I recovered 3 mL of cyclohexene, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.
I recovered 3 mL of cyclohexene, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.27QAP
Related questions
Question
I recovered 3 mL of cyclohexene, how many grams of water was produced in the reaction please show how?
Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.
Please ASAPPPP you guys didn't answer the last one
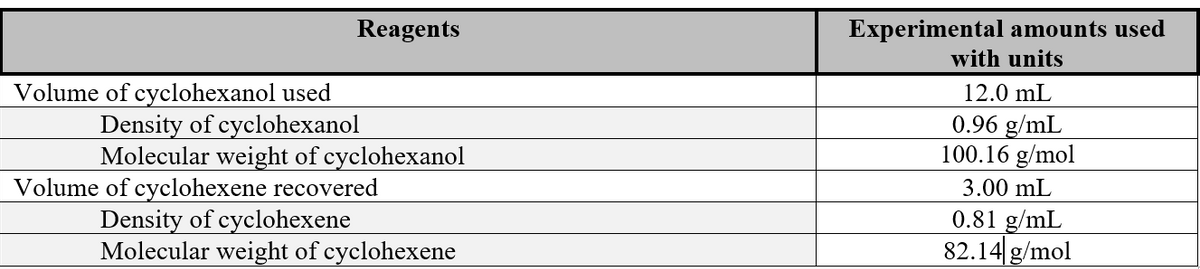
Transcribed Image Text:Reagents
Volume of cyclohexanol used
Density of cyclohexanol
Molecular weight of cyclohexanol
Volume of cyclohexene recovered
Density of cyclohexene
Molecular weight of cyclohexene
Experimental amounts used
with units
12.0 mL
0.96 g/mL
100.16 g/mol
3.00 mL
0.81 g/mL
82.14 g/mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 4 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

