
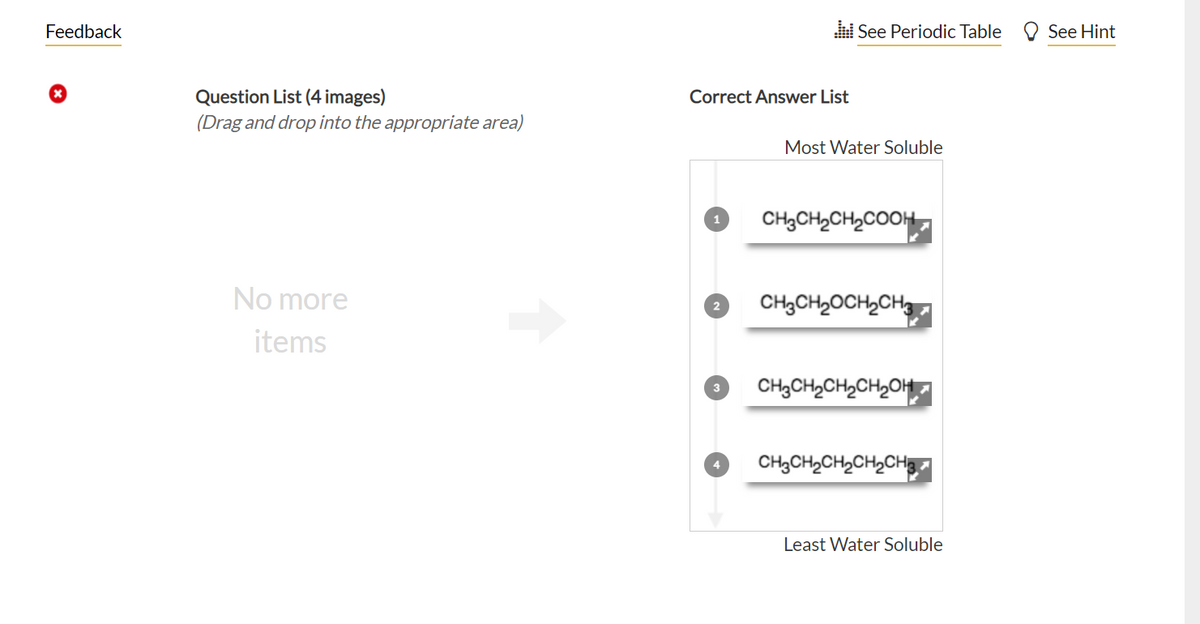
Q: Q31ls 30. It is the linear structure of CH3CO(CH2)3AND2CH3: رمند منو نمید ا = || =
A: linear structure: also known as skeletal structure, a representation of molecular structure in…
Q: Consider the reaction coordinate diagram shown. Which step has the greatest rate constant going in…
A: When the reactant molecules convert into products, they require the minimum amount of excess energy.…
Q: Which represents the correct arrow-pushing for the following transformation? A. B. C. D. HeBr H¬Br:…
A: The arrow represents the movement of electrons from higher electron density to lower electron…
Q: Given the information about the following acids i) HW pka = 2 ii) HX pKa = 10 iii) HY pKa = 12 iv)…
A: Dissociation of a weak acid is given by:he acid dissociation constant of this acid is given by:For…
Q: 1. A sample of a funeral shroud taken from an ancient Egyptian tomb is found to have C-14 activity…
A: Carbon-14, or C-14, is a type of carbon found in living things. When a living organism dies, the…
Q: With the help of a neat flow chart, describe the manufacture of ethyl alcohol by fermentation…
A: The flow chart for the fermentation process of ethyl alcohol can be represented as :Raw Materials:…
Q: Which of the following bonds would you expect to have the largest frequency of vibration? Α) 1Η-1Η…
A: The frequency of bond vibration depends on the strength of the bond and the masses of the bonded…
Q: 4. HO Which of the following compounds can potentially be prepared as a single enantiomer? -OH OH ОН…
A: Enantiomers, also called optical isomers, are produced when atoms in a molecule are arranged in a…
Q: a sample of gold and quarts weighs 100g the density of gold 19.3 and quarts 2.65 and for sample 0.4(…
A: We are given the density of the sample, which is . We are also given the densities of gold and…
Q: Name two uses of lipids as hormones and two uses as vitamins in human body
A: Lipids, essential components of our diet, are diverse molecules with vital roles in our body.…
Q: In the above redox reaction, use oxidation numbers to identify the element oxidized, the element…
A: Answer:Oxidizing agent is the specie that oxidizes others and itself gets reduced during the…
Q: Calculate the mass of iron (III) oxide (Fe₂O3) that contains a million (1.00 x 106 ) iron atoms. Be…
A: number of iron atoms = 1.00 × 106 atomsmass of iron(III) oxide (Fe2O3) = ?Note:molar mass of…
Q: Draw the major product(s) of the following reactions including stereochemistry when it is…
A: Aklkyns are rich in electron density. So, usually they do not undergo nucleophilic addition…
Q: Given the standard potential for hydroxide formation is -1.59V and the standard potential for Mn²*…
A: This problem is based on electrochemistry and Second law of Thermodynamics.Here, we have to use…
Q: Sulfur hexafluoride gas is collected at 29.0 °C in an evacuated flask with a measured volume of 5.0…
A: The temperature, pressure, volume, and mole of the gas are related by the ideal gas equation. The…
Q: In the following reaction, iodine monochloride (ICI) effectively serves as a source of an…
A: OMe is an ortho/para directing group
Q: What reagent(s) is needed to complete the transformation below? Options: BH3, then NaOH/H2O2…
A: Find out suitable reagent for carried out above transformation.When bromination of alkenes is…
Q: I balanced the equation but I can’t seem to do the next part
A:
Q: The answer is 0.4 atm, please show steps, thanks.
A: Answer:Limiting reagent is the reactant that exhuast first in the chemical reaction and decides the…
Q: a. 0.180 M sodium chloroacetate (Ka (CICH₂COOH) = 1.36 × 10 °)? [H₂O+]-[ = M -3 b. 3.90 × 10-³ M…
A:
Q: A solution is made by dissolving 11.4 g of potassium cyanide, KCN, in enough water to make exactly…
A:
Q: At 25.0 degrees Celsius the Henry’s Law constant for phosphine (PH3) gas in water is 8.1 x 10^-3…
A:
Q: At 298 K, Kc is 2.2×10^5 for the reaction F(g)+O2(g)⇌O2F(g). What is the value of Kp at this…
A:
Q: In Breckenridge, Colorado, the typical atmospheric pressure is 520. torr. What is the boiling point…
A:
Q: Select the final major organic product of this reaction. ویت ملی OH CHLU CH₂ ?
A: Carboxylic acids are acidic in nature. If a carboxylic acid reacts with a base it donate a proton…
Q: 56.6 mL of 3.00 M Ca(OH)2 is added to 100.00 mL of an HNO3 solution before the equivalence point is…
A: The question is based on the concept of titration.We are titrating nitric acid with calcium…
Q: Classify each chemical reaction: KBr(aq) + AgNO3 (aq) 1 reaction KNO,(aq) + AgBr(s) CH, OCH (1) +…
A:
Q: Please don't provide handwritten solution .... do these reactions give identical products, show…
A: Hydroboration is the addition of borane to unsaturated to form alkyl borane. In BH3, hydrogen is…
Q: 6. Describe the differences between A,Gº and A,G and what each of them represents.
A:
Q: How many sp2 carbon does Vitamin A contain? 5 10 4 8 Vitamin A OH
A: Hybridization of carbon (C) in hydrocarbons:• Alkane-C-C- in this case all bonds are single and the…
Q: Given the table below, what is the value of Kc for the following reaction? 3 A (aq) = B(aq) + C(aq)…
A:
Q: The Arndt-Eistert synthesis involves a rearrangement (known as a Wolff Rearrangement) with…
A: The given reaction scheme is .We have to give the mechanism of the above reaction.
Q: 1. Provide stepwise synthesis including reagents and intermediates for the following syntheses. OH…
A: Alcohols reacts with thionyl chloride to give an alkyl halide.. Thus, cyclohexanol reacts with…
Q: Ag2O NH OH 0 0 OH
A:
Q: Which of the following compounds would NOT give an IR spectrum in the typical range of 650-4000…
A: For a molecule to be IR active there must be a change in dipole moment as a result of the vibration…
Q: Draw a structural formula for the major organic product of the reaction shown below. CH3(CH2)5CH2…
A: Given reaction is addition of Gilman reagent on alkenyl halide.Gilman reagent is represented as…
Q: A 1.41 L buffer solution consists of 0.334 M propanoic acid and 0.158 M sodium propanoate. Calculate…
A: Given that,Total volume of the buffer solution is V = 1.41 L.The concentration of the propanoic acid…
Q: Some bond dissociation energies in kJ/mol are listed. 1°, 2°, 3° means primary, secondary,…
A: In the given reaction a primary cyano group is converted to a tertiary cyano group.Now, in the…
Q: Carbon disulfide and oxygen react to form carbon dioxide and sulfur dioxide, like this: CS₂(g)…
A: LeChatelier's principle: When factors like concentration, pressure, temperature, inert gases that…
Q: Draw the major product that forms for the following reaction. Use a line structure which means that…
A: Alkanes react with bromine in the presence of light to give alkyl bromide. The reaction proceeds by…
Q: What is the major organic product in the ollowing reaction? HO p-TsOH OH ? 1 S What is the major…
A: Given is organic reaction.Given reactant contain two functional group: ester and ketone.Reagent…
Q: How many moles of Al are needed to produce 6.0 moles of Al2S3 via the chemical reaction 2Al +35…
A: Balanced chemical equation : 2Al + 3S ➡️ Al2S3 When 2 moles of aluminium react with 3 moles of…
Q: Which of the alkynes below, after undergoing an acid- catalyzed hydration, would be expected to…
A: The Alkyne containing a carbon carbon triple bond having a general formula is CnH2n.Note ~ Since you…
Q: Determine the volume. in ft, of 2 lb of a two-phase liquid-vapor mixture of Refrigerant 134A at 28°F…
A: Given:To Find:The pressureThe volumeHere,
Q: Explain why the following reaction takes place as shown. Be sure to fully explain why the reaction…
A: Question is based on the concept of organic reactions. We need to explain the product formation of…
Q: [Review Topics] [References] Use the References to access important values if needed for this…
A: Multiple questions
Q: 5) Show how one might synthesize each of the following compounds starting with either benzyl bromide…
A: We have synthesized the below-given compounds from either benzyl bromide or allyl bromide.
Q: A B Energy Energy Br Br TM = Reaction Coordinate ||| CH3OH heat CH3OH heat Energy QUO C Energy…
A: In given reactions alkyl bromide is converted into alkene in the presence of methanol and heat.So,…
Step by step
Solved in 3 steps with 2 images

- #4. answer is on bracket. thank uPlease answer super fast and answer all questions and show calculations For the image attached For 1. a Mass of metal: Trial 1 is 35.0228 g Trial 2 is 35.0915 g Trial 3 is 34.0821 g Mass of water: Trial 1 is 20.0177 g Trial 2 is 20.0250 g Trial 3 is 20.0168 g For delta t of water: Trial 1 is 15.5 C Trial 2 is 15.7 C Trial 3 is 15.1 C For delta t of metal Trial 1 is 80.1 C Trial 2 is 80.2 C Trial 3 is 79.5 C For B my calculated Specific heat is: Trial 1 is 0.462 Trial 2 is 0.467 Trial 3 is 0.4664- Please I want answer sub-parts (c) and (d) with steps and clear typing or hand writing. Many thanks
- Can someone answer nos. 5 to 10? Thank you very much! If it's not too much, provide an explanation for each number. Thanks!Answer #1 and #2. Prioritize answering #2.Second-degree Equation Step 1. Copy the given table on your graphing paper, after your student details. X (% concentration) 0.50 1.00 1.50 2.00 2.50 3.00 Y (oil separation) 5.10 3.60 2.60 2.00 1.40 1.00 Log Y Step 2. Initially plot the x and y values on a regular graphing paper (or millimeter graphing paper - if available, preferred for precision). Label then connect the points to observe curved line. Step 3. Since the line is curved, the y-intercept cannot be easily determined because you can’t exactly determine the direction of the line once extrapolated. To obtain a straight line, first, compute the logarithm values of all the y values up to the 2nd decimal place (for the purpose of easy plotting later) and fill in the table. Step 4. On another graphing paper, plot then label the x and log y. Using a dotted line, extrapolate the straight line to the y axis. Label this point as “a” for the y-intercept. Step 5.…

