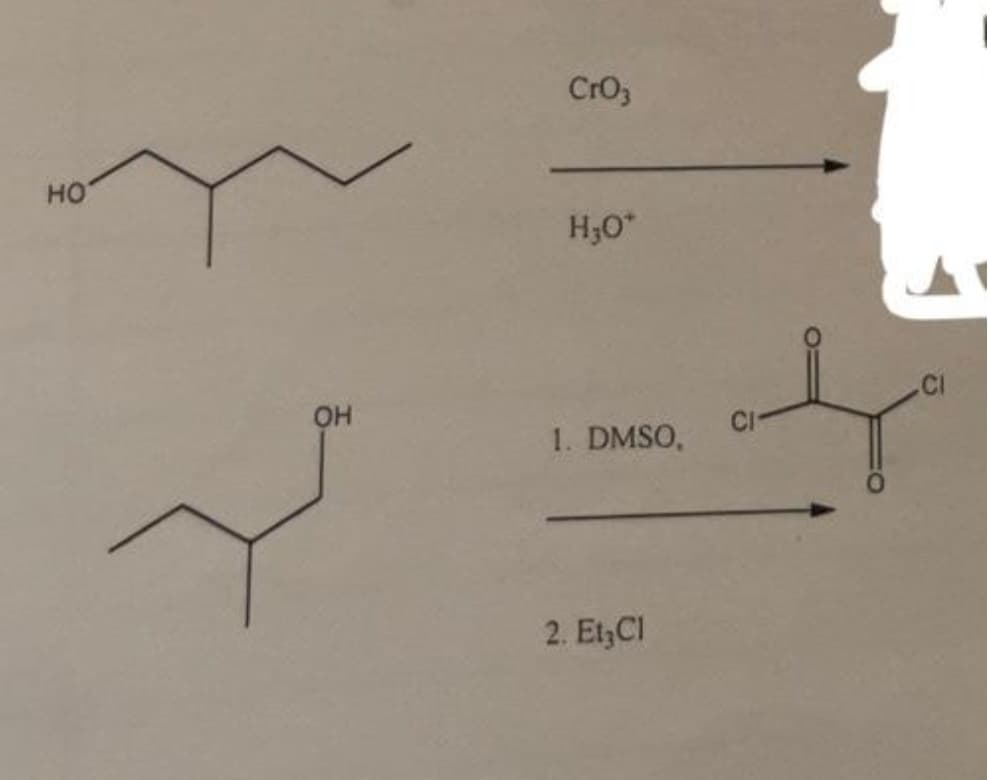
Q: Bài 33: Hoàn thành các phản ứng sau NABH, COOCH, [1] LIAIH, HO [2] H,0 c. (CH,),N' a. CH,OH [1]…
A: (a) When ketonic group and ester is reduced by sodium borohydride, NaBH4 then due mild reducing…
Q: CI с. AICI3 1) HNO3, H2SO4 d. 2) Zn / HCI CI 1) СО, НСI е. 2) SO3, H2SO4
A: Acetanilide -HN- COCH3 group is ortho – para directing. Because of steric hinderance and the…
Q: 1. MeCl, AICI3 2. NBS, ROOR 3. PhSNa
A: Since you have posted multiple questions, we are entitled to answer the first only. Please repost…
Q: 1. 2 Li, Et,0 2. 0.5 equiv. Cul. 3. CH3(CH2)½Br -CI
A:
Q: 7 1 5.3Li +1H → 2 He+ O a. He O b. 2, 1H O C. 3 O On d. 4 4Be
A: A radioactive reaction can be balanced by summing up the atomic number and mass number on both sides…
Q: 14 helit ds pod 1. B gh 2.1-buOK S. BH, in THE 4 Ho, NaOH (8) BH (A) 114 (B) (C) (D) OH
A: Since you have posted multiple questions, we are entitled to answer the first only. 24) The reaction…
Q: radidactive research, particulary in studies mass (in mg) oudsoud g of Na, 32PO, after 40.0 days?…
A: Radioactive decay follow first order kinectics. Half life = 14.3 days
Q: NABHz CN NH3, Hg ot H3 Ot Por3, KoC (cH3h. 0s \. BH3 2. +202, O4- crqele DCC
A:
Q: Is this the correct synthesis of the proposed reaction?
A: Based on the given reaction, all the steps seems to be fine except the oxidation of methyl group to…
Q: 1. 2 Li, Et,0 2. 0.5 equiv. Cul 3. CH3(CH2)2Br CI
A: The major organic product for the given reaction scheme is to be determined. Alkyl halide on…
Q: Cacs) t Sn Cl4 (ag) Cassi t Cu SO4(a)- Mgrs)+ Z,(NC), (u) > -> Mg(s) + SnCl4(a8)→ Zn(S)t SnCl4(a%)→…
A: Complete reaction A reaction equation where all ions(main chemical species and spectator ions) are…
Q: 12, land Fe2", Fe3- 2(3) + 2FE²-(aq) eTextbook and Media Hint Mg, Mg2* and Cr, Cr3+ Co. Co2 and…
A:
Q: Complete the following shorthand notations: ? (р, а) Li 31P (a, ?) ES 15 242 66Cm (α, n) ? 25Mg…
A:
Q: What do the peaks of the mass spec of C6H10 show?
A: The structure of the unknown molecules can be obtained by analyzing their spectroscopical results…
Q: (27)Al (p,y) (28)Si (40)Ar (n,B') (41)K (14)N (a,p) (17)o (200)Hg (p,a) (6)Li (n,a)
A: Nuclear reaction:
Q: 1A 8A H 2A 3A 4A SA 6A 7A He Li Be BCNOF Ne Na Mg 38 48 5B 6B 7B - 8B - 18 28 AI Si P S CI Ar K Ca…
A: Electronegativities increase from left to right across the periodic table, because of the increase…
Q: oni. 3. r MgBr a. 1. 2. H20 1. Zeq. a My Br HzO
A:
Q: Prxn O Pb2*(aq) + SO42-(aq) PbSO4(s) 5 C(s) + 4 KNO3(s) 5 CO2(g) + 2 N2(g) + 2 K20(s) O O3(g) –…
A:
Q: 4. Rb + SgRb,S 5 NH, + HCI → NH,CI 6 Li+H,0- LIOH + H,
A: Since, Balanced equation means number of each atom in both side are present in equal number.
Q: Sstepr Og Meé Nhe fh
A:
Q: 1A 8A H 2A 3A 4A 5A 6A 7A He Li Be B C NO F Ne Na Mg 38 48 5B 6B 7B - 8B - 1B 28 A1 si PS CI Ar K Ca…
A: Ionic compounds are formed by ions. The metal atom loses electrons and forms cations and the…
Q: H,SO, (aq) + Mg(OH), (aq) (aq) (1) + 2.
A:
Q: 1. excess KNH2, heat 2. O3 3. Н-О CI CI. 1. NaNH2 2. isobutyl iodide 3. На, Pt 1.9-BBN 2. HаОг, NaOн
A: In an organic reaction reactants reacts in the presence of reagents to form products. Alkyne…
Q: (c) 1. O, -78 °C ? 2. Zn, HOAC
A: O3 is oxidizing agent . it oxidised alkene to carbonyls.
Q: Bno O, H 1. THE -78 to 0C NH H. H. 2. THE 1h, rt. -OTMS NH 85% 3.
A: Step 1: N- actylation followed by carbanion addition at imine carbon. Michael type addition…
Q: Give a clear explanation handwritten answer...
A: Calculations have been done in the following step.
Q: F[OH-] = 1.45 x 102, find the pOH.
A: pOH is the measure of acidity or basicity. Lesser the value of pOH more basic is the solution. pOH…
Q: Rb Sr 8 As 50 3
A:
Q: 7. ZnCI Pd CI 8. ОН CH»l2 Cu-Zn
A:
Q: With the information given in the table, what would the percent by mass of the reacted sodium…
A: Dear student, we dont answer graded questions. We answer only homework help questions. Please post…
Q: Q/ Prepare 0.1 N and 0.1 M of 98% H,SO4 in 250 ml. M.wt of H,SO, = 98 g/mol, and its Specific…
A: Let 100% of the solution of H2SO4 is equivalent to 100 g. Thus, 98% H2SO4 is equivalent to 98 g…
Q: МОМОМОМ OMOM BF3-OEt, A RO" OTBS ÓTBS 2 R= H (52%) 3 R= MOM (30%)
A:
Q: (d) 0.15 MCa(CIO3)2 lons: CIO,2- O CIO- CIO2- O Ca2+ O Ca* O CIO3 [cation*] = i M [anion] = i M
A: Cation is a species containing more of protons than electrons. Thus, they possess a positive charge.…
Q: PE RCENT C 19) Determin the the fo a) C,„H2 12 22
A: mass of C = 12 g/mol
Q: NaCI Not lonic v NO2 Cus CO PF3 MgF2 >
A:
Q: 1A 8A H 2A 3A 4A SA 6A 7A He Li Be BCNOFNE Na Mg 38 4B 5B 6B 7B 8B - 1B 26B AI si P S CI Ar K Ca sc…
A: The question is based on the concept of atomic properties. we have to write isoelectronic species of…
Q: jatot pug eleeron lount ) [ eulbpy), CNO) ce] (i) 2+ 3+
A:
Q: он 1. Pd L, Et,N, MECN Me HO 2. NABH,, MEOH çI . ElN, CH,Cl, 3. Me
A: The first step of the reaction is the Heck coupling reaction and proceeds via following mechanism :…
Q: 2- а. Ст (aq) + Cl>(9) + СrО7? (ад) + CГ (аq) V b. Ni*+ (aq) + Ba(s) — Ва* (аq) + Ni(s) V ||
A: Answer:- this question is answered by using the simple concept of calculation of standard cell…
Q: 2HW3.pdf ge 2 of 3 n) o) p) Br CI -Br v) w) zt x Q Search 오
A: Name the following compounds, need part N, V, and O.
Q: + KNO, → NâNO, + RUH KOH CH, + 0, → Co, + 2 H0 on Fe + Caso + Mg(OH) NH OH + HBr 3 HÓ + NH BI NaBr →…
A:
Q: RÜNO, BeF2 à + 4) Be(NO3)2 + RbF
A: The question is based on the concept of chemical reaction. a reaction has been provided. we have to…
Q: 1A BA H 2A 3A 4A SA 6A 7A He u Be BCNO F Ne Na Mg 3e48 58 68 78 r 8e- 18 28 R Si S CI Ar KCa sc TI V…
A: Groups and periods The vertical columns in periodic table are called groups. The horizontal rows in…
Q: अ @मांयंपल H is Pa=4.81 Pe=4'05 bak , 6= 4.63ba stendertonange in gibas fee enesy च अंट ? bak, P=451…
A:
Q: 7) ( HO 8) Me O,-78 °C then Zn, CH,COH
A:
Q: एमरखन 120 लो गाणकीं aso 240 1ons. Ble0 00 12ut the Oea albue n the Giuph1gee
A:
Q: मुजठुन प्रूककोटेट व जेह्छकाइ नेणकोत (onpoand डे कa तेट किष्र कट निनणास जटवलेीनक्त- 2CHY(8)+ 2NH3cg)t…
A:
Q: Draw the organic products formed in each reaction.
A: a. Ans: On reacting excess of NH3 with a haloalkane gives a primary amine as the predominant…
Q: Smallest Largest 1A H 2A neon Li Be Na Mg 38 4B 5B 6B 7B8B K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Rb…
A: Given element are Neon, Krypton, helium, argon We have to arrange the given atom in increasing…
Q: LAu Br2 0 Na 0DLDA 2)Br : )LDA )CH3I CUD 70NA 2) CH3I O HCON 2)Br H30t D Na H Its0+ 2Br
A: Product of the reaction has to be identified.(a)Acid-Catalysed Bromination of Ketones: ketones…
Step by step
Solved in 3 steps with 1 images


