Q: Enter your answer in the provided box. The vapor pressure of a liquid doubles when the temperature…
A: Vapor pressure of a solvent or solution is proportional to the temperature of the solvent or the…
Q: Please complete the retrosynthesis for both of these problems. Thank you.
A:
Q: How do you calculate the molar ΔHsoln for a compound using coffee cup
A: To calculate the molar enthalpy of solution (ΔHsoln) for a compound using coffee cup calorimetry,…
Q: Explain the relationship between color intensity and amount of reaction.
A: The relationship between color intensity and the amount of reaction is that the intensity of color…
Q: A chemist adds 450.0 ml. of a 0.51M barium acetate (Ba(C₂H,0₂),) solution to a reaction flask.…
A: Definition of one molar or M solution is that one mole of solute is being dissolved in 1 L of the…
Q: = O SIMPLE REACTIONS Predicting precipitation Complete the table below by deciding whether a…
A:
Q: A 6.00 mol sample of solid A was placed in a sealed 1.00 L container and allowed to decompose into…
A: Initial moles of sample A are 6.00 molesVolume is 1.00 LEquilibrium concentration of B = 1.40 M
Q: 3. 50.0 mL of 1.0 M sodium hydroxide and 50.0 mL of 1.0 M hydrochloric acid, both at 22.5 °C, were…
A: Given :---- Mass of sodium hydroxide solution (mNaOH) = 50.0 gMass of hydrochloric acid solution…
Q: Choose the best reagents to complete the reaction shown below. •1118 1. PhMgBr 2. H3O+ OH Br AL A B…
A:
Q: A website promoting the use of alternative energy vehicles and hybrid technologies claims that "A…
A: Gallons original quantity = 500.0 gallonsCO2 produced from 500 gallons = 5 tonsGasoline primary…
Q: For the following reaction write out the equilibrium law : a) 2C4H10(g) + 13O2(g)…
A:
Q: Complete the following fission reaction: In+235U. 92 → 131 Sn + ? + 2n + energy 50
A:
Q: For the following nuclides: vii. Iodine-131 viii. Plutonium-236 a. Determine the most likely mode…
A: Radioactive decay is the spontaneous process through which an unstable atomic nucleus transforms…
Q: Consider NaBr and LiF. The LiF has a stronger ionic bond because A. The number of protons and…
A: The LiF has a stronger ionic bond than NaBr
Q: The conjugate acid of HSO4 is H₂SO4. HSO4. HSO3. H₂SO4. SO4²-
A: • Acid and Base:Acid: acids are proton (H+) donor.Base: bases are proton (H+) acceptor.• Conjugate…
Q: 1. Which quantity does not increase when the temperature of a reaction system is raised? a.…
A: Quantity does not increase when the temperature of a reaction system is raised
Q: Identify the correct classification for the reaction 2 PbSO 4 →2 PbSO 3 + 0 2 combination…
A: Combination reaction: A + B ----> AB Decomposition reaction: AB -----> A + B Single…
Q: A chemist prepares a solution of aluminum chloride (AIC13) by measuring out 8.17 g of aluminum…
A: Given,The mass of aluminum chloride ( AlCl3 ) = 8.17 gVolume of the solution = 100. mL
Q: Calculate the [H+] in a solution that has a pH of 9.88.
A: pH of any solution is the potential of hydrogen ions . It is given by :pH = -log[H+ ] where [H+ ] =…
Q: Give only typing answer with explanation and conclusion Write the main product in the following…
A: Aldol condensation is acid as well as base catalyzed reaction. This reaction involves the formation…
Q: ? Reactions You May Utilize: 1. BH3 2. H₂O₂ / "OH 3. SOC1₂ 4. Ph3P=CH₂ 5. 2 Liº 6. CH3OH 7. HI 8.…
A: Borane is used for reduction of double bond . Peroxide is used for oxidation of borane . Dichromate…
Q: Calculate the solubility at 25 °C of AgBr in pure water and in 0.41 M Nal. You'll probably find some…
A: Concentration of NaI = 0.41 M
Q: Draw the possible products of this epoxide ring- opening reaction. Use a dash or wedge bond to…
A: Epoxide ring opening is a chemical reaction in which an epoxide ring (a three-membered cyclic ether)…
Q: 11) (12) 13) & CH₂ + CH3NH₂/H+ N(CH3)₂ CH 3 CH ₂ CH CH CH ₂ енз PLATHY □ EOH OLAINY o 2) H+ H₂O₂…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: 3. Draw the expected major and minor products for the following reaction. Label one Markovnikov…
A: Hydrohalogenation of alkene.
Q: I recovered 3 mL of cyclohexene, how many grams of water was produced in the reaction please show…
A: Details
Q: Please provide answers of all table and get the thumbs up please provide me correct solution I Will…
A:
Q: Calculate the energy required to heat 1.40 kg of ethanol from 1.1 °C to 17.9 °C. Assume the specific…
A: Heat absorbed by the ethanol is given by:Where,q = heat absorbedm = mass of the ethanolCv = heat…
Q: Draw the products of the two step reaction sequence shown below. Use a dash or wedge bond to…
A: Under basic condition the reaction occurs via SN2 mechanism and the less hindered side of epoxide is…
Q: Show how to make the following from acetoacetic ester. L ماک how acet aceste 0 OH ti make ester.…
A: We have to synthesise the given molecules form acetoacetic ester.
Q: What are the moles of N2 present at equilibrium at 450oC if 1.00 mol N2 and 4.00 mole H2 in a 10.0 L…
A: The equilibrium constant is given by the ratio of product concentration to the concentration of…
Q: Calculate the probability that a ClO molecule treated as a harmonic oscillator will be found at a…
A: We have find out the answer.
Q: Draw the products of the reaction shown below. Ignore inorganic byproducts. If the reaction results…
A: This reaction is an example of an electrophilic aromatic substitution reaction.In electrophilic…
Q: If dissolving 1.5 g of a solute into 100 mL of water caused the temperature of the solution to…
A: Given => mass of solute = 1.5 g Volume of water = 100 ml Temperature of solution increases by…
Q: When a molecule absorbs a photon, one of its electrons is raised to the ______ state. quantum…
A: Photons are smallest units of energy. They are the smallest unit of electromagnetic radiation. These…
Q: Determine the equilibrium constant for the system N2O4 2NO2 at 25°C. The concentrations are shown…
A: Given reaction: The expression of the equilibrium constant, ... (1)
Q: IZ Br₂ / FeBr 3
A:
Q: 16. Step 1 Step 2 Step 3 Students in a SCH4U class were examining the reaction between…
A:
Q: A buffer solution contains 0.492 M NaH₂PO4 and 0.358 M K₂HPO4. Determine the pH change when 0.102…
A: Concentration of NaH2PO4 = 0.492 MConcentration of K2HPO4 = 0.358 MMoles of HI = 0.102 molVolume of…
Q: N ОН 2- S2O3, Ag, H
A: Organic transformation
Q: Curved arrows are used to illustrate the flow of electrons. Follow the curved arrows and draw the…
A: Mechanism for Organic transformation.
Q: H An H 1 2 -H H 3 4 H H a. 1 is aromatic, 2 is aromatic, 3 is aromatic, 4 is not aromatic b. 1 is…
A: For any molecule to be aromatic: 1. compound must be cyclic 2. compound must be conjugated 3.…
Q: Name the following compound: CI H₂C-CH₂-CH-CH₂-CH₂-CH-CH-CH₂ H₂C-CH₂ она Select one: O a.…
A: The IUPAC name of the compound is given by using the rules of IUPAC nomenclature.The rules can be…
Q: shows a mixture of molecules: carbon - nitrogen oxygen key O hydrogen. O sulfur chlorine wing…
A: When the given quantities of the reactant molecules involved in the chemical equation are not…
Q: The pH of an aqueous solution that contains 3.4 x 10 -2 M NaOH is ______. What is the pOH?
A: Concentration of NaOH solution = [NaOH] = 3.4 × 10-2 MpH of the solution = ?pOH of the solution = ?
Q: CI AICI 3
A: In the given reaction an aromatic hydrocarbon is reacting with an alkyl chloride in the presence of…
Q: Label the following NHCHOI 95 12° 3° or quarterNary. (H) -NH₂
A:
Q: An aqueous solution is 3.50 % by mass potassium bromide, KBr, and has a density of 1.02 g/mL. The…
A: % By mass = 100 x Mass of solute/Mass of solutionNumber of mol = Mass/molar massMole fraction of…
Q: Draw the two products of the reaction shown below. Assume only 1 equivalent of HBr is consumed in…
A: Hydrolysis of ethers typically involves the cleavage of the ether bond (R-O-R') by water (H2O) in…
Q: Draw the product of the reaction shown below. Ignore inorganic byproducts. .O₂N. NaOCH3 Drawing NO2…
A: This reaction is an example of nucleophilic aromatic substitution reaction.Nucleophilic aromatic…
Could you show a plausible Mechanism for this Mannich Condensation and give a short explanation ?
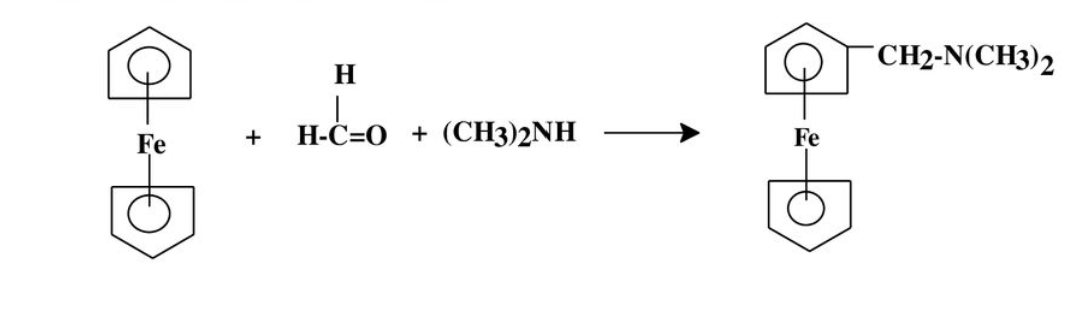
Step by step
Solved in 3 steps with 1 images

- Rank the following groups in order of decreasing priority. −Cl, −CH3, −SH, −OHRank the following groups in order of decreasing priority. −CH=CH2, −CH3, −C≡CH, −HRank the following groups in order of decreasing priority. a.−COOH, −H, −NH2, −OH b.−H, −CH3, −Cl, −CH2Cl c. −CH2CH3, −CH3, −H, −CH(CH3)2 d.−CH=CH2, −CH3, −C≡CH, −H
- Assign a priority order (from least to highest priority) to each of the following sets of groups: I. -OCH II. -NH(CH3)2 III. CH2NH2 IV. -OH A. IV, I, III, II B. III, II, IV, I C. II, III, IV, I D. III, IV, I, IIWhich of the following would be has the highest priority according to the sequence rules? a. —SH c. —CH 3 b. —NH 2 d. —OHZn + H2SO4=
- 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.Rank the following groups in order of decreasing priority. a.−F, −NH2, −CH3, −OH b.−CH3, −CH2CH3, −CH2CH2CH3, −(CH2)3CH3 c.−NH2, −CH2NH2, −CH3, −CH2NHCH3 d.−COOH, −CH2OH, −H, −CHO e.−Cl, −CH3, −SH, −OH f.−C≡CH, −CH(CH3)2, −CH2CH3, −CH=CH2

