Course: Chem 120 - Organic Che x O LOCAL clicker x b My Questions | bartleby i cpp.edu/-Isstarkey/local/clicker/bonding/acid-base.html Which is the stronger acid? Explain briefly. Which is the stronger acid? Explain briefly. H20 NH3 но- -CN но- но- -CH A B A) H,O is the stronger acid because HO is more stable than H,N. - CN A) A is the stronger acid because 110- is more stable. B) H,0 is the stronger acid beeause HO is less stable than H,N". B) A is the stronger acid because °o CN is more stable. C) B is the stronger acid because H0--CH, is more stable. D) B is the stronger acid because ®o o is more stable. C) NH, is the stronger acid because H,N is more stable than HO", D) NH, is the stronger acid because H,N' is less stable than HO". E) It's impossible to predict acid strength without pK, data. E) It's impossible to predict acid strength without pk, data. Arrange the following hydrogens in the order of DECREASING pK, values (from least acidic to most acidic). Arrange the following compounds in order of INČREASING acid strength (from least acidic to most acidic). OH- 0=c-C-CH-CF3 OH 1 A)I>Il > III OH HCI CH,OH II III B) III > II >1 C) II > III>I A) III < II < I B) III < II>II E) II>I> III C) I < III < II D) II < II < I E) I < II < III
Course: Chem 120 - Organic Che x O LOCAL clicker x b My Questions | bartleby i cpp.edu/-Isstarkey/local/clicker/bonding/acid-base.html Which is the stronger acid? Explain briefly. Which is the stronger acid? Explain briefly. H20 NH3 но- -CN но- но- -CH A B A) H,O is the stronger acid because HO is more stable than H,N. - CN A) A is the stronger acid because 110- is more stable. B) H,0 is the stronger acid beeause HO is less stable than H,N". B) A is the stronger acid because °o CN is more stable. C) B is the stronger acid because H0--CH, is more stable. D) B is the stronger acid because ®o o is more stable. C) NH, is the stronger acid because H,N is more stable than HO", D) NH, is the stronger acid because H,N' is less stable than HO". E) It's impossible to predict acid strength without pK, data. E) It's impossible to predict acid strength without pk, data. Arrange the following hydrogens in the order of DECREASING pK, values (from least acidic to most acidic). Arrange the following compounds in order of INČREASING acid strength (from least acidic to most acidic). OH- 0=c-C-CH-CF3 OH 1 A)I>Il > III OH HCI CH,OH II III B) III > II >1 C) II > III>I A) III < II < I B) III < II>II E) II>I> III C) I < III < II D) II < II < I E) I < II < III
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter4: Polar Bonds, Polar Reactions
Section: Chapter Questions
Problem 28E: The following are equivalent ways of asking about the acidity of an H atom: • What is the most...
Related questions
Question
Please help me with all of this pretty please :(
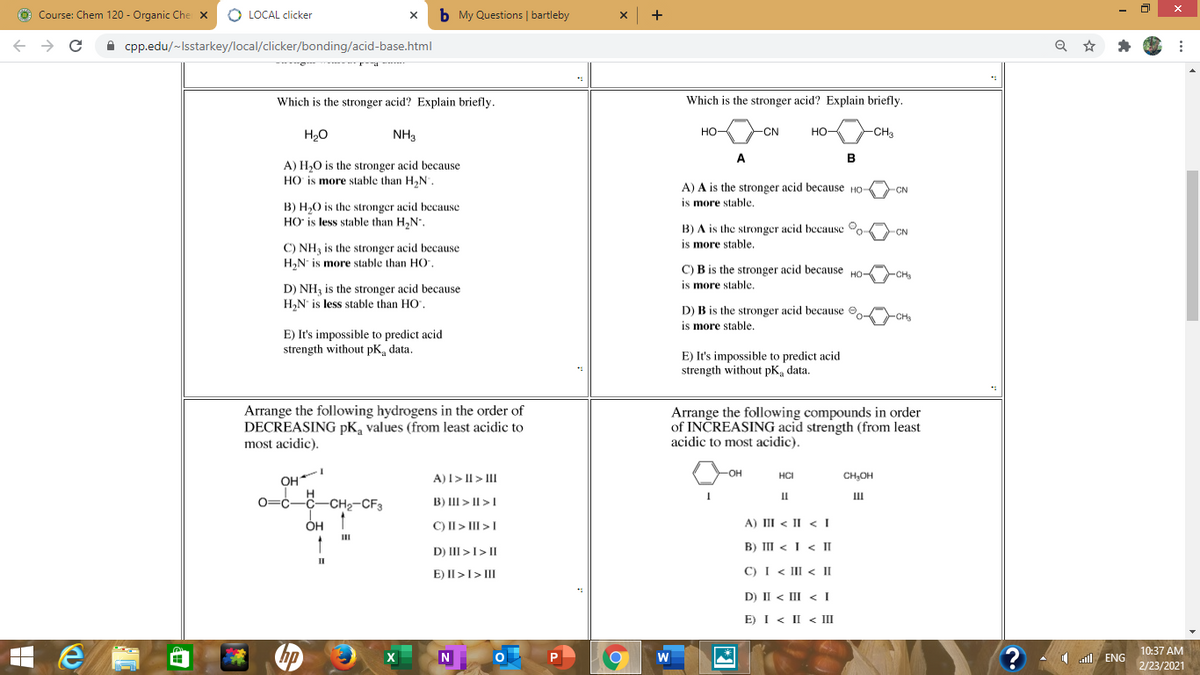
Transcribed Image Text:O Course: Chem 120 - Organic Che x
O LOCAL clicker
b My Questions | bartleby
->
A cpp.edu/~Isstarkey/local/clicker/bonding/acid-base.html
Which is the stronger acid? Explain briefly.
Which is the stronger acid? Explain briefly.
H20
NH3
но
-CN
HO
CHa
A
B
A) H,0 is the stronger acid because
HO is more stable than H,N.
A) A is the stronger acid because HO
is more stable.
CN
B) H,0 is the stronger acid because
HO' is less stable than H,N.
B) A is the stronger acid because o
is more stable.
-CN
C) NH3 is the stronger acid because
H,N is more stable than HO,
C) B is the stronger acid because HO-
CH
is more stable.
D) NH3 is the stronger acid because
H,N' is less stable than HO".
D) B is the stronger acid because O
is more stable.
CHs
E) It's impossible to predict acid
strength without pK, data.
E) It's impossible to predict acid
strength without pK, data.
Arrange the following hydrogens in the order of
DECREASING pK, values (from least acidic to
most acidic).
Arrange the following compounds in order
of INČREASING acid strength (from least
acidic to most acidic).
HO-
OH
0=c-C-CH2-CF3
A)I> II > III
HCI
CH,OH
II
II
B) III > II >1
ÓH
C) II> III >I
A) III < II < I
II
B) III < I < II
D) III >I> II
E) II >I> III
C) I < III < II
D) II < III < I
E) I < II < III
10:37 AM
hp
1 l ENG
2/23/2021
図
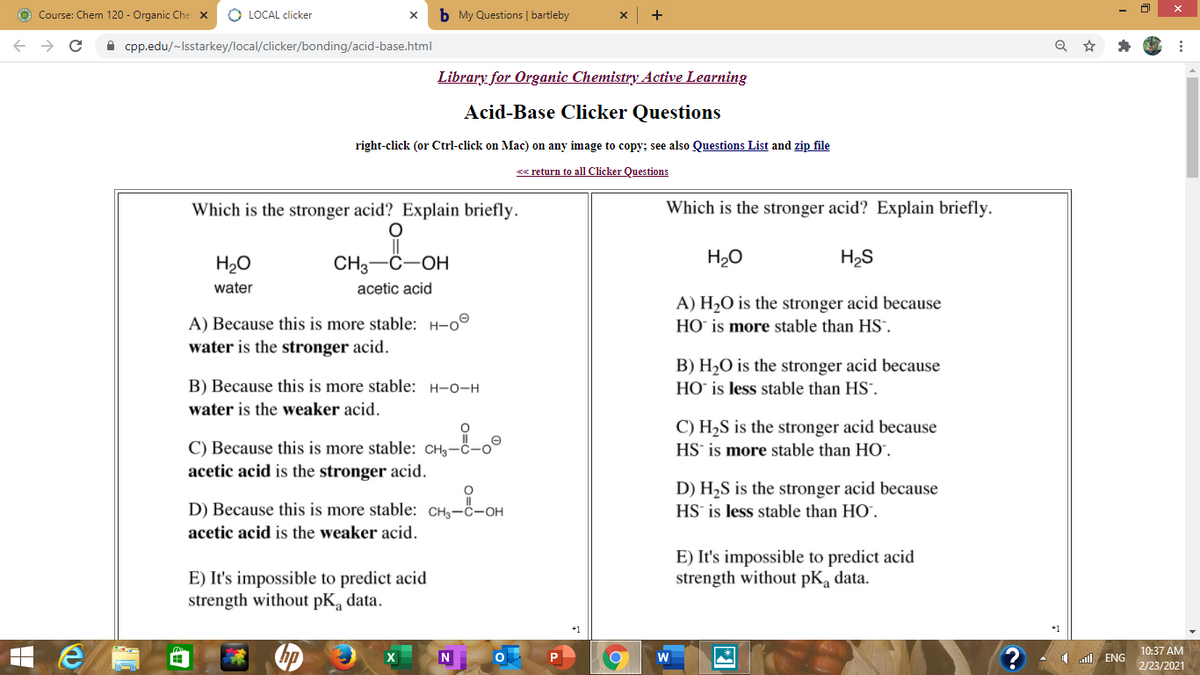
Transcribed Image Text:ở Course: Chem 120 - Organic Che X
O LOCAL clicker
b My Questions | bartleby
->
A cpp.edu/~Isstarkey/local/clicker/bonding/acid-base.html
Library for Organic Chemistry Active Learning
Acid-Base Clicker Questions
right-click (or Ctrl-click on Mac) on any image to copy; see also Questions List and zip file
< return to all Clicker Questions
Which is the stronger acid? Explain briefly.
Which is the stronger acid? Explain briefly.
H20
CH3-C-OH
H2O
H2S
water
acetic acid
A) H2O is the stronger acid because
HO` is more stable than HS".
A) Because this is more stable: H-o°
water is the stronger acid.
B) H2O is the stronger acid because
HO` is less stable than HS.
B) Because this is more stable: H-o-H
water is the weaker acid.
C) H2S is the stronger acid because
HS' is more stable than HO.
C) Because this is more stable: CHg-c-o°
acetic acid is the stronger acid.
D) H2S is the stronger acid because
D) Because this is more stable: CH3-ċ-OH
HS is less stable than HO`.
acetic acid is the weaker acid.
E) It's impossible to predict acid
strength without pKa data.
E) It's impossible to predict acid
strength without pK, data.
10:37 AM
illl ENG
2/23/2021
因
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
